Validation of the Thai Version of the Movement Disorder Society-Sponsored Revision of the Unified Parkinson's Disease Rating Scale
Article information
Abstract
Objective
This study aims to validate the Thai translation of the Movement Disorder Society-sponsored revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS).
Methods
The English version was translated into Thai and then back-translated into English. The translated version underwent 2 rounds of cognitive pretesting to assess the ease of comprehension, ease of use and comfort with the scale. Then, it underwent large clinimetric testing.
Results
The Thai version was validated in 354 PD patients. The comparative fit index (CFI) for all four parts of the Thai version of the MDS-UPDRS was 0.93 or greater. Exploratory factor analysis identified isolated item differences in factor structure between the Thai and English versions.
Conclusion
The overall factor structure of the Thai version was consistent with that of the English version based on the high CFIs (all CFI ≥ 0.90). Hence, it can be designated the official Thai version of the MDS-UPDRS.
Parkinson’s disease (PD) is a common neurodegenerative disorder. In the 1980s, the Unified Parkinson’s Disease Rating Scale (UPDRS) was developed to provide a unified, comprehensive instrument for the assessment of different domains of symptoms and disabilities in PD patients [1]. The Movement Disorder Society (MDS) commissioned a review of the UPDRS in 2001. The revised version, the MDS-sponsored revision of the UPDRS (MDS-UPDRS), was published for use in 2008 after passing rigorous clinimetric testing [2].
The MDS-UPDRS is divided into 4 parts as the original UPDRS with some changes [2]. Part I includes ‘‘non-motor experiences of daily living,’’–Part IA is rater rated, and Part IB is rated by the patient and/or caregiver. Part II includes ‘‘motor experiences of daily living,’’ to be filled by the patient and/or caregiver. Part III is the ‘‘motor examination,’’ like the UPDRS but with new additions of items and choices in tremor rating for improved consistency. Part IV concerns ‘‘motor complications’’ like the UPDRS but with a revised assessment method.
Here, we present the results of the clinimetric assessments of the Thai translated version of the MDS-UPDRS.
MATERIALS & METHODS
Six institutions in Thailand were involved in this study. The Institutional Review Board of each center approved the study (IRB No. 542/57, IRB No. MTU-EC-IM-5-159/57). Informed consent was obtained from all participants. The study consisted of 3 phases—translation, cognitive pretesting of the translated version, and validation (clinimetric testing).
Translation
The English version of the MDS-UPDRS questionnaire was translated into Thai (SS, AP) and then independently back-translated into English (SP, PJ, NL) by movement disorder neurologists fluent in both English and Thai. The back-translation was reviewed by the USA team (GTS, CGG, NLP). Suggestions for changes were incorporated into the Thai translation. Once the finalized version was developed, it went through the cognitive pretesting phase.
Cognitive pretesting
Cognitive pretesting is used to qualitatively assess whether upon completion of the questionnaire, the task is at an appropriate difficulty level for the assessor and the assessed, the assessed can maintain interest and attention, and the assessed feels comfortable and is able to comprehend the questionnaire [3]. The purpose is to assess the ease of comprehension, ease of use, and comfort with the scale. The cognitive pretesting packets have specific questions for both raters and patients and request feedback on specific scale items. The scale items that were different when back-translated from Thai to English and the items that were used in this phase of the English version were chosen for cognitive pretesting.
Three raters (OP, PS, PP) not involved in the original translation administered the cognitive pretesting package to a total of 10 patients. Once no problems were noted, the final translation (Official Working Document) was obtained. This version then underwent large clinimetric testing for validation.
Validation
The Thai version of the MDS-UPDRS was administered to native Thai-speaking PD patients. Once all the data were collected, they were analyzed by a team in the USA.
Statistical analysis
Factor analysis
M-plus Version 7 (Muthen & Muthen, Los Angeles, CA, USA) was used to perform the primary confirmatory and secondary exploratory factor analyses. Factor estimation was conducted using an unweighted least squares approach that minimizes the sum of squared differences between observed and estimated correlation matrices and ignoring diagonal elements. To assist in interpretation, orthogonal CF-varimax rotation was used to minimize the correlation between factors.
To perform the statistical analysis, 5 subjects were needed for each item of the questionnaire [4]. Given there are 65 items on the MDS-UPDRS, a sample of at least 350 was required. If there were missing values from a subject, only the part with those missing values was excluded from the analysis. Hence, the sample size of various parts may vary.
Primary analysis
Confirmatory factor analysis (CFA) was conducted to address the primary interest in determining whether the factor structure for the English language MDS-UPDRS [2] can be confirmed with data collected using the Thai translation.
The CFA constructing Thai data to fall into the factors defined in the English language data was conducted separately for MDS-UPDRS Parts I to IV. The comparative fit index (CFI) was used to evaluate the CFA results. According to the protocol, a successful translation and an official translation of the MDS-UPDRS would require CFIs for parts I–IV of the translated MDS-UPDRS of 0.90 or greater relative to the English version. To validate the model fit, we further examined the mean- and variance-adjusted weighted least squares.
Secondary analysis
In the secondary analysis, to explore the underlying factors without the constraint of a predefined factor structure, an exploratory factor analysis (EFA) was performed in the Thai version of the MDS-UPDRS Parts I–IV. For each part of the MDS-UPDRS, the number of factors was chosen using a scree plot.
A scree plot [5] is a line plot of the eigenvalues on the y-axis and the number of factors on the x-axis. It displays a downward curve, starting high on the left, descending toward the right, and then flattening out after the cutting-off point. After the factors are chosen, items with factor loadings greater than 0.40 were retained. To assist in the interpretation of the factors, orthogonal CF-varimax rotation was used to minimize the correlation among the factors.
RESULTS
Cognitive pretesting
Questions included for this phase were cognitive impairment, anxious mood, handwriting, freezing, hand movements, arising from chair, time spent with dyskinesia, and functional impairment of dyskinesia.
Sixteen patients along with their examiners were interviewed in two rounds. In the first round of cognitive pretesting, word changes were suggested for items in Parts I, II, and IV. The second round of cognitive pretesting demonstrated successful revisions to the above listed items. This version, called the Official Working Document, was then validated in a large group of patients.
Validation
Factor analysis
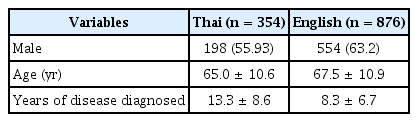
The demographic characteristics of the sample are shown in Table 1. The Thai dataset included 354 native Thai-speaking PD patients who were examined using the Official Working Document of the Thai MDS-UPDRS. Supplementary Table 1 (in the online-only Data Supplement) shows the frequency of responses for each answer choice of each item for both the Thai and English datasets.
Primary analysis
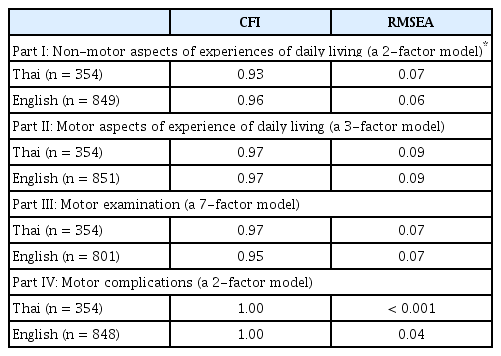
The CFA models for each part of the MDS-UPDRS are displayed in Table 2. The CFI was 0.93 or greater for all four parts of the Thai MDS-UPDRS in comparison with the factor structure of the English language. Dopamine dysregulation syndrome did not load on any factor in the original English version and was thus excluded from this analysis [5].
Secondary analysis
The EFA analysis for the Thai and English datasets differed in some areas. The results are shown in Supplementary Table 2 (in the online-only Data Supplement). Supplementary Figure 1 (in the online-only Data Supplement) shows the scree plots for all four parts.
Several factors were extracted from the scree plot—2 from Part I, 3 from Part II, 7 from Part III, and 2 from Part IV. In contrast to the English version, in Part I, “cognitive impairment and light headedness on standing” did not load on any factors, while fatigue loaded on both factors. For Part II, “handwriting” loaded on Factor 2, but loaded on Factor 1 in the English version. “Eating tasks” and “tremor” loaded on Factor 2 instead of on Factor 3 in the original English version. “Getting out of bed” loaded on Factor 3, but loaded on Factor 2 in the English version. In Part III, both the English and Thai versions loaded on different factors in 12 of the 33 items, and 9 items loaded on more than one factor. Both versions demonstrated that most items loaded on different factors had multiple cross-loadings. “Time spent with dyskinesias” in Part IV did not load on any of the factors. “Time spent in the OFF state” of the Thai version loaded on Factor 2, while it loaded on Factor 1 in the English version. “Complexity of motor fluctuations,” “functional impact of fluctuations,” and “painful OFF state dystonia” in the Thai version loaded on both Factor 1 and Factor 2.
Internal consistency
To evaluate the internal consistency of the Thai version of the MDS-UPDRS, Cronbach’s alpha was calculated for each part. In addition, 95% confidence intervals were reported based on 1,000 bootstrap samples (Supplementary Table 3 in the online-only Data Supplement). All four parts have good internal consistency with alpha larger than 0.8.
DISCUSSION
The MDS-UPDRS is a standard scale to evaluate different domains of PD. It has been translated into many languages and is used worldwide. The Thai version of MDS-UPDRS encountered a few hurdles in translation and cognitive pretesting. Some of the sentences were too long, which made the patients confused. This was resolved by asking the questions in parts. For example, for question 1.1 on cognitive impairment: “Over the past week have you had problems remembering things, following conversations, paying attention, thinking clearly, or finding your way around the house or in town?” The question was asked in parts, such as, “Over the past week have you had problems remembering things?”; wait for an answer, then ask “following conversations?”; wait for an answer, and so on. Once this was done and a few words were changed, the questionnaire was properly understood and passed the cognitive pretesting phase.
Because the developers suggested that each part of the scale be used individually [2], we conducted clinimetric assessments for each part separately. For validation of the Thai translation, we required a minimum CFI of 0.90. The minimum CFI we obtained was 0.93 (Part I), and the maximum was 1.00 (Part IV). Therefore, we considered the Thai translation’s factor structure to be consistent with the English language version.
We expected variations between the target language factor structure and the unconstrained factor structure of the Thai translation. This is a common occurrence in CFA and represents the natural variability from sample to sample. As expected, we found isolated item differences in the factor structure between the original English version and the Thai translation. We found cross-loadings for different items in the various factors of the Thai translation. These differences in the factor loadings across the two versions are most likely due to cultural differences. The differences are also seen in other language translations, albeit differently [6-8]. Nonetheless, the overall factor structures were confirmed to be consistent with the English version, hence rendering them suitable for use as a validated translation.
Overall, our results met the criteria for validation of the Thai translation of the MDS-UPDRS, with noted high CFIs for each part of the scale. The Thai version demonstrated a global factor structure similar to that of the English version, and thus, the Thai translation is considered an official Thai version of the MDS-UPDRS.
Supplementary Materials
The online-only Data Supplement is available with this article at https://doi.org/10.14802/jmd.21104.
Supplementary Table 1.
Distribution of Responses of MDS-UPDRS by Language
Supplementary Table 2.
Comparison of English language and Thai Exploratory Factor structures for Parts I-IV of the MDS-UPDRS
Supplementary Table 3.
Internal consistency of each MDS-UPDRS part
Supplementary Figure 1.
Scree plot for exploratory factor analysis. A: Part I: Non-motor aspects of experiences of daily living. B: Part II: Motor aspects of experiences of daily living. C: Part III: Motor examination. D: Part IV: Motor complications.
Notes
Conflicts of Interest
The authors have no financial conflicts of interest.
Funding Statement
This research was funded by the Royal College of Physicians of Thailand.
Author Contributions
Conceptualization: Nancy LaPelle, Pablo Martinez-Martin, Glenn T. Stebbins, Christopher G. Goetz, Sheng Luo. Formal analysis: Xuehan Ren, Sheng Luo. Funding acquisition: Roongroj Bhidayasiri, Priya Jagota. Investigation: Priya Jagota, Prachaya Srivanitchapoom, Sitthi Petchrutchatachart, Surat Singmaneesakulchai, Apichart Pisarnpong, Praween Lolekha, Suwanna Setthawatcharawanich, Parnsiri Chairangsaris, Natlada Limotai, Pawut Mekawichai, Pattamon Panyakaew, Onanong Phokaewvarangkul, Jirada Sringean, Yuvadee Pitakpatapee. Methodology: Nancy LaPelle, Pablo Martinez-Martin, Glenn T. Stebbins, Christopher G. Goetz, Sheng Luo. Project administration: Priya Jagota, Roongroj Bhidayasiri. Resources: Nancy LaPelle, Pablo Martinez-Martin, Glenn T. Stebbins, Christopher G. Goetz, Roongroj Bhidayasiri. Supervision: Roongroj Bhidayasiri, Glenn T. Stebbins, Christopher G. Goetz. Validation: Glenn T. Stebbins, Christopher G. Goetz, Sheng Luo. Writing—original draft: Priya Jagota, Xuehan Ren, Sheng Luo. Writing—review & editing: Prachaya Srivanitchapoom, Sitthi Petchrutchatachart, Surat Singmaneesakulchai, Apichart Pisarnpong, Praween Lolekha, Suwanna Setthawatcharawanich, Parnsiri Chairangsaris, Natlada Limotai, Pawut Mekawichai, Pattamon Panyakaew, Onanong Phokaewvarangkul, Jirada Sringean, Yuvadee Pitakpatapee, Nancy LaPelle, Pablo Martinez-Martin, Glenn T. Stebbins, Christopher G. Goetz, Roongroj Bhidayasiri.
Acknowledgements
The official Thai version of the MDS-UPDRS is available at https://www.movementdisorders.org/MDS/MDS-Rating-Scales/MDS-Unified-Parkinsons-Disease-Rating-Scale-MDS-UPDRS.htm.