Analysis of Semiology, Lesion Topography and Treatment Outcomes: A Prospective Study on Post Thalamic Stroke Holmes Tremor
Article information
Abstract
Objective
Holmes tremor (HT) comprises rest, postural and intention tremor subtypes, usually involving both proximal and distal musculature. Perturbations of nigro-striatal pathways might be fundamental in the pathogenesis of HT along with cerebello-thalamic connections.
Methods
Nine patients with an HT phenotype secondary to thalamic stroke were included. Epidemiological and clinical records were obtained. Structural and functional brain imaging were performed with magnetic resonance imaging (MRI) or computed tomography (CT) and positron emission tomography (PET), respectively. Levodopa was administered in sequentially increasing dosage, with various other drugs in case of inadequate response. Longitudinal follow-up was performed for at least three months. The essential tremor rating assessment scale (TETRAS) was used for assessment.
Results
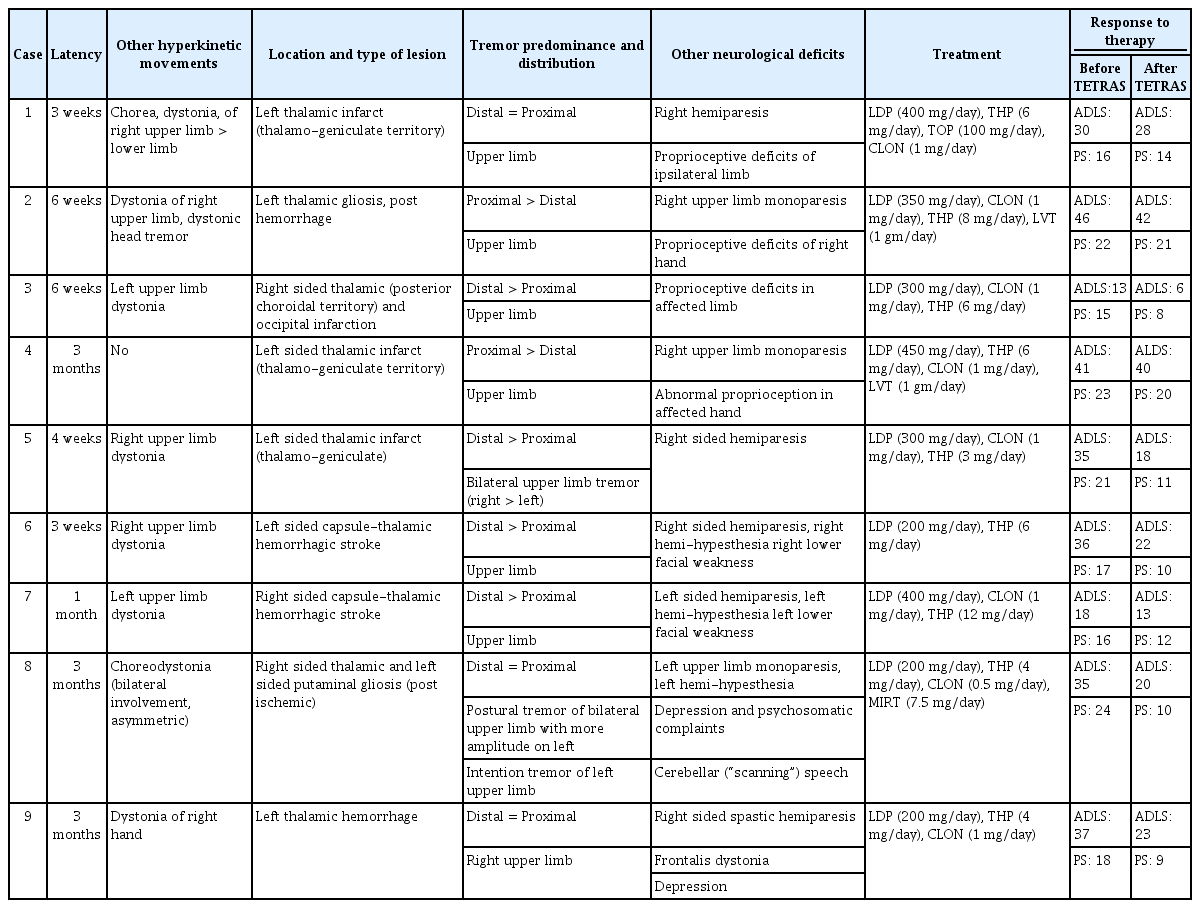
The mean latency from stroke to tremor onset was 50.4 ± 30.60 days (range 21–90 days). Dystonia was the most frequently associated hyperkinetic movement (88.8%). Tremor was bilateral in 22.2% of participants. Clinical response was judged based on a reduction in the TETRAS score by a prefixed value (≥ 30%), pertaining to which 55.5% (n = 5) of subjects were classified as responders and the rest as non-responders. The responders showed improvement with significantly lower doses of levodopa than the remaining nonresponders (240 ± 54.7 mg vs. 400 ± 40.8 mg; p = 0.012).
Conclusion
Although levodopa is useful in HT, augmenting the dosage of levodopa beyond a certain point might not benefit patients clinically. Topography of vascular lesions within the thalamus might additionally influence the phenomenology of HT.
INTRODUCTION
More than a century after the original description by Gordon Holmes, the pathophysiology of Holmes tremor (HT) remains an enigma. Previous nomenclature such as “rubral tremor”, “thalamic tremor”, and “mesencephalic tremor” have been largely abandoned owing to numerous studies that have elucidated the network of various regions in the brain, which might contribute to the genesis of HT [1-3]. This “network hypothesis” proposes the concurrent functional defect of both cerebello-thalamic or dentato-rubral-thalamic and dopaminergic nigrostriatal pathways as requisite for the genesis of such a tremor [3,4]. The most recent consensus is to consider HT as a clinical syndrome that comprises rest, postural and intention tremor subtypes, usually involving both proximal and distal musculature, at a low frequency (< 5 Hz) [5].
Vascular lesions are by far the most common etiological cause of HT, with a variable latency from initial insult to onset of tremor ranging from as low as one week to more than a decade [3,6]. This thereby underscores the role of neuronal plasticity and the reorganization of different neuronal circuits within proposed brain regions, the intricacies of which are presently beyond our understanding. Irrespective of the etiology, however, the therapeutic response to pharmacological agents, including dopaminergic medication, has been heterogeneous [7]. In contrast, the response to deep brain stimulation (DBS) surgery has been more uniform and encouraging [2,8].
In concurrence with Nsengiyumva et al. [6], we believe that the anatomical localization of the HT lesion harbors significance with regard to its therapeutic prospects. Herein, we elucidate a series of patients who presented with HT, with variable latency following a thalamic vascular insult, as well as look at the natural history of such lesions and therapeutic response on follow-up.
MATERIALS & METHODS
The investigators included patients from the outpatient department (OPD) of a tertiary care neurology institute in Kolkata, India, between July 2021 and June 2022. A total of approximately 230 patients presenting with tremor were screened in the movement disorder clinic within the stipulated period, of which 11 patients with the HT phenotype were identified (4.78%). One patient was excluded because brain imaging could not be performed and was lost to follow-up. Nine patients had thalamic lesions, while one showed a brainstem lesion. For the present study, only patients with thalamic lesions were included. The diagnosis of HT was clinical, based on the definition in the Consensus statement of the International Parkinson disease and Movement disorder society [5].
All the patients were critically examined, and their findings were analyzed by three neurologists (AKD, AM and AB), two of whom (AM and AB) are movement disorder specialists at the same institute. All patients were followed up for at least three months after the initial evaluation and start of therapy. The following data were collected: age, sex, latency from initial insult to onset of tremor, neurological and radiological features, therapy provided, and response.
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee of Institute of Post Graduate Medical Education & Research (IPGME & R), Kolkata (IPGME&R/IEC/2023/290) and with the 1975 Helsinki declaration and its later amendments. Written informed consent was duly obtained from the patients included in the study. This included consent to 1) participate in the study, 2) for video-recording, and 3) presentation or publication of videos for academic purpose.
Treatment of all patients was started within one week of presentation to the movement disorder clinic. Tremor severity was quantified as per the essential tremor rating assessment scale (TETRAS). Patients were treated with sequentially increasing doses of levodopa with other add-on drugs as necessitated by response and tolerability. Patients were classified into responders and nonresponders based on a predetermined cutoff reduction in the performance score (PS) subset of TETRAS by 30% or more. The longitudinal follow-up duration was at least three months, and patients were independently assessed by all three investigators in the same clinical setting using TETRAS; however, a common consensus was agreed upon by all the investigators regarding initial diagnosis and outcomes.
Brain imaging was performed with either magnetic resonance imaging (MRI) using a 3 Tesla scanner or computed tomography (CT) using a 128-slice machine. Functional imaging was performed with a fluorodeoxyglucose positron emission tomography (FDG-PET) scan. MRI sequences used for evaluation were axial, coronal, and sagittal T1, T2 weighted images, axial fluid attenuated inversion recovery (FLAIR) sequence, axial diffusion weighted image (DWI), axial apparent diffusion coefficient (ADC) maps, axial gradient echo (GRE) sequence and time of flight (TOF) angiography images of cerebral vasculature. Images were assessed independently by a neuroradiologist (S.M.) who was blinded to the clinical features of each patient.
Data were organized in Microsoft Excel (Microsoft, Redmond, WA, USA) and analyzed in IBM SPSS v.23 (IBM Corp., Armonk, NY, USA). Categorical variables are expressed as frequencies and percentages. Continuous variables are expressed as the mean and standard deviation. Comparisons between groups were performed using the Mann‒Whitney U test after checking for assumptions.
RESULTS
Patient demographics
Nine patients were included for analysis, all of whom were of Bengali ethnicity. The cohort comprised 4 male and 5 female patients. The mean age of presentation was 53.3 ± 8.4 years (range 40–70 years). The mean latency from initial vascular insult to onset of tremor was approximately 50.4 ± 30.60 days (range: 21–90 days) (Table 1).
Etiology and radiological features
Fifty-five percent of the patients (n = 5) presented following ischemic strokes, while the remaining patients had sequelae of hemorrhagic stroke, all involving the thalamus. Among the ischemic thalamic lesions (n = 5), thalamoperforating and/or posterior choroidal circulation was implicated in n = 3 (60%); the remaining patients (n = 2; 40%) had affection of the thalamogeniculate vasculature. Involvement of the occipital lobe and putamen was noted in one patient each (Figure 1).
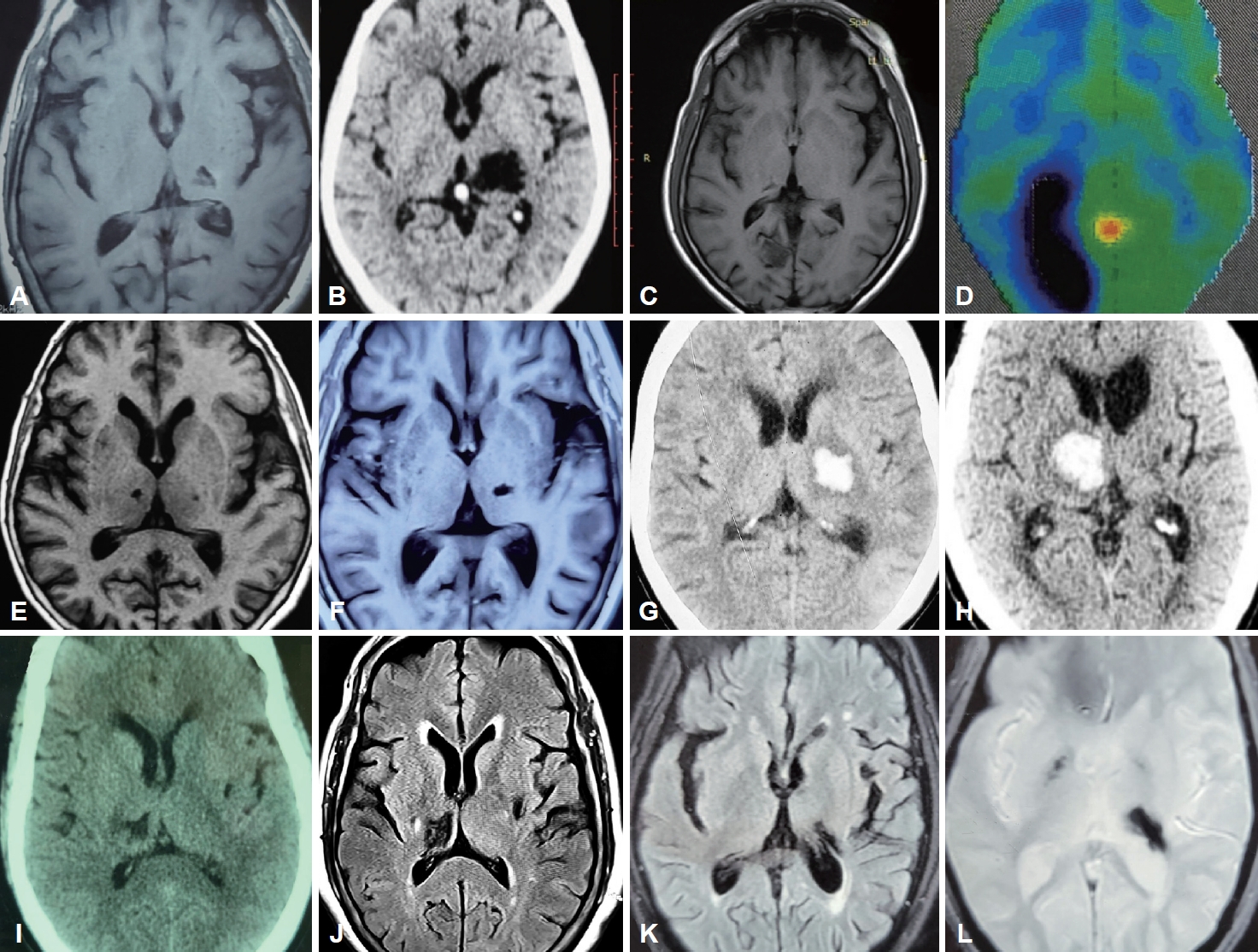
Summary of neuroimaging findings of all patients in the series. A: Axial T1 weighted MRI image of Patient 1 shows gliotic foci in the left thalamus. B: Axial CT scan brain of Patient 2 shows a encephalomalacia of the thalamus on the left side. C: Axial T1 weighted MRI brain of Patient 3 shows gliosis of the right side of the thalamus and occipital lobe. D: Corresponding axial FDG-PET image of the same patient showing severe hypometabolism in the right occipitoparietal lobe and adjacent parietal cortex, with mild hypometabolism in the bilateral inferior frontal, bilateral temporal cortex and right thalamus. E: Axial T1 weighted MRI image of Patient 4 shows two gliotic lesions in the bilateral thalamus, lesion on left side being the presumable culprit. F: Axial T1 MRI brain of Patient 5 shows a small hypointense lesion on the left side of the thalamus and discrete hypo-intensities in the right globus pallidus. G: Axial CT scan of the brain of Patient 6 shows a hematoma encroaching in the left capsule-thalamic region. H: Axial CT scan of the brain of Patient 7 shows a hematoma of the right side of the thalamus with mass effect. I: Axial CT brain scan of Patient 8 showing hypodense gliotic foci on the right thalamus and a smaller one on the left putamen. J: Axial T2 FLAIR MRI image of Patient 8 shows two gliotic foci- one on the right thalamus and the other on the left putamen. K: Axial T2 FLAIR MRI image of Patient 9 showing a hypointense lesion encroaching on the left thalamus. L: Axial gradient echo (GRE) sequence MRI scan of Patient 9 showing blooming artifact on the left thalamus and a smaller foci on the right internal capsule suggestive of hemosiderin residue from an old hemorrhage. MRI, magnetic resonance imaging; CT, computed tomography; FDG-PET, fluorodeoxyglucose positron emission tomography; FLAIR, fluid attenuated inversion recovery.
Clinical features
Dystonia was the most frequently associated hyperkinetic movement of the affected limb (n = 8; 88.8%). Frontalis and cervical dystonia were noted in one patient each (n = 1; 11.1%). A single patient had voice tremor (n = 1; 11.1%), while another had dysarthria (n = 1; 11.1%). Chorea was a feature in one patient (n = 1;11.1%). The tremor was bilateral in two patients (n = 2; 22.2%), while lower limb tremor was prominent in a single patient (n = 1; 11.1%). Tremor was noted to be distal predominant in n = 4; 44.4% of patients. Hemiparesis/monoparesis was the most commonly associated neurodeficit (n = 8; 88.8%), followed by proprioceptive defects of the ipsilateral limb (n = 4; 44.4%) and hemihypesthesia (n = 2; 22.2%). Two patients had cranial nerve deficits (n = 2; 22.2%). Depression/psychosomatic symptoms were noted in two participants (n = 2; 22.2%) (Tables 1, 2).
Treatment outcomes
All patients were managed with pharmacological therapy, which included sequentially increasing doses of levodopa, followed by various combinations of trihexyphenidyl, clonazepam, baclofen and levetiracetam (Table 2). The mean dosage of levodopa administered was 311.11 ± 96.1 mg (range: 200–450 mg). All patients were followed up for at least 3 months following the onset of therapy. The patients were classified into responders and nonresponders as per preset criteria (30% or more reduction in performance score of TETRAS score). Five subjects (55.5%) were deemed to be responders after independent assessment by all three investigators at 3 months or beyond of follow-up, while the remaining (n = 4; 44.4%) were categorized as nonresponders. The mean performance score (PS) score of TETRAS in responders prior to therapy was 19.0 ± 3.5, which was significantly lower after therapy at 9.6 ± 1.1 (p = 0.014). The mean levodopa dose in responders was 240 ± 54.7 mg, which was significantly lower than the levodopa dosage in nonresponders (400 ± 40.8 mg; p = 0.012) (Table 3).
DISCUSSION
It is now largely accepted that HT is a heterogeneous clinical entity, with significant clinical variations both in its semiology and etiology, a fact that was acknowledged by Gordon Holmes himself more than a century ago [6,9]. Holmes, in his seminal work, attributed the classic tremor to lesions of the red nucleus or the cerebello-rubral system, emphasizing that the former is phylogenetically a part of the thalamencephalon. The phenomenon that a thalamic vascular insult can give rise to a high-amplitude, low-frequency tremor with all three components is well documented [10-12]; this observation is in congruence with the putative role of the thalamus in the so-called “long loop”, i.e., the dentate-rubro-thalamo-cortico-pontine-cerebellar pathway [13]. Although thalamic lesions have been shown to account for several new-onset HT phenotypes [3], various other anatomical locations have also been similarly implicated in its genesis; these include the red nucleus, globus pallidus (GP), ponto-mesencephalic junction, cerebellar cortex, and vermis [2]. The involvement of the nigro-striatal pathway, however, is speculative and is derived mostly from the observation that HT is responsive to levodopa and the asymmetry of uptake demonstrated in functional dopaminergic studies [2,3,14].
Thalamus and Holmes’ tremor
The authors described nine cases that presented with an HT phenotype with or without other associated hyperkinetic movements, with variable latencies following a thalamic vascular insult. The relative paucity of resting tremors in some of these patients is in accordance with previous observations that thalamic lesions are unlikely to generate resting state tremors, unless particularly severe [10,11,15-17]. According to a plausible hypothesis, synchronized oscillations of the inferior olive (IO) nucleus, because of perturbations of the so-called “short loop”, i.e., dentatoolivary-cerebellar pathway, are believed to cause regular, involuntary tremors such as static and rest tremors [13]. In HT, nigrostriatal pathways are often implicated along with cerebello-thalamic tracts, and the former might contribute to the generation of the restrest tremor. According to the recent lesion connectome concept in HT, the involved thalamic nuclei are the ventralis oralis posterior (VOP) and pulvinar [1] rather than the ventralis intermediate (VIM), which is commonly targeted in DBS for tremor. Furthermore, a study using multimodal 3D medical imaging in HT revealed a differing clinical picture and therapeutic response in HT depending on the involved thalamic nuclei and their connections [18]. Hence, clinical phenotyping could be pivotal in the selection of appropriate target(s) for DBS in refractory HT.
Among the patients with thalamic infarctions (patients 1, 3, 4, 5 and 8), the vascular territory was thalamo-geniculate with involvement of the lateral part of the thalamus in two patients (patients 1 and 4), while the posterior choroidal artery/thalamoperforate territories were implicated in three subjects (patients 3, 5 and 8). This is in coherence with works of previous authors [10,12,15] who had implicated the thalamogeniculate and, in few cases, the posterior choroidal territories in the pathogenesis of involuntary abnormal movements following thalamic vascular insult. It is worth noting that involvement of the ventrolateral thalamic nuclei, particularly the VIM nucleus, is fundamental to the genesis of tremor and hyperkinetic movements, as it is an integral part of the cerebello-thalamo-cortical loop [19]. Whether the interruption occurs at the level of the VIM or prior to entry of the cerebello-thalamic fibers into the nucleus, however, is unknown, since it has been shown that the VIM lies outside the brain circuit responsible for HT [1]. In fact, the lesion connectome concept in HT affects the VOP and pulvinar nuclei as culprits for tremor genesis. Previously, Krystkowiak et al. [10] and Lehéricy et al. [12] suggested lesions of the centromedian (Cm) thalamic nucleus as a cause of dystonia; however, more intricate networks might be at play.
The authors noted that both the tremor and associated dystonia improved with levodopa therapy, which might be due to the functional proximity of the paramedian thalamic territories to the mesencephalon and the nigro-striatal tract (Figure 2). The concept of a direct connection between the mesencephalic dopaminergic system and the paramedian thalamic nuclei groups, i.e., the “mesothalamic pathway”, was proposed by Freeman et al. [20]. Lesions of this pathway are believed to cause retrograde degeneration of the upstream basal ganglia and nigrostriatum. A recent meta-analysis underscores levodopa as the most effective pharmacological agent for HT [21], thus strengthening previous hypotheses emphasizing the role of nigrostriatal dopaminergic denervation in the pathogenesis of HT. However, this pathway alone might not be enough to explain all aspects of the HT phenotype.
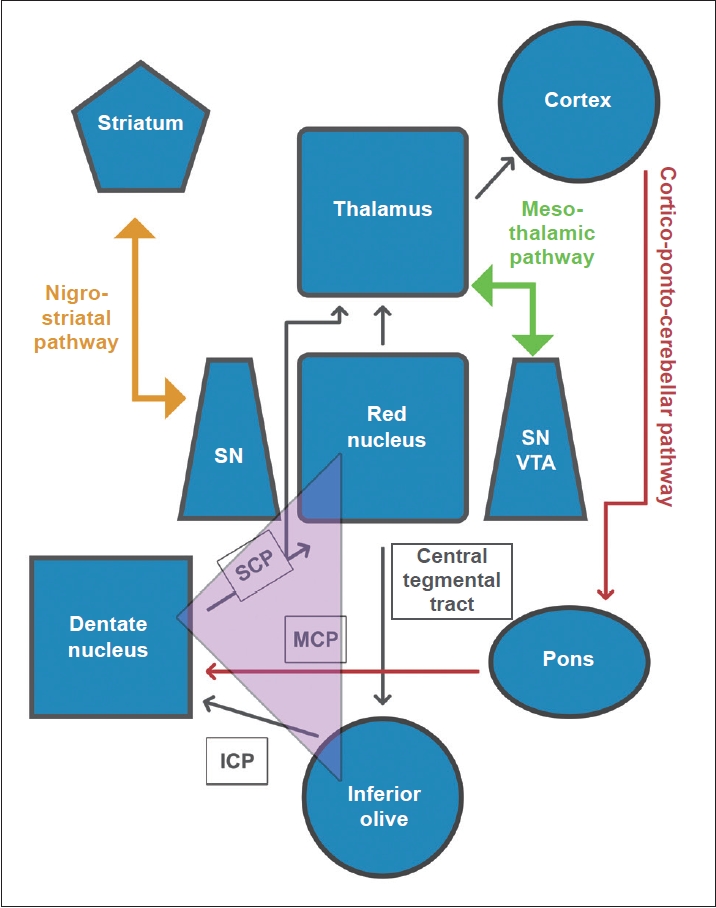
Schematic representation depicting 1) connections between the inferior olive, red nucleus and contralateral dentate nucleus as part of the Guillain-Mollaret triangle, 2) connections between the cortex and pons to contralateral cerebellum as part of the cortico- ponto-cerebellar pathway, 3) connections between the dentate nucleus, contralateral red nucleus, contralateral thalamus and cortex as part of the dentato-rubro-thalamo-cortical pathway (“long loop”), 4) adjacent connections between mesencephalic dopaminergic neurons of the ventral tegmental area (VTA) and substantia nigra (SN) and the thalamus as the meso-thalamic pathways, and 5) connections between SN and the striatum as the nigro-striatal pathway. The functional proximity among various discrete neuroanatomical substrates renders them susceptible to perturbations following a lesion among any one or more of these pathways. Purple triangle indicates Guillain-Mollaret triangle. SCP, superior cerebellar peduncle; MCP, middle cerebellar peduncle; ICP, inferior cerebellar peduncle.
Dystonia in HT
Dystonia was the most commonly associated hyperkinetic movement, and it was present in the majority of patients (8/9; 88.8%). The distribution of dystonia mostly followed the tremor-affected limb, although it also involved the lower limbs in 2 patients. An irregular, jerky, dystonic head, and neck tremor was a feature of one of the patients in the series (patient 2). Bilateral involvement of the upper limbs was noted in two of the subjects (patients 5 and 8). It has been previously described that movement disorders resulting from thalamic lesions are mostly mixed phenomena [15,17]. Dystonia was present in nearly 83% of cases in a recent study on HT (Table 1) [22]. The authors found that dystonia was ameliorated following levodopa therapy, which might implicate pallido-thalamic and nigro-striatal pathways in the pathogenesis of dystonia in HT.
Chorea in HT
Another interesting observation in the present series is that one patient (patient 1) had initially developed choreiform movement of the affected extremity, which was coupled with dystonic posturing. Chorea is an exceedingly rare manifestation of thalamic stroke. Acute pure hemichorea has been previously reported in thalamic lacunar infarction [23]; however, the patient in the present study had a delayed onset by a few weeks. This might be attributable to perturbations in thalamostriatal connections and cerebral plasticity.
Functional imaging, role of FDG-PET and abnormal sensory feedback
Recovery in motor function is linked to synaptic plasticity and reorganization of motor tracts in the frontal motor and premotor cortex as well as recruitment of uncrossed pyramidal fibers from the opposite motor cortex and contralateral cerebellum [24]. Hence, it is appealing to consider that these newly formed pyramidal motor/cerebellar pathways might be unstable and misdirected, with perturbed sensory-motor feedback and feed-forward mechanisms, leading to the development of tremors in the affected limb. The authors found hypometabolism on brain FDG-PET in the bilateral inferior frontal cortex in one patient (patient 3) who had ischemic stroke involving the right occipital cortex and medial thalamus. Functional aberrations of thalamo-striato-cortical connections might have contributed to decreased metabolism in areas that are otherwise anatomically discrete from the primary site of vascular insult.
Presently, there is increasing evidence to support the role of abnormal sensory motor integration in the pathogenesis of many movement disorders. In this regard, proprioceptive afferent impulses are particularly important for smooth coordination and execution of movements [25]. The basal ganglia have numerous reciprocal connections with the frontal and limbic cortex and the thalamic nuclei, which in turn are regulated by dopamine and dopamine receptors. These connections comprise two distinct loops, namely, the direct and indirect pathways, which exert facilitatory and inhibitory influences on the motor cortex, respectively. Successful execution of motor control, however, is critically linked to appropriate sensory feedback; therefore, the basal ganglia serve as a gateway to sensory feedback for motor control [26]. The cerebellum directly receives rich sensory information, and this plays an important role in motor coordination [27]. Thus, the authors hypothesize that disrupted central processing of sensory information might play a significant role in the genesis of hyperkinetic movements such as tremor, which might additionally reduce the efficacy of treatment modalities that mostly focus on motor circuitry.
Treatment of HT
HT is a source of major debilitation to the patient, owing to its large amplitude and refractoriness to conventional pharmacotherapy. Efforts have been made to treat it with several agents, namely, levodopa and anticholinergics; however, outcomes have been variable and largely unsatisfactory [2,3]. Surgical options such as DBS have achieved considerable success in ameliorating all tremor subtypes, including HT [28]. However, the most common therapeutic target for HT DBS therapy, the VIM, has been associated with remission in less than half of such cases [1]. Therefore, in the future, it may be beneficial to shift the focus to other targets, such as the GP, subthalamic nucleus (STN), and other thalamic nuclei (such as the VOP), which are more integral parts of the HT circuit [1]. In the present study, the mean dosage of levodopa used to achieve definitive clinical response was significantly lower in the responder cohort. The authors observed that increasing the daily dosage of levodopa beyond 300 mg/day does not add much clinical benefit; in contrast, it causes various side effects (nausea, vomiting, dizziness) and tolerability issues. What exactly should be the cutoff daily dosage beyond which therapeutic benefit is unlikely is a question that needs to be addressed through future controlled prospective studies in larger cohorts of patients.
Dopamine transporter scan and appraisal of the role of the nigrostriatal pathway
Likewise, the role of the nigrostriatal pathway in the pathogenesis of HT has been a subject of contention for decades. Several researchers have attempted to evaluate cases of HT of various etiologies with functional dopamine uptake scans [2,4,7,29-36] (Table 4). Remy et al. [7] were the first to examine functional aberrations at both the pre- and postsynaptic dopaminergic levels and demonstrated significant presynaptic reductions in dopamine uptake as well as a response to dopaminergic therapy. Similar observations of reduced or absent presynaptic dopamine uptake and dopaminergic therapy response were reported by Strecker et al. [31], Guedj et al. [32], Seidel et al. [4], and Juri et al. [35]. However, this is debatable since other authors have reported contrary observations. Sung et al. [33] and Paviour et al. [29] failed to document a dopaminergic therapeutic response despite a reduction in striatal presynaptic dopamine uptake. Hertel et al. [30] and Gajos et al. [2] reported cases of midbrain and thalamic HT, respectively, with normal uptake in pre- and postsynaptic domains (Table 4). Considering the inconsistencies in these studies, it would be premature to assume that the involvement of nigrostriatal is sine qua non for the genesis of HT. Despite these controversies, the role of levodopa in the treatment of HT remains undeniable and should be part of a therapeutic trial of all such patients.
The present study has a few limitations. First, the number of participants was small, and no statistically significant conclusion can be drawn based on the present findings. Second, functional brain imaging was not used in the evaluation of every patient due to various contraindications and lack of logistic support; hence, the assumptions are chiefly based on clinical and structural radiological findings and clinical outcomes, leaving scope for investigator bias. Third, the TETRAS scale used for quantifying tremor severity was originally meant for essential tremor phenotypes and does not take into consideration the remaining component of tremor. However, the patients were independently assessed by movement disorder specialists with concurrence of opinion regarding improvement.
In hindsight, thalamic lesions led to distinct, heterogeneous tremor phenotypes with variable responses to therapy. Dystonia was frequently associated with tremor. The phenotype of HT was not uniform, and it varied according to the localization of the lesion within the thalamus. The tremor improved with levodopa in the majority of the patients. An increase in the daily dosage of levodopa beyond a certain point did not add any clinical benefit, and those who responded did so at significantly lower doses than those refractory to treatment.
Supplementary Material
The online-only Data Supplement is available with this article at https://doi.org/10.14802/jmd.23095.
Video 1.
Case 1: Irregular, large amplitude tremor of the right upper limb, present at rest, which accentuates the assumption of an anti-gravity posture, with prominent intention tremor of the ipsilateral extremity. Case 2: Large amplitude tremor of the right upper limb, present at rest, which accentuates on assuming anti-gravity posture as well as intention tremor. There is involvement of the proximo-distal musculature of the ipsilateral extremity as well as a dystonic neck and head tremor.
Video 2.
Case 3: Tremor at rest of first left hand (mostly at first two metacarpo- phalangeal joints), which accentuates on assuming anti-gravity posture with prominent dystonic posturing of fingers of ipsilateral hand. An intention tremor component is noted on finger-to-nose testing of the left upper limb.
Video 3.
Case 4: Large amplitude, proximal predominant postural tremor on forward extension as well as wing-beating posture of the right upper limb. Case 5: Bilateral (asymmetric, right > left) rest tremor of upper limb, which accentuates on assuming anti-gravity posture (re-emergent postural tremor) as well as a kinetic (intention) component. A voice tremor is evident.
Video 4.
Case 6: Irregular, jerky, proximo-distal postural tremor of the right upper limb with intention tremor of the ipsilateral extremity upon finger-to-nose testing. Case 7: Dystonic posturing of outstretched fingers of the left hand, with postural and intention tremors of the ipsilateral extremity.
Video 5.
Case 8: Choreodystonic movement of both upper limbs (asymmetric, left > right) at rest and extended posture. A postural and intention tremor of the left upper limb was noted with proximo-distal muscle involvement. Case 9: Slow, myorhythmic rest tremor, with postural and intention tremor of the right upper extremity. Dystonic posturing of the fingers of the right hand noted.
Notes
Conflicts of Interest
The authors have no financial conflicts of interest.
Funding Statement
None
Author contributions
Conceptualization: Amlan Kusum Datta, Adreesh Mukherjee. Data curation: Amlan Kusum Datta, Adreesh Mukherjee. Formal analysis: all authors. Investigation: Amlan Kusum Datta, Adreesh Mukherjee, Sudeshna Malakar. Methodology: Amlan Kusum Datta, Adreesh Mukherjee, Atanu Biswas. Project administration: Atanu Biswas, Amlan Kusum Datta. Resources: Amlan Kusum Datta, Adreesh Mukherjee, Atanu Biswas. Software: Amlan Kusum Datta, Adreesh Mukherjee, Atanu Biswas. Supervision: Atanu Biswas, Adreesh Mukherjee. Validation: Sudeshna Malakar, Adreesh Mukherjee, Atanu Biswas. Visualization: Amlan Kusum Datta, Adreesh Mukherjee, Atanu Biswas. Writing—original draft: Amlan Kusum Datta. Writing—review & editing: Adreesh Mukherjee, Atanu Biswas, Sudeshna Malakar.