Articles
- Page Path
- HOME > J Mov Disord > Volume 16(3); 2023 > Article
-
Letter to the editor
Pallidal Deep Brain Stimulation for Refractory Celiac-Related Myoclonus -
Jinyoung Youn1,2,3,4
 , Elizabeth Slow3,4
, Elizabeth Slow3,4 , Robert Chen3,4,5
, Robert Chen3,4,5 , Andres M. Lozano6,7
, Andres M. Lozano6,7 , Alfonso Fasano3,4,6,8
, Alfonso Fasano3,4,6,8

-
Journal of Movement Disorders 2023;16(3):325-327.
DOI: https://doi.org/10.14802/jmd.23006
Published online: June 9, 2023
1Department of Neurology, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea
2Neuroscience Center, Samsung Medical Center, Seoul, Korea
3Edmond J. Safra Program in Parkinson’s Disease, Morton and Gloria Shulman Movement Disorders Clinic, Toronto Western Hospital, University Health Network, Toronto, ON, Canada
4Division of Neurology, University of Toronto, Toronto, ON, Canada
5Division of Brain, Imaging and Behavior, Systems Neuroscience, University Health Network, University of Toronto, Toronto, ON, Canada
6Krembil Brain Institute, Toronto, ON, Canada
7Division of Neurosurgery, University of Toronto, Toronto, ON, Canada
8Center for Advancing Neurotechnological Innovation to Application (CRANIA), Toronto, ON, Canada
- Corresponding author: Alfonso Fasano MD, PhD Movement Disorders Center, Toronto Western Hospital, 399 Bathurst St, 7McL410, Toronto M5T 2S8, ON, Canada / Tel: +1-416-603-5800 / Fax: +1-416-603-5004 / E-mail: alfonso.fasano@uhn.ca
Copyright © 2023 The Korean Movement Disorder Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- Dear Editor,
- Gluten-related disorders are a broad spectrum of immunemediated disorders, and celiac disease (CD) is the most common and well-known among these disorders. CD can present with various neurological issues, with ataxia being the most common neurological symptom and myoclonus being the most common movement disorder after ataxia [1]. Although most neurological symptoms may improve or stabilize with a gluten-free diet, myoclonus is less responsive [2]; thus, an effective treatment for CD-related myoclonus is needed. Given the notion that deep brain stimulation (DBS) of the globus pallidus pars interna (GPi) can improve conditions such as myoclonus-dystonia (MD) or post-hypoxic myoclonus (PHM) [3,4], GPi DBS might be a treatment option for CD-related myoclonus. Herein, we report the case of a patient with CD-related myoclonus ataxia who showed partial benefit from GPi DBS.
- A 60-year-old man presented with a 6-month history of jerky movement of the left arm in the absence of any other symptoms (previously reported as case 1) [5]. He had a several-year history of diarrhea but no prior diagnosis of CD, and he denied any family history of CD, myoclonus, or balance problems. Although he was a heavy alcohol user at presentation, all his symptoms worsened even after he quit drinking. The diagnostic workup included brain magnetic resonance imaging, which showed mild diffuse atrophy; negative autoimmune/metabolic screening results (antinuclear antibodies, extractable nuclear antigen panel, antineutrophil cytoplasmic antibodies, thyroid-stimulating hormone, anti-thyroglobulin antibody, and anti-thyroid peroxidase); and negative results in screening for a panel of autoimmune and paraneoplastic antibodies (against AMPA, amphiphysin, CV2.1, DPPX, GABAB receptor, GAD65, Hu, NMDA receptor, PNMA2, recoverin, Ri, SOX1, Titin, Tr, VGKC, Yo, and Zic4). CD was diagnosed on the basis of flattened villi on endoscopy and elevated serum tissue transglutaminase immunoglobulin A antibodies of 23 U/mL (normal range up to 10 U/mL). Electrophysiological studies demonstrated findings consistent with cortical myoclonus, such as electropositive polyspikes equally represented between Cz and C4, giant median nerve somatosensory evoked potential, and cortical potentials 11 ms before myoclonus in the deltoid muscle with jerk-locked back-averaging as reported previously [5].
- The patient started a gluten-free diet and various medication trials, including perampanel (4 mg/day), clonazepam (2 mg/day), valproate (2,000 mg/day), levetiracetam (3,000 mg/day) and mycophenolate mofetil (700 mg/day), but there was no benefit, and myoclonus spread to his legs. The patient also noticed a progressive balance impairment even after quitting drinking, and he required a walker one year after disease onset. GPi DBS was offered 2.5 years after disease onset when he had generalized myoclonus mostly affecting the left arm and both legs owing to refractory CD (Supplementary Video 1 in the online-only Data Supplement). He also demonstrated mild dysmetria, greater on the left, and mild dystonia of the neck and right arm. He could stand with a wide base but was unable to walk.
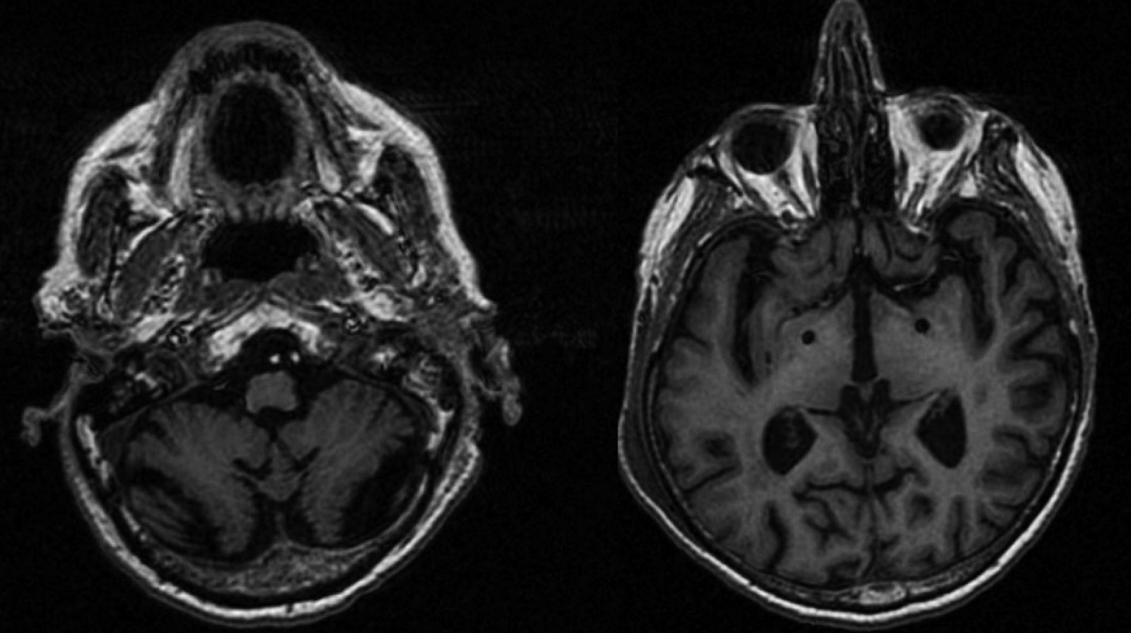
- GPi DBS surgery was performed (Medtronic Activa PC, Dublin, Ireland, connected to 3387 leads) (Figure 1). After surgery, his clinical course was complicated by pneumonia and lower leg weakness due to radiculopathy as confirmed by electrophysiological evaluation, but he recovered gradually. Six months after surgery, his myoclonus was slightly better with stimulation. The total score of the Unified Myoclonus Rating Scale changed from 90 to 83, and the score of action myoclonus in particular changed from 56 to 53 [6]. In addition to the improvements observed in our evaluation, the patient reported improvement in using his arms after pallidal stimulation. However, there were no improvements in imbalance or gait, but these symptoms mainly resulted from ataxia, even though myoclonus was also present (Supplementary Video 2 in the online-only Data Supplement). The optimal parameters were double monopolar stimulation with 3.0 V on contacts 0 and 1 on the right and monopolar stimulation with 2.5 V on contact 8 on the left, while a pulse width of 60 µs and frequency of 140 Hz were kept the same on both sides. The patient did not have any stimulation-induced side effects.
- Although limited to only a single case, our experience shows that myoclonus can partially improve with GPi DBS, which could be considered a treatment option for these patients, for whom myoclonus is typically resistant to conservative treatment approaches [2]. Among the myoclonic disorders, MD is the most common myoclonus disorder responsive to pallidal stimulation [4], in keeping with the observation that pallidal neuronal firing is modulated by the occurrence of myoclonus [4]. While the myoclonus in MD is subcortical, the myoclonus in CD is of cortical origin [5,7]. Based on the different responses to GPi DBS, pallidal stimulation could be effective mainly for subcortical myoclonus, although its effect on cortical myoclonus is still unclear. GPi DBS is also effective for refractory myoclonus in PHM, although the mechanism of pallidal stimulation in PHM is still poorly understood [3,7]. The myoclonus in PHM is known as mainly cortical myoclonus, but patients with PHM might often present both types of myoclonus [3]. Unlike MD and PHM, myoclonus in CD originates from the cortex, as electrophysiologically confirmed in our patient [5]. One study found that pallidal stimulation with low frequency, which was less effective than stimulation with high frequency in our patient, modulates the uptake of cortical glucose in PMH [7], and this result may explain the partial improvements observed in our patient. Therefore, there could be a common pathway for myoclonus per se, and pallidal stimulation could be effective for both cortical and subcortical myoclonus, although the benefits would be limited for cortical myoclonus compared to subcortical myoclonus.
- Considering the modest improvement in our patient, we can conclude that GPi DBS may be of limited utility for cortical myoclonus compared to subcortical myoclonus. In fact, despite the improvement in myoclonus, especially in the upper limbs, our patient’s gait and balance did not improve. Considering that, compared to the upper limbs, the patient had more severe myoclonus in the lower limbs, where weakness and ataxia were also present, pallidal stimulation might not have a sufficient symptomatic effect on the lower limbs. Although our patient was a heavy drinker, ataxia became prominent and worsened even after he had quit drinking. Gait ataxia–the most common neurological issue in CD–is conceivably resistant to DBS. In conclusion, this is the first case report of GPi DBS being used in the treatment of CD-related myoclonus, and an improvement in myoclonus in the upper limbs was observed.
Supplementary Material
Video 1.
Video 2.
-
Ethics Statement
This study was approved by the institutional review board of University Health Network (15-8777). Our patient provided written informed consent and authorization for the videotaping and publication of his videotape.
-
Conflicts of Interest
The authors have no financial conflicts of interest.
-
Funding Statement
This study in part was funded by the University of Toronto and University Health Network Chair in Neuromodulation (AF).
-
Author contributions
Conceptualization: Alfonso Fasano. Data curation: all authors. Formal analysis: all authors. Funding acquisition: Alfonso Fasano. Investigation: all authors. Methodology: all authors. Project administration: Alfonso Fasano. Supervision: Elizabeth Slow, Robert Chen, Andres M. Lozano, Alfonso Fasano. Writing—original draft: Jinyoung Youn, Alfonso Fasano. Writing—review & editing: Jinyoung Youn, Alfonso Fasano.
Notes
- 1. Vinagre-Aragón A, Zis P, Grunewald RA, Hadjivassiliou M. Movement disorders related to gluten sensitivity: a systematic review. Nutrients 2018;10:1034.ArticlePubMedPMC
- 2. Zis P, Hadjivassiliou M. Treatment of neurological manifestations of gluten sensitivity and coeliac disease. Curr Treat Options Neurol 2019;21:10.ArticlePubMedPDF
- 3. Ramdhani RA, Frucht SJ, Kopell BH. Improvement of post-hypoxic myoclonus with bilateral pallidal deep brain stimulation: a case report and review of the literature. Tremor Other Hyperkinet Mov (N Y) 2017;7:461.ArticlePubMedPMC
- 4. Magarinos-Ascone CM, Regidor I, Martinez-Castrillo JC, Gómez-Galán M, Figueiras-Méndez R. Pallidal stimulation relieves myoclonus-dystonia syndrome. J Neurol Neurosurg Psychiatry 2005;76:989–991.ArticlePubMedPMC
- 5. Swinkin E, Lizárraga KJ, Algarni M, Garcia Dominguez L, Baarbé JK, Saravanamuttu J, et al. A distinct EEG marker of celiac disease-related cortical myoclonus. Mov Disord 2021;36:999–1005.PubMed
- 6. Frucht SJ, Leurgans SE, Hallett M, Fahn S. The unified myoclonus rating scale. Adv Neurol 2002;89:361–376.PubMed
- 7. Kim MJ, Park SH, Heo K, Chang JW, Kim JI, Chang WS. Functional neural changes after low-frequency bilateral globus pallidus internus deep brain stimulation for post-hypoxic cortical myoclonus: voxel-based subtraction analysis of serial positron emission. Brain Sci 2020;10:730.ArticlePubMedPMC
REFERENCES
Figure & Data
References
Citations

- Pallidal deep brain stimulation for patients with myoclonus-dystonia without SGCE mutations
Jun Ikezawa, Fusako Yokochi, Ryoichi Okiyama, Ayako Isoo, Takashi Agari, Tsutomu Kamiyama, Akihiro Yugeta, Maya Tojima, Takashi Kawasaki, Katsushige Watanabe, Satoko Kumada, Kazushi Takahashi
Journal of Neurology.2024;[Epub] CrossRef
Comments on this article
 KMDS
KMDS
 E-submission
E-submission
 PubReader
PubReader ePub Link
ePub Link Cite
Cite