Articles
- Page Path
- HOME > J Mov Disord > Volume 10(1); 2017 > Article
-
Review Article
Functional Neuroanatomy for Posture and Gait Control - Kaoru Takakusaki
-
Journal of Movement Disorders 2017;10(1):1-17.
DOI: https://doi.org/10.14802/jmd.16062
Published online: January 18, 2017
The Research Center for Brain Function and Medical Engineering, Asahikawa Medical University, Asahikawa, Japan
- Corresponding author: Kaoru Takakusaki, MD, PhD, The Research Center for Brain Function and Medical Engineering, Asahikawa Medical University, 2-1, 1-1 MidorigaokaHigashi, Asahikawa 078-8511, Japan Tel: +81-166-68-2884 Fax: +81-166-68-2887 E-mail: kusaki@asahikawa-med.ac.jp
• Received: December 13, 2016 • Accepted: December 15, 2016
Copyright © 2017 The Korean Movement Disorder Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted noncommercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
- Here we argue functional neuroanatomy for posture-gait control. Multi-sensory information such as somatosensory, visual and vestibular sensation act on various areas of the brain so that adaptable posture-gait control can be achieved. Automatic process of gait, which is steady-state stepping movements associating with postural reflexes including headeye coordination accompanied by appropriate alignment of body segments and optimal level of postural muscle tone, is mediated by the descending pathways from the brainstem to the spinal cord. Particularly, reticulospinal pathways arising from the lateral part of the mesopontine tegmentum and spinal locomotor network contribute to this process. On the other hand, walking in unfamiliar circumstance requires cognitive process of postural control, which depends on knowledges of self-body, such as body schema and body motion in space. The cognitive information is produced at the temporoparietal association cortex, and is fundamental to sustention of vertical posture and construction of motor programs. The programs in the motor cortical areas run to execute anticipatory postural adjustment that is optimal for achievement of goal-directed movements. The basal ganglia and cerebellum may affect both the automatic and cognitive processes of posturegait control through reciprocal connections with the brainstem and cerebral cortex, respectively. Consequently, impairments in cognitive function by damages in the cerebral cortex, basal ganglia and cerebellum may disturb posture-gait control, resulting in falling.
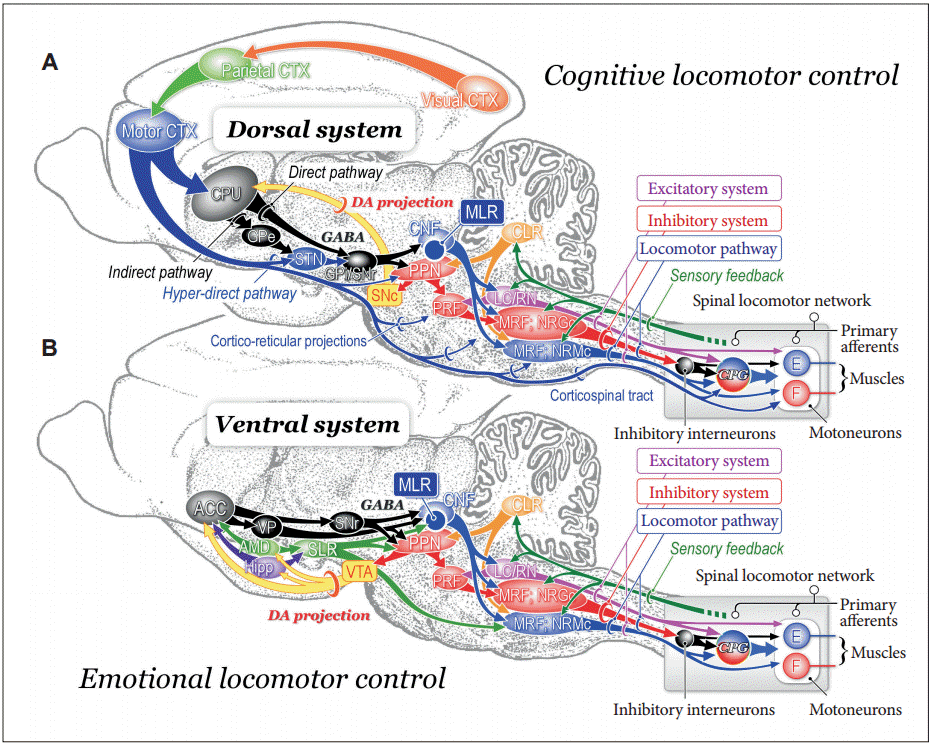
- Figure 1 illustrates our recent understanding of basic signal flows involved in motor control. Sensory signals arising from external stimuli and/or internal visceral information have various functions. For example, they are to be utilized for cognitive processing such as production of working memory which guides future behavior. Alternatively, they may affect emotional and arousal states. Sensory signals are further available to detect and correct postural instability by acting on the cerebral cortex, cerebellum, and brainstem. Accordingly, animal initiates movements depending on either a “cognitive reference” or an “emotional reference” [1,2].
- Voluntary movements are derived from intentionally-elicited motor commands arising from the cerebral cortex to the brainstem and spinal cord. On the other hand, emotional reference may contribute to emotional motor behavior which is generated by the projections from the limbic-hypothalamus to the brainstem, such as fight or flight reactions [1,3,4]. Regardless of whether the initiation is volitional or emotional, goal-directed behaviors are always accompanied by automatic process of postural control including balance adjustment and muscle tone regulation. The subject is largely unaware of this process which is evoked by sequential activations of neurons in the brainstem and spinal cord. Basic locomotor motor pattern is generated by spinal locomotor networks that is termed as the central pattern generators (CPG). However, in order to learn motor skills or behave in unfamiliar circumstance, the subject requires cognitive posture-gait control that depends on cognition of self-body information together with spatial localization of objects in extra-personal space.
- The cerebellum regulates the cognitive and automatic processes of posture-gait control by acting on the cerebral cortex via the thalamocortical projection and on the brainstem, respectively. Both the feedforward information from the cerebral cortex via the cortico-ponto-cerebellar pathway and real-time sensory feedback via the spinocerebellar tract to the cerebellum may play major roles in these operations. The basal ganglia may also contribute to the modulation of each process though its gamma-aminobutyric acid (GABA)-ergic projections to the cerebral cortex and brainstem [2,5,6]. The degree of GABAergic influence from the basal ganglia is regulated by the midbrain dopaminergic neurons [7].
GENERAL SCHEMA OF POSTURE-GAIT CONTROL
- In the absence of their forebrain, like a decerebrate cat, it can walk, trot and gallop. When the decerebration is made at precollicular-postmammillary level, the cat initiates locomotion by electrical or chemical stimulation applied to the mesencephalic or midbrain locomotor region (MLR) [1,8,9]. However, if the neuraxis is transected slightly higher at the precollicular-premammillary level, cats can spontaneously elicit locomotion with well-coordinated postural control [10]. Therefore, a critical region exists between these decerebrate levels. This area is recognized as the subthalamic locomotor region (SLR), which mostly corresponds to the lateral hypothalamic area. Stimulation of the SLR evoked locomotion after a large lesion was made in the MLR area [11], indicating that the SLR has direct connections with the brainstem locomotor pathway beyond the MLR. However, the walking in the decerebrate preparations is machine-like and is neither goal-directed nor adaptive to the environment. Hence, the SLR connections to the MLR are likely important for normal control of posture and gait.
- So far three locomotor regions have been identified in animals: the MLR in the mesopontine tegmentum, the SLR and the cerebellar locomotor region (CLR) in the mid-part of the cerebellum [12]. Human imaging study demonstrated that the organization of these supraspinal locomotor centers was preserved during the transition to bipedal locomotion human [13]. The clinical relevance of these centers has so far been largely neglected.
- Role of the mesencephalic area in the control of posture and locomotion
- The MLR appears to be present in all classes of vertebrates [14]. It likely includes the cuneiform nucleus (CNF) and the pedunculopontine tegmental nucleus (PPN), although the precise location of the locomotor regulation still remains a matter of debate. The PPN is located in the ventrolateral part of the caudal mesencephalic reticular formation, composed of a heterogeneous population of neurons, containing GABA and glutamate in addition to acetylcholine [15]. Different neuronal types within the PPN area may have different functions with their own inter-connections to multiple parts of the brain. There are connections to cerebral cortex, multiple basal ganglia and limbic areas, the thalamus, the brainstem, the spinal cord and the cerebellum [16]. This key location including multiple segregate functions, renders figuring out the precise function of these regions quite complicated [17].
- In the experiments using decerebrate cats, activation of neurons in the PPN suppress muscle tone via its cholinergic projections to the pontine reticular formation (PRF), while activation of neurons in the CNF mostly elicits locomotion [5,15,18-20]. Microstimulation of the transition zone between the two areas induced a mixture of locomotor rhythm with associated muscle tone suppression (Figure 2). Moreover, blocking the PRF by injecting atropine sulfate, blocked the PPN-induced atonia but facilitated MLR-induced locomotion, indicating that cholinergic PPN neurons not only control the level of muscle tone but also modulate the locomotor pattern, and do this by effects at the pontine level [15,18]. Studies in rats by Sherman et al. [21] show non-cholinergic neurons just medial to the PPN have projections to the spinal cord, while the cholinergic neurons of the PPN do not. This area at the mesopontine junction may be the true MLR. The dorsal neurons of this MLR area (laterodorsal tegmental nucleus) with spinal projections are active in locomotion, while the ventral neurons are active in standing and do not have spinal projections.
- Functional organization of the reticular formation in the control of posture
- It is generally agreed that the reticulospinal tract (RST) contributes to regulation of the level of muscle tone. There may exist functional organization in the pontomedullary reticular formation (PMRF) in relation to the control of postural muscle tone (Figure 3) [15]. Direct recording of reticulospinal neurons (RSNs) revealed that those located in the dorso-medial part of the PMRF are active during the period of muscle tone suppression or muscular atonia (Figure 3Aa), and those in the ventromedial part are active during reflex standing due to decerebrate rigidity or hypertonus (Figure 3Ab). Accordingly, functional topographical organization may exist in the PMRF in the control of postural muscle tone. On the other hand, during MLR-elicited locomotion or spontaneously evoked locomotion in high-decerebrated preparation, RSNs located in both the dorsomedial and ventromedial PMRF were modulated in response to step cycles (Figure 3Ac), indicating that muscle tone-related RSNs participate in the execution of locomotion so that locomotor rhythm and muscle tone can be simultaneously regulated by the reticulospinal system during locomotion.
- Presence of such an organization was supported by experiments using microstimulation applied to the PMRF (Figure 3B). Stimulation applied to the dorsomedial part of the PMRF resulted in general suppression of muscle tone (Figure 3B, red areas), and those applied to the ventromedial PMRF induced general augmentation of muscle tone (Figure 3B, blue areas). Neuroanatomical studies revealed that RSNs in the dorsomedial and ventromedial PMRF descend through ventral and lateral funiculi, indicating that the ventral and lateral RST are responsible for muscle tone suppression and augmentation, respectively. On the other hand, tegmental reflex, or asymmetrical postural figures, which was characterized by extension of the unilateral limb and flexion of the contralateral limb, was evoked from the area between the inhibitory and excitatory areas and the lateral part of the PMRF where few RSNs arise in the cat (Figure 3B, green areas).
- In addition to the RST, vestibulospinal tract (VST) plays an important role in the control of postural equilibrium by its similar architectonic organization of descending fibers within the spinal cord with the RST [22,23]. Matsuyama and Drew [24,25] examined firing property of neurons in the RST and VST in the cat during locomotion on an inclined surface. Specifically, the VST controls primarily the overall level of postural muscle tone, while the RST has an additional role in determining the relative level of different muscles, particularly when the pattern is asymmetric.
- Locomotor pathway and its control by the forebrain structures
- In the “locomotor pathway”, signals from the MLR also activate medullary RSNs, in turn commanding the spinal locomotor network to generate the oscillatory pattern of locomotion [8,26]. However, the SLR and CLR may also activate this reticulospinal locomotor pathway through distinct and direct projections [27]. Signals from the MLR may also activate monoaminergic descending pathways including the coerulospinal and raphespinal tracts, acting as a muscle tone excitatory system [28-30]. Then we focus on the inputs of the forebrain structures to the midbrain MLR/PPN area. In decerebrate cats, the basal ganglia control locomotion and posture using different GABAergic output pathways of the substantia nigra reticulata (SNr); the lateral part of the SNr blocks the PPN-induced muscle tone suppression, whereas the medial part of the SNr suppresses the MLR-induced locomotion [18,20,31]. Recent rat studies confirm that inhibitory input from the SNc (GABAergic and dopaminergic) to ventral MLR regulate posture, while inhibitory projection from the GABAergic SNr to the dorsal MLR regulates locomotion [21]. In Parkinson’s disease (PD), GABAergic outputs of the basal ganglia are abnormally increased [7], so excessive SNr-inhibition of the MLR may cause gait disturbance and muscle rigidity [5,18].
- However, it is unknown whether these mechanisms are the same for bipedal humans and quadrupedal animals. It is also unclear what drives or dictates the SNr-induced control of movements. There are sub-compartments in the basal ganglia; neostriatum-dorsal pallidal pathway (“dorsal pathway”, Figure 4A) and ventral striatum-ventral pallidal pathway (“ventral pathway”, Figure 4B). The nucleus accumbens, as a component of the limbic system, may be important in releasing locomotion, via GABAergic projections disinhibiting the MLR via the ventral pallidum [32,33] and via fibers to the SNr (Figure 4B) [34]. Because the nucleus accumbens also receives inputs from the hippocampus and amygdala, the ventral pathway may be implicated in reward-oriented locomotor behaviors, as it receives inputs from ventral tegmental area, hippocampus and amygdala. On the other hand, the more recently evolved parts of the basal ganglia make up the dorsal system (Figure 4A) [35]. These parts may achieve locomotor control depending on cognitive behavioral context, such as sensoryguided locomotor control.
- Spinal control of posture and gait
- Spinal circuitry involved in the stretch reflex, reciprocal inhibition, non-reciprocal inhibition (or autogenic inhibition) and flexion reflexes are involved in the control of posture. Particularly, stretch reflex and non-reciprocal inhibition (Ib inhibition) play major role in static control of posture. On the other hand, interneuronal networks involved in reciprocal Ia inhibition and flexion reflexes including crossed-extension reflex and are critical to produce postural figures with extension-flexion movements of left-right leg alternation during walking. Integration of all spinal reflex networks therefore can be essential to full execution of muscle tone regulation during movement. While spinal reflex networks generate rhythm and pattern of locomotor movements through the activation of the CPG, they play crucial role in supporting body during stance phase of locomotion as well. Because spinal preparations in quadruped animals do not express postural reflex described above, neural networks within the spinal cord alone does not enable to control postural equilibrium [36,37], and integration of descending supraspinal signals and peripheral sensory afferents at the level of spinal cord is necessary for full execution of postural control.
- Organization of the spinal locomotor network
- Once animals start locomotion, muscle tone must be regulated depending on locomotor cycles. A particular group of spinal interneuronal networks that generates rhythmic activity in the absence of rhythmic inputs is termed CPG [1,36,37]. The rhythmic interneuronal activity is sent to the second-order interneurons in the intermediate region (lamina IV-VII of Rexed), which shape “locomotor patterns” of each limb’s movements [26,38]. The signals are then transmitted to the target motoneurons innervating ipsilateral limb muscles through their excitatory and inhibitory actions [26,36]. Reciprocal Ia interneurons, classical Ib interneurons and Renshaw cells are likely included in this group [36]. On the other hand, lamina VIII interneurons projecting to the contralateral side contribute to the left-right alternations of limb movements [39,40]. The rhythm and pattern are transmitted back to the supraspinal structures via the spinothalamic, spinoreticular and spinocerebellar tracts so that the supraspinal structures monitor all events in the spinal cord [36].
- Spinal control of muscle tone during locomotion
- Activity of the spinal locomotor networks is modulated by sensory afferents in a phase dependent manner [36,38,41,42]. For example, proprioceptors in muscles at the hip joint are primarily responsible for regulating the stance phase. Afferents from proprioceptors in extensor muscles regulate transition from stance to swing phase. It should be critically noted that signals in Ib afferents from tendon organ in ankle extensor muscles inhibit homonymous motoneurons at rest, while they excite extensor motoneurons during stance phase [36,41]. The functional consequence of this “reflex reversal” is that the swing phase is not initiated until the extensor muscles are unloaded and the forces exerted by these muscles are low.
- Skin afferents also exert a powerful influence on the CPG [26,36,41]. Skin receptors are important to detect obstacles and adjust stepping to avoid them such as the “stumble-corrective reaction [43]. Importantly, the corrective flexion movements are produced only if the paw is stimulated during the swing phase. An identical stimulus applied during stance phase elicits the opposite response, an excitation of extensor muscles that reinforces the ongoing extensor activity. This is another example of the reflex reversal. The reflex reversal phenomenon is critically involved in postural control during locomotion. However, its mechanisms have not been elucidated.
BRAINSTEM AND SPINAL CORD; CORE-STRUCTURES OF POSTURE-GAIT CONTROL
- Classical lesion studies in the cat
- Even cerebral cortex was removed, the kitten can alive more than several years [44]. They could eat and exhibit periods of rest, become active, search for fool, and were able to remember the location of food. They utilized the visual and haptic senses with respect to external space. However, in the adult cats, skilled locomotor performance was disturbed when lesions were made in the motor-related cortical regions [45]. If the caudate nucleus of the cat is selectively removed both sides [46], a remarkable behavior develops referred to as the “compulsory approaching syndrome”. The cat faithfully followed any moving object that catches its attention, seemingly unable to terminate the locomotor behavior. This was referred to as “visually-determined cortical automatism [47]”. The main manifestations consisted of loss of drive (apathy), obsessive-compulsive behavior, cognitive deficits, stimulus-bound perseverative behavior, and hyperactivity [46]. On the other hand, removal of both the cerebral cortex and the striatum (diencephalic cat; the thalamus and hypothalamus were preserved) resulted in the cats walking incessantly, even though they did not attend to any environmental stimuli [48].
- Forebrain structure including the cerebral cortex, the limbic-hypothalamic structures, and the basal ganglia as well as the cerebellum control posture and gait largely by acting on the reticulospinal system through their direct and indirect connections via the MLR/PPN area (Figure 4). These cortical and subcortical projections may enable animals to express volitional and emotional motor behaviors depending on the context [2,14].
- Control by the cerebral cortex
- While basic locomotor synergy was not largely disturbed if pyramidal tracts were bilaterally disconnected [49], skilled locomotor task was severely impaired. Liddell and Phillips [50] found after unilateral or bilateral pyramidal lesions that the cats became ‘helplessly immobile’, unable to take a step without slipping or falling, when they were required to walk along a narrow beam or horizontal ladder. Such a skilled performance became more severely damaged by postcruciate than by precruciate lesions. After postcruciate lesions including both the somatosensory and parietal cortices, the cat refused to walk on narrow trucks [45]. The precruciate area, which corresponds to supplementary motor area (SMA) and premotor area (PM) of the primates, may be involved in movement initiation. On the other hand, the postcruciate cortices may utilize specific somatosensory inputs to fulfil a role in the regulation of ongoing movements [51] in the manner of anticipatory or feed-forward adjustments [52]. Skilled posture-gait control, therefore, can be achieved on the basis of knowledges of the orientation and motion of the body in space as well as motion perception and spatial localization of objects in extra-personal space [53-55]. Such a knowledge is provided by integration of vestibular, somatosensory and visual sensory signals which occurs at both the cerebral cortex and cerebellum (Figure 5A) [56].
- Precise visuomotor coordination occurred at the cerebral cortex plays critical roles in the execution of precise arm-hand movements such as reaching and grasping [57]. Similarly, when a walking subject encounters obstacles, each leg must be placed with a high degree of accuracy through the visuo-parieto-frontal cortical projection, as in the subject has to modify the leg trajectory in each step in order to achieve appropriate foot placement [58]. Such a visuomotor coordination is particularly necessary in quadrupeds because an obstacle is no longer within the visual field by the time the hindlimbs are stepping over it. When the posterior parietal cortex was bilaterally removed, the cat’s hindlimbs did not step over the obstacles if their forelimbs cleared them [59]. Therefore, the posterior parietal cortex must be engaged to register and store the temporospatial relationship between the obstacle and one’s bodily information, such as body schema, in short-term memory that is utilized to produce motor programs in the motor cortices (Figure 5B) so that the cat can precisely modify the limb trajectory [59,60]. To successfully achieve such an accurate control of limb movements during walking, posture must be optimized in advance to the purposeful action so that bodily equilibrium can be maintained. Therefore, the visuo-parieto-frontal cortical projection (Figure 5B) is critically involved in the fulfilment of ongoing purposeful control via anticipatory adjustments of posture [61]. It follows that both the intentional limb movements and anticipatory postural adjustment are programmed at motor cortical areas (Figure 5B and D).
- Then, what part of the motor cortex contributes to the programming of the anticipatory postural adjustments? One of the most candidate areas is the SMA and PM (Figure 5B and C). In bipedal walking monkey, an inactivation of the leg area of the primary motor cortex (M1) by injecting muscimol (GABAA agonist) partly paralyzed the contralateral leg [62]. On the other hand, muscimol injections into the trunk/leg regions of the bilateral SMA largely disturbed postural control without inducing paralysis [63]. When it was injected into the dorsal PM, spontaneous walking was maintained; however, the monkey could not start walking using sensory cue. These findings indicate the SMA and PM may contribute to postural control during bipedal walking and initiation of gait, respectively.
- Studies using neural tracers have demonstrated abundant cortico-fugal projections to the PMRF from the premotor cortices (SMA/PM) in quadruped [64] and biped [65] animals. Recent studies have focused onto the importance of cortico-pedunculopontine projection in terms of motor control. Probabilistic diffusion tractography in rhesus monkey as well as humans, shows that the SMA is strongly connected to the lateral PPN, while the dorsal PM is connected to the medial PPN [66]. The RST innervates whole spinal segments [22] so that it controls postural muscle tone and symmetric postural figures (Figure 3) [15]. Therefore, the cortico-reticular and RST may achieve anticipatory postural adjustment (Figure 5D) Possibly, the SMA contributes to the anticipatory postural adjustment for step initiation, which is impaired in PD patients [67]. On the other hand, the PM/SMA may forward programs of precise leg-foot movement to the M1 [68], which, in turn, sends motor command via the corticospinal tract.
- Consequently, cognitive information in the temporoparietal cortex is essential for accurate gait control particularly when the subject encounters an unfamiliar environment. Therefore, the deficiency in the information processing from the temporoparietal cortex to the frontal cortex (frontoparietal network) may cause errors in anticipatory postural adjustment and gait difficulties such as the “freezing of gait”. It follows that deficits in cognitive function in elder persons and in patients with Alzheimer’s disease are at higher risk of falling particularly when more cognitive tasks are required [69,70].
- Next critical question as to the cortical control of posture is “how does the brain acquire access to an internal estimate of body motion and postural verticality?” Postural vertical is supported by a sense of verticality which is synthesized by visual, somatosensory and vestibular information [71-75]. Among them, the vestibular sensation is superior to others in terms of absoluteness of sensation because it always refers the gravity [76], and the vestibular system provides the brain with sensory signals about three-dimensional head rotations and translations. Vestibulothalamic projections are bilateral and mainly involve the posterior thalamus [77-79]. While there was no cortical area that receives inputs exclusively from vestibular afferents, there are multiple presentations of vestibular information in the cerebral cortex [80], such as the frontal eye field, PM, somatosensory cortex, ventral intraparietal cortex, medial superior temporal area, and parieto-insular vestibular cortex (PIVC). The PIVC has particularly dense connections with other vestibular-relating cortical areas, and receives information from other sensory modalities [81-83]. Now, both the posterior thalamus and PIVC are areas of interest for the internal model of postural verticality [84-88]. Because the PIVC has descending projections to the contralateral vestibular nuclei [79,89-91], the vestibular cortical systems possibly contribute to upright standing by acting to the vestibulospinal system based on the internal model of postural verticality (Figure 5B).
- Postural verticality is often disturbed in pathological conditions such as “pusher syndrome” after stroke and “Pisa syndrome” in advanced PD. Stroke patients with pusher syndrome actively push away from the ipsilesional side and have a tendency to fall towards their paretic, contralesional side (the left side for right-hemisphere patients). They had lesions including the parietal insular cortex or posterior thalamus [84,87,88,92]. This phenomenon is more prominent when patients are upright compared to when lying down. Now the pusher phenomenon can be arising from a conflict or mismatch between visual and postural vertical [84,87,88]. However, it is still uncertain whether the same pathophysiological mechanism is operating in Pisa syndrome.
- Posture-gait control by the cerebellum
- Postural control by the cerebellum highly depends on sensory afferents. Signals from the labyrinth ascend the vestibular nerve to the floccules and vermis of the cerebellum in addition to the vestibular nuclei. The fastigial nuclei (FN) receive a copy of the output of the spinal cord in addition to peripheral sensory information via spinocerebellar tracts [93]. The FN also receive visual [94] and vestibular [95] information. These multisensory features may provide “an error-correction mechanism”, which permits FN neurons to affect motor functions such as coordinating postural responses during walking which entail changes in limb position. The FN may therefore send highly integrated bodily information to the posture-gait related areas in the brainstem and motor cortical areas [96].
- Output from the FN to the brainstem contributes to the control of postural muscle tone. Electrical stimulation applied to the mid-part of the cerebellar white matter in decerebrate cats either increased [97] or reduced the level of muscle tone [98]. The cerebellar stimulation possibly activated the excitatory RST and VST of both sides so that extensor muscle tone was bilaterally increased. On the other hand, the decrease in the level of muscle tone is considered to be due to withdrawal of excitatory influence upon motoneurons [98]. Because, postural muscle tone is generally reduced by the damage of the medial part of the cerebellum, the cerebellum contributes to the activation of antigravity muscles. Possibly, the FN regulates posture-gait subprograms in the brainstem and spinal cord by inparallel activation of fastigio-spinal, fastigio-reticular, and fastigio-vestibular pathways [27,99,100]. Therefore, the deficiency in these pathways may reduce the degree of α–γ linkage in patients with cerebellar diseases, resulting in hypotonia. The hypotonia state reduces the accuracy of the sensory feedback so that posture-gait control can be seriously disturbed.
- Reciprocal connections between the cerebellum and cerebral cortex (Figure 5C) may be critically involved in the cognitive and programing processes of postural control. More recently, it has been recognized that cerebellar output reaches vast areas of the cerebral cortex including prefrontal and posterior parietal cortices in addition to motor-related areas [101]. The FN in the cerebellum, as well as the vestibular cortex (PIVC), is critically involved in encoding internal postural model in space and self-motion [56]. Some studies have suggested the presence of FN projections to the parietal cortex [102,103], motor cortex and multimodal visual areas [104]. Reciprocal connection between parietal cortex and the cerebellum may be involved in perception of body motion in space (Figure 5B). Such a bodily information can be utilized to maintain upright posture during standing and to achieve anticipatory postural adjustment. The latter may involve reciprocal connections between the motor cortical area (SMA/PM) and cerebellum (motor loop) in order to construct motor programs (Figure 5C) [105,106]. Accordingly, cerebellar disease patients may have problems in cognitive process of postural control. However, the perception of verticality in patients with cerebellar ataxia may only deteriorate in a more advanced stage of the disease [107]. In addition, only few abnormalities of anticipatory postural adjustment were found in the cerebellar disease patients compared to controls, while the patients appeared to be less able to use predictive information [108]. Because the cerebellum is reciprocally connected with the basal ganglia [109], it is possible that basal ganglia in addition to the cerebral cortex may complement the cerebellar role of cognitive process of postural control.
- Posture-gait control by the basal ganglia in relation to PD
- Because posture-gait control is seriously impaired in PD, the basal ganglia has long been functionally regarded to be predominantly involved in motor control but is increasingly recognized to play additional roles in sensory processing, cognition, and behavior [110-112]. Here, emphasis has been placed on the mechanisms of posture-gait impairment in PD so that understanding the role of basal ganglia to the postural control is facilitated. Based on our recent understanding, postural disturbances in PD attribute to following mechanisms: 1) disturbances in the dopaminergic and cholinergic systems, 2) impairment of cognitive functions due to failure of integrative sensory processing that allows to produce internal postural model (body schema), 3) failure in motor programming due to reduced activity in the motor cortical areas, and 4) disturbances in posture-gait areas in the brainstem [15,18,113].
- Recent clinical studies suggest that the postural impairments in PD is based on dysfunction of both the dopaminergic and cholinergic systems. Disturbances in these chemospecific systems may critically contribute to posture-gait failure in this disease. For example, a damage of dopamine (DA) neurons in the substantia nigra pars compacta (SNc), which project to the basal ganglia nuclei, is considered to increase in the GABAergic inhibitory output from the basal ganglia [7,114,115]. This may strongly inhibit thalamocortical systems and brainstem structures (Figure 1).
- In addition, cholinergic neurons in the PPN (brainstem) and basal forebrain (BF) are seriously damaged in PD [116-119]. Indeed, a reduction of the thalamic cholinergic innervation in patients with PD has no cognitive and motor impairments but exhibits an increase in postural sway speed [120]. Cholinergic PPN neurons project to the non-specific thalamocortical system [121-123], basal ganglia nuclei including DA neurons in the SNc and PMRF [124-126]. On the other hand, cholinergic BF projections to the cerebral cortex are necessary for attentional performance and cognitive processing [127].
- Therefore, disturbances in attention, sensori-motor integration and cognitive processing in PD can be largely attributed to the damage of the cholinergic systems. Accordingly, both of the excessive inhibition from the basal ganglia and the damage of cholinergic systems may impair both the cortical and subcortical, particularly the brainstem, functions.
- Cortical activity was substantially reduced in patients in PD compared control subject during walking [128,129]. This may cause failure of integrative sensory processing, which in turn, disturb construction internal postural model and motor programming. It has been shown that a loss of cholinergic neurons in the BF and PPN associates with fallers in PD [116,130]. Müller and Bohnen [131] suggested that reduced activities in the cholinergic PPN neurons may disturb multi-sensory integration at the level of the thalamus. This may also explain why patients in PD with more severe posture-gait instability have a high risk of developing dementia [131]. Impairment of the integration of sensory information, particularly proprioception [132] and vestibular graviception [133], may result in deficits of internal model of postural verticality which is possibly constructed at the temporoparietal association area including the vestibular cortex. Therefore, asymmetry in the activity of the left and right vestibular cortices may induce leaning upright posture, which is often called as Pisa syndrome. Pisa syndrome is a dystonic lateroflexion of the trunk with a postural disturbance resembling the leaning tower of Pisa, and is more often observed in patients with advanced PD [134]. The marked lateroflexion become worsen during walking but is almost completely alleviated by passive mobilization or supine positioning [135]. Because PD without Pisa syndrome also had deficiencies in postural verticality compared to healthy controls [132,136-138], mismatch between proprioception and vestibular gravitation in PD may alter subjective postural verticality, resulting in Pisa syndrome. Alternatively, asymmetry of the basal ganglia output, which is due to cholinergic-dopaminergic imbalance in the striatum [134,139,140] or disturbance of the pallidal output [141,142], may also elicit left-right disproportion of the thalamocortical processing of vestibular information.
- The motor cortical areas including the M1, SMA, and PM have connections with the basal ganglia and cerebellum, constituting motor loop that contributes to execution and motor programming of voluntary movements (Figure 5C) [105,106]. Because of increasing inhibitory output from the basal ganglia to the thalamocortical projections in addition to reduced cognitive information processing, the capability of producing motor programs in response to changes in circumstance can be deteriorated. In fact, the SMA contributes to the anticipatory postural adjustment for step initiation via corticofugal projections to the PPN and PMRF, and this process is seriously impaired in PD patients [67]. Also, the dorsal part of the PM is involved in sensory-guided gait control as suggested in bipedally walking monkey [62]. Because the activity of dorsal part of the PM was increased during visually-guided paradoxical gait in PD, posture-gait programs in SMA/PM became reusable by the activation of visuo-motor pathway [129]. Accordingly, failure in motor programming in PD can be due to the decrease in excitability of the motor cortical areas in addition to impairment of sensory processing in the temporoparietal cortices. Recently, role of the cerebellum in the pathophysiology of PD is highly recognized [143,144]. Because the cerebellum has reciprocal connections with the basal ganglia (Figure 5C) in addition to the cerebral cortex and brainstem, there is a need to elucidate whether the cerebellum participates in compensatory mechanisms associated with the disease or contributes to the pathophysiology of PD.
- We propose that reduced excitability in the mesopontine tegmentum including the PPN/MLR can be also involved in posture-gait failure in PD [6,15,18,20,113]. In decerebrate cats, the basal ganglia control locomotion and posture using different GABAergic output pathways of the SNr; the lateral part of the SNr blocks the PPN-induced muscle tone suppression, whereas the medial part of the SNr suppresses the MLR-induced locomotion [18]. Recent studies in rodents confirm that inhibitory input from the SNr to the glutamatergic neurons in the MLR regulates locomotion [21,145]. In PD, GABAergic outputs of the basal ganglia are abnormally increased [7,114], so excessive SNr-inhibition of the MLR may cause gait disturbance and muscle rigidity acting on MLR and muscle tone inhibitory region in the PPN [18]. Muscle tone rigidity in PD is called as “lead-pipe like rigidity” which is characterized by co-contraction of extensor and flexor muscles. Such a co-contraction is observed in neck, back and leg muscles, resulting in flexion posture including camptocormia [146].
- PD patients with cholinergic cell loss in the PPN showed more severe motor disabilities with gait and posture, which were associated with L-3,4-dihydroxyphenylalanine (L-DOPA)-resistant akinesia [147,148]. Subsequent post-mortem study in PD patients established a correlation between the occurrence of falls and freezing and the loss of cholinergic PPN neurons. However, the degree of neuronal loss in the CNF was not significantly different between fallers and non-fallers in PD patients [147]. In PD patients, individual neurons in the dorsal PPN increased their firing rates with increased stepping frequency [149]. Moreover, gait speed in PD patients was correlated with a power of alpha-oscillations (7–10 Hz) of field potentials recorded from the PPN area [150].
- As one of clinical procedures for alleviating gaitposture deficiency in PD, deep brain stimulation (DBS) targeting the PPN with the aim of stimulating remaining cholinergic neurons [151-155]. The first studies using DBS in advanced PD patients concluded that low-frequency stimulation of the PPN could be effective to control freezing of gait and falls. However, further clinical studies concluded that freezing of gait were mildly improved by PPN-DBS but some results were rather disappointing [156,157]. These results emphasize the need to determine the optimal surgical target [158,159]. Ferraye et al. [156] suggest that the most suitable targets are located slightly posterior to the PPN pars compacta, probably in the ventral part of the CNF where stimulation-induced locomotion has been reported in animals [18]. This area possibly corresponds to the subcuneiform nucleus as described by Alam et al. [160] and Karachi et al. [161] also suggest that it may be the case that treating PD patients suffering from failure of gait initiation versus falling may require specifically targeting the CNF and the dorsal part of the PPN, respectively.
- In human, Fling et al. [162] used functional neuroimaging approach and revealed strong functional connectivity between the SMA and PPN/MLR area, which was positively correlated with freezing severity in patients of PD. In contrast, connectivity between the STN and SMA was lost. They suggested that the former connectivity may potentially due to a maladaptive compensation, and the latter may reflect the reduced automatic control of gait by the basal ganglia. A study using diffusion tensor imaging revealed the connection between the cerebellum and the PPN in PD patients without freezing of gait. However, freezers of patients in PD showed the absence of cerebellotegmental connectivity and increased visibility of the decussation of corticopontine fibers in the anterior pons [163]. These findings highlight the importance of corticopontine-cerebellar pathways in the pathophysiology of gait when the cerebellotegmental connection that may contribute to automatic execution of gait control is damaged in freezers of PD.
HIGHER-ORDER REGULATION OF POSTURE-GAIT CONTROL
Anticipatory postural adjustment
Maintenance of vertical posture
Action on the brainstem structures
Cerebellar actions on the cerebral cortex
Disturbances in the dopaminergic and cholinergic systems
Impairments in sensory processing and cognition
Failure in motor programming
Reduced activity in posture-gait area in the midbrain
Reorganization of cortico-cerebello-brainstem pathways in PD
- This work is partially supported by grants from JSPS KAKENHI (Grant Numbers 26120004 and 25290001) and from Japan Agency for Medical Research and Development (AMED) for K.T. KT is also supported by granting foundations from the QOLER Medical Group and Sasson Hospital.
Acknowledgments
Figure 1.Basic signal flow involved in postural control. Multisensory signals from the visual, vestibular, auditory, somatosensory (proprioceptive), and visceral receptors act on various sites in the central nervous system. These signals may provide cognitive and emotional references to the cerebral cortex and limbic system, respectively, so that the subject may elicit either voluntary movements or emotional motor behavior depending on the context. In each case, automatic process of postural control, such regulation of postural muscle tone and basic postural reflexes, by the brainstem and spinal cord is required. On the other hand, cognitive postural control is particularly important when the subject learns motor skills and behaves in unfamiliar circumstance. See text for detail explanation. Modified from Takakusaki. Mov Disord 2013;28:1483-1491, with permission of Wiley [6].
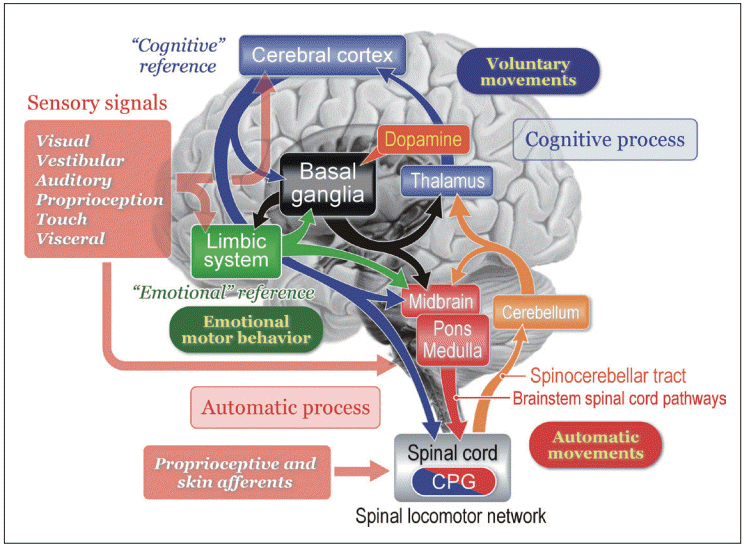

Figure 2.Effects of midbrain stimulation on posture and locomotion in decerebrate cat preparation. A: Stimulation sites in the right mesopontine tegmentum. Stimulation consists of 30 μA in intensity and 50 Hz in frequency with a duration of 10 seconds. B: Effects of stimulation applied to each site in (A) on left and right soleus muscle electromyograms. Stimulation of the dorsal part of the CNF induced muscle tone augmentation. While stimulation of the ventral CNF and the dorsal PPN induced locomotor rhythm, the latter was accompanied by a decrease in muscle tone. Stimulation of the PPN and PRF corresponding to the nucleus reticularis pontis oralis (NRPo) immediately suppressed soleus muscle activities. C: Topography of stimulus effects in the mesopontine tegmentum. Locomotion was evoked by stimulating the CNF (blue). Stimulation of the locus coeruleus (LC) and dorsolateral CNF induced hypertonia (violet; muscle tone augmentation). Ventrolateral part of the PPN and NRPo, induced muscular atonia (red) and hypotonia (orange). Stimuli applied to the locomotion-evoking sites and atonia-evoking sites elicited a mixture of rhythmic limb movements and muscle tone suppression (green). Modified from Takakusaki et al. J Neural Transm (Vienna) 2016;123:695-729, with permission of Springer [15]. CNF: cuneiform nucleus, PPN: pedunculopontine tegmental nucleus, IC: inferior colliculus, SCP: superior cerebellar peduncle.
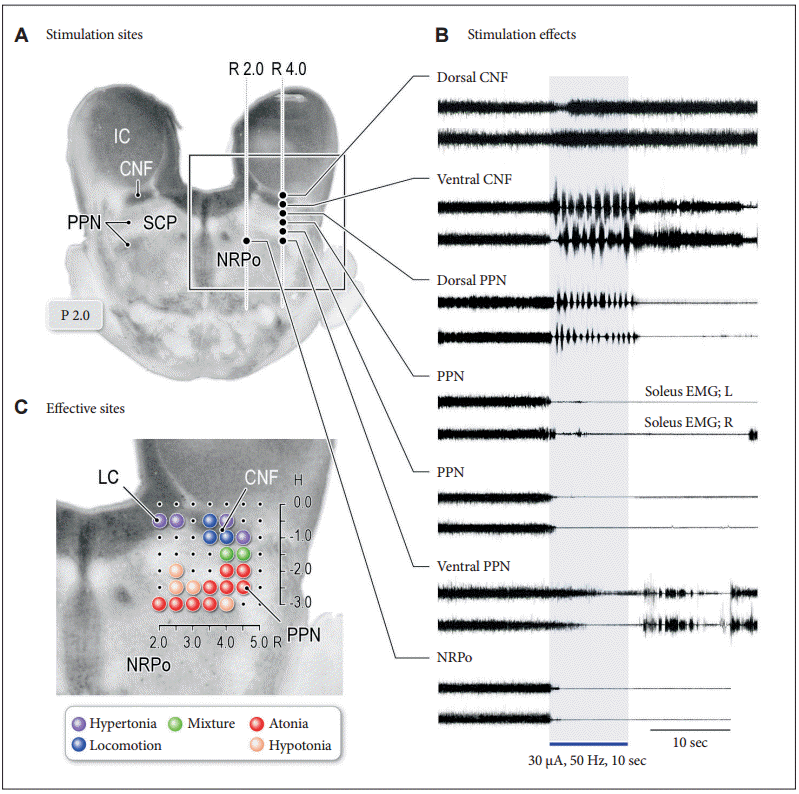

Figure 3.Functional organization of medullary reticulospinal systems in decerebrate cats. A: Locations of the medullary reticulospinal neurons relating to muscle tone suppression (a), muscle tone augmentation (hypertonus) (b), and locomotion (c). During reflex standing of the decerebrate cats, reticulospinal neurons with a firing frequency more than 10 Hz during reflex standing of decerebrate cats are judged as hypertonus-related reticulospinal neurons (b; n = 76). When carbachol (long-acting cholinomimetic agents) was injected into the pontine reticular formation muscle tone of decerebrate cats was abolished. Reticulospinal neurons of which firing frequency was increased to more than 10 Hz during carbachol-induced atonia are judged as atonia-related reticulospinal neurons (a; n = 75). During reflex standing (decerebrate rigidity) these cells usually had no spontaneous firing. Locomotion-related neurons (n = 59) were judged as those displaying rhythmic firing relating to step cycles of locomotion. Recording was made in both high decerebrated cats which displayed spontaneous locomotion and normal decerebrated cats with stimulation of the MLR. B: Results obtained from five animals are superimposed on representative coronal planes of the caudal pons and medulla. Sites from which either suppression (red), augmentation (blue), or tegmental reflexes (green) was elicited in more than three out of five animals are marked. Sites from which the stimulation induced postural changes in more than four animals are indicated by darker colored squares; conversely, light colored squares indicate that the postural changes were induced in three animals. Modified from Takakusaki et al. J Neural Transm (Vienna) 2016;123:695-729, with permission of Springer [15]. P: pyramidal tract, MLF: medial longitudinal fasciculus, 5ST: spinal trigeminal tract, NRPc: nucleus reticularis pontis caudalis, TB: trapezoid body, RM: nucleus raphe magnus, SO: superior olive, NRGc: nucleus reticularis gigantocellularis, NRMc: nucleus reticularis magnocellularis, RPa: nucleus raphe pallidus, NRPv: nucleus reticulars parvocellularis.
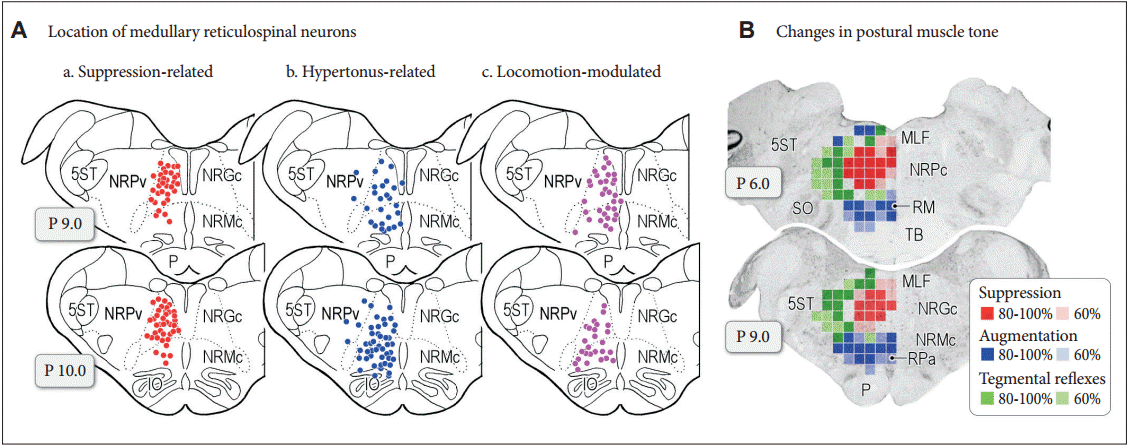

Figure 4. Neuronal mechanisms of cognitive (A) and emotional (B) control of locomotion in the cat. A: Dorsal system for cognitive locomotor control. A visuo-motor pathway from the visual cortex to motor cortex via the parietal cortex contributes to this control. Corticofugal projections act on to the basal ganglia nuclei, brainstem and spinal cord. Dopaminergic projection from the substantia nigra pars compacta (SNc) to the caudate-putamen (CPu) may be involved in learning the locomotor behaviors. GABAergic output from the basal ganglia nuclei (internal segment of the globus pallidus and substantia nigra pars reticulata; GPi/SNr) acts on MLR/PPN area may control locomotion and posture. Efferents from the midbrain locomotor region (MLR) recruit excitatory system, inhibitory system and locomotor pathway. The excitatory system arises from the LC and the raphe nuclei. The inhibitory system which arises from cholinergic neurons in the PPN sequentially activates PRF neurons, medullary reticulospinal neurons in the nucleus reticularis gigantocellularis (NRGc) and spinal inhibitory interneurons. The inhibitory interneurons may inhibit both motoneurons and interneurons. The locomotor pathway consists of reticulospinal neurons arising from the ventromedial medulla corresponding to the nucleus reticularis magnocellularis (NRMc). Cholinergic and glutamatergic projections from the PPN excite SNc-DA neurons. These descending tracts act on CPGs in spinal cord so that muscle tone and locomotion are regulated. Efferents from the (CLR may excite locomotor pathway. B: Ventral system for emotional locomotor control. Efferents from the amygdala (AMD) and hippocampus (Hipp) project to the nucleus accumbens (NAc). GABAergic NAc neurons project to ventral pallidum (VP) and the SNr, which control activity of the MLR/PPN neurons. Efferents from the AMD and the Hipp also act on lateral hypothalamic area, which corresponds to the SLR. DA projections from the ventral tegmental area (VTA) may contribute to the rewardoriented locomotor behaviors. Modified from Snijders et al. Ann Neurol 2016;80:644-659, with permission of Wiley [164]. E: extensor motoneurons, F: flexor motoneurons, PRF: pontine reticular formation, PPN: pedunculopontine tegmental nucleus, LC: locus coeruleus, RN: raphe nuclei, DA: dopamine, CLR: cerebellar locomotor region, SLR: subthalamic locomotor region, CNF: cuneiform nucleus, CTX: cortex, GPe: external segment of the globus pallidus.


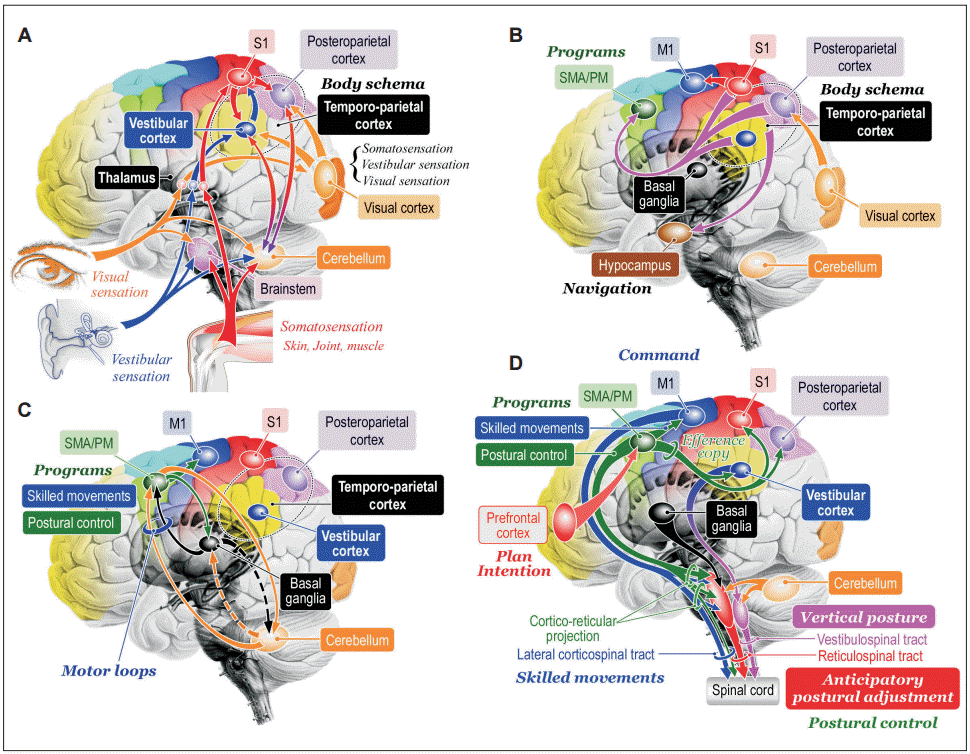
Figure 5.Hypotheses of cognitive process of posture-gait control. A: Cognition of bodily information. Sensory signals flowing into the central nervous system converge to the brainstem, cerebellum, thalamus, and cerebral cortex. At the level of cerebral cortex, signals from the visual cortex, vestibular cortex and primary sensory cortex (S1) is integrated and internal model of self-body, such as body schema and verticality can be constructed at the temporoparietal cortex including the vestibular cortex and posteroparietal cortex. Reciprocal connection between the temporoparietal cortex and cerebellum may contribute to this process. B: Transmission of the bodily information. The bodil y information is then transmitted to the supplementary motor area (SMA) and premotor area (PM) where the information can be utilized as materials to produce motor programs. Similarly, the information is transferred to hippocampus and is used to navigate further behaviors. C: Motor programming. The motor cortical areas closely cooperate with the basal ganglia and cerebellum so that appropriate motor programs are constructed. D: Postural control by corticofugal projections to the brainstem and spinal cord. The bodily information generated at the vestibular cortex may be utilized for sustention of vertical posture via cortico-vestibular and vestibulospinal tract. Signals from the prefrontal cortex including plans and intentions may trigger to run motor programs in the SMA/PM, which may include those for purposeful movements and associating postural control. The postural control program may be utilized to generate anticipatory postural adjustment via cortico-reticular and reticulospinal tract. Then motor programs are sent to the M1 so that goal-directed purposeful skilled movements can be achieved. Modified from http://dx.doi.org/10.1080/01691864.2016.1252690, with permisson of Taylor & Francis [165].

- 1. Grillner S. Control of locomotion in bipeds, tetrapods, and fish. In: Brookhart JM, Mountcastle VB, editors. Handbook of physiology. The nervous system II. Bethesda, MD: American Physiological Society; 1981:1179–1236.
- 2. Takakusaki K. Forebrain control of locomotor behaviors. Brain Res Rev 2008;57:192–198.ArticlePubMed
- 3. Sinnamon HM. Preoptic and hypothalamic neurons and the initiation of locomotion in the anesthetized rat. Prog Neurobiol 1993;41:323–344.ArticlePubMed
- 4. Takakusaki K, Takahashi K, Saitoh K, Harada H, Okumura T, Kayama Y, et al. Orexinergic projections to the cat midbrain mediate alternation of emotional behavioural states from locomotion to cataplexy. J Physiol 2005;568(Pt 3):1003–1020.ArticlePubMedPMC
- 5. Takakusaki K, Tomita N, Yano M. Substrates for normal gait and pathophysiology of gait disturbances with respect to the basal ganglia dysfunction. J Neurol 2008;255 Suppl 4:19–29.ArticlePubMed
- 6. Takakusaki K. Neurophysiology of gait: from the spinal cord to the frontal lobe. Mov Disord 2013;28:1483–1491.ArticlePubMed
- 7. DeLong MR, Wichmann T. Circuits and circuit disorders of the basal ganglia. Arch Neurol 2007;64:20–24.ArticlePubMed
- 8. Armstrong DM. Supraspinal contributions to the initiation and control of locomotion in the cat. Prog Neurobiol 1986;26:273–361.ArticlePubMed
- 9. Mori S. Integration of posture and locomotion in acute decerebrate cats and in awake, freely moving cats. Prog Neurobiol 1987;28:161–195.ArticlePubMed
- 10. Hinsey JC, Ranson SW, McNattin RF. The role of the hypothalamus and mesencephalon in locomotion. Arch NeurPsych 1930;23:1–43.Article
- 11. Shik ML, Orlovsky GN. Neurophysiology of locomotor automatism. Physiol Rev 1976;56:465–501.ArticlePubMed
- 12. Mori S, Matsui T, Kuze B, Asanome M, Nakajima K, Matsuyama K. Stimulation of a restricted region in the midline cerebellar white matter evokes coordinated quadrupedal locomotion in the decerebrate cat. J Neurophysiol 1999;82:290–300.ArticlePubMed
- 13. Jahn K, Deutschländer A, Stephan T, Kalla R, Wiesmann M, Strupp M, et al. Imaging human supraspinal locomotor centers in brainstem and cerebellum. Neuroimage 2008;39:786–792.ArticlePubMed
- 14. Grillner S, Georgopoulos AP, Jordan LM. Selection and initiation of motor behavior. In: Stein PSG, Grillner S, Selverston AI, Stuart DG, editors. Neurons, networks, and motor behavior. Cambridge, MA: The MIT Press; 1997:3–19.
- 15. Takakusaki K, Chiba R, Nozu T, Okumura T. Brainstem control of locomotion and muscle tone with special reference to the role of the mesopontine tegmentum and medullary reticulospinal systems. J Neural Transm (Vienna) 2016;123:695–729.ArticlePubMedPMCPDF
- 16. Mena-Segovia J. Structural and functional considerations of the cholinergic brainstem. J Neural Transm (Vienna) 2016;123:731–736.ArticlePubMedPDF
- 17. Petzold A, Valencia M, Pál B, Mena-Segovia J. Decoding brain state transitions in the pedunculopontine nucleus: cooperative phasic and tonic mechanisms. Front Neural Circuits 2015;9:68.ArticlePubMedPMC
- 18. Takakusaki K, Habaguchi T, Ohtinata-Sugimoto J, Saitoh K, Sakamoto T. Basal ganglia efferents to the brainstem centers controlling postural muscle tone and locomotion: a new concept for understanding motor disorders in basal ganglia dysfunction. Neuroscience 2003;119:293–308.ArticlePubMed
- 19. Takakusaki K, Habaguchi T, Saitoh K, Kohyama J. Changes in the excitability of hindlimb motoneurons during muscular atonia induced by stimulating the pedunculopontine tegmental nucleus in cats. Neuroscience 2004;124:467–480.ArticlePubMed
- 20. Takakusaki K, Obara K, Nozu T, Okumura T. Modulatory effects of the GABAergic basal ganglia neurons on the PPN and the muscle tone inhibitory system in cats. Arch Ital Biol 2011;149:385–405.PubMed
- 21. Sherman D, Fuller PM, Marcus J, Yu J, Zhang P, Chamberlin NL, et al. Anatomical location of the mesencephalic locomotor region and its possible role in locomotion, posture, cataplexy, and parkinsonism. Front Neurol 2015;6:140.ArticlePubMedPMC
- 22. Kuze B, Matsuyama K, Matsui T, Miyata H, Mori S. Segment-specific branching patterns of single vestibulospinal tract axons arising from the lateral vestibular nucleus in the cat: a PHA-L tracing study. J Comp Neurol 1999;414:80–96.ArticlePubMed
- 23. Matsuyama K, Takakusaki K, Nakajima K, Mori S. Multisegmental innervation of single pontine reticulospinal axons in the cervico-thoracic region of the cat: anterograde PHA-L tracing study. J Comp Neurol 1997;377:234–250.ArticlePubMed
- 24. Matsuyama K, Drew T. Vestibulospinal and reticulospinal neuronal activity during locomotion in the intact cat. I. Walking on a level surface. J Neurophysiol 2000;84:2237–2256.ArticlePubMed
- 25. Matsuyama K, Drew T. Vestibulospinal and reticulospinal neuronal activity during locomotion in the intact cat. II. Walking on an inclined plane. J Neurophysiol 2000;84:2257–2276.ArticlePubMed
- 26. Rossignol S. Neural control of stereotypic limb movements. In: Rowell LB, Sheperd JT, editors. Handbook of physiology, section 12. Exercise: regulation and integration of multiple systems. New York, NY: Oxford University Press; 1996:173–216.
- 27. Mori S, Matsui T, Kuze B, Asanome M, Nakajima K, Matsuyama K. Cerebellar-induced locomotion: reticulospinal control of spinal rhythm generating mechanism in cats. Ann N Y Acad Sci 1998;860:94–105.ArticlePubMed
- 28. Fung SJ, Barnes CD. Evidence of facilitatory coerulospinal action in lumbar motoneurons of cats. Brain Res 1981;216:299–311.ArticlePubMed
- 29. Mori S, Kawahara K, Sakamoto T, Aoki M, Tomiyama T. Setting and resetting of level of postural muscle tone in decerebrate cat by stimulation of brain stem. J Neurophysiol 1982;48:737–748.ArticlePubMed
- 30. Sakai M, Matsunaga M, Kubota A, Yamanishi Y, Nishizawa Y. Reduction in excessive muscle tone by selective depletion of serotonin in intercollicularly decerebrated rats. Brain Res 2000;860:104–111.ArticlePubMed
- 31. Takakusaki K, Saitoh K, Harada H, Okumura T, Sakamoto T. Evidence for a role of basal ganglia in the regulation of rapid eye movement sleep by electrical and chemical stimulation for the pedunculopontine tegmental nucleus and the substantia nigra pars reticulata in decerebrate cats. Neuroscience 2004;124:207–220.ArticlePubMed
- 32. Sławińska U, Kasicki S. Theta-like rhythm in depth EEG activity of hypothalamic areas during spontaneous or electrically induced locomotion in the rat. Brain Res 1995;678:117–126.ArticlePubMed
- 33. Swanson LW, Mogenson GJ. Neural mechanisms for the functional coupling of autonomic, endocrine and somatomotor responses in adaptive behavior. Brain Res 1981;228:1–34.ArticlePubMed
- 34. Lynd-Balta E, Haber SN. Primate striatonigral projections: a comparison of the sensorimotor-related striatum and the ventral striatum. J Comp Neurol 1994;345:562–578.ArticlePubMed
- 35. Robertson B, Kardamakis A, Capantini L, Pérez-Fernández J, Suryanarayana SM, Wallén P, et al. The lamprey blueprint of the mammalian nervous system. Prog Brain Res 2014;212:337–349.ArticlePubMed
- 36. Rossignol S, Dubuc R, Gossard JP. Dynamic sensorimotor interactions in locomotion. Physiol Rev 2006;86:89–154.ArticlePubMed
- 37. Rossignol S, Barrière G, Frigon A, Barthélemy D, Bouyer L, Provencher J, et al. Plasticity of locomotor sensorimotor interactions after peripheral and/or spinal lesions. Brain Res Rev 2008;57:228–240.ArticlePubMed
- 38. McCrea DA, Rybak IA. Organization of mammalian locomotor rhythm and pattern generation. Brain Res Rev 2008;57:134–46.ArticlePubMedPMC
- 39. Jankowska E. Interneuronal relay in spinal pathways from proprioceptors. Prog Neurobiol 1992;38:335–378.ArticlePubMed
- 40. Matsuyama K, Takakusaki K. Organizing principles of axonal projections of the long descending reticulospinal pathway and its target spinal lamina VIII commissural neurons: with special reference to the locomotor function. In: Westland TB, Calton RN, editors. Handbook on White Matter: Structure, Function and Changes. New York, NY: Nova Science Publishing; 2009:335–356.
- 41. Pearson KG. Generating the walking gait: role of sensory feedback. Prog Brain Res 2004;143:123–129.ArticlePubMed
- 42. Frigon A, Sirois J, Gossard JP. Effects of ankle and hip muscle afferent inputs on rhythm generation during fictive locomotion. J Neurophysiol 2010;103:1591–1605.ArticlePubMed
- 43. Forssberg H. Stumbling corrective reaction: a phase-dependent compensatory reaction during locomotion. J Neurophysiol 1979;42:936–953.ArticlePubMed
- 44. Bjursten LM, Norrsell K, Norrsell U. Behavioural repertory of cats without cerebral cortex from infancy. Exp Brain Res 1976;25:115–130.ArticlePubMed
- 45. Adkins RJ, Cegnar MR, Rafuse DD. Differential effects of lesions of the anterior and posterior sigmoid gyri in cats. Brain Res 1971;30:411–414.ArticlePubMed
- 46. Villablanca JR. Why do we have a caudate nucleus? Acta Neurobiol Exp (Wars) 2010;70:95–105.ArticlePubMedPDF
- 47. Denny-Brown D. The midbrain and motor integration. Proc R Soc Med 1962;55:527–538.ArticlePubMedPMC
- 48. Villablanca J, Marcus R. Sleep-wakefulness, EEG and behavioral studies of chronic cats without neocortex and striatum: the ‘diencephalic’ cat. Arch Ital Biol 1972;110:348–382.PubMed
- 49. Eidelberg E, Yu J. Effects of corticospinal lesions upon treadmill locomotion by cats. Exp Brain Res 1981;43:101–103.ArticlePubMed
- 50. Liddell EGT, Phillips CG. Pyramidal section in the cat. Brain 1944;67:1–9.ArticlePDF
- 51. Brooks VB, Stoney SD Jr. Motor mechanisms: the role of the pyramidal system in motor control. Annu Rev Physiol 1971;33:337–392.ArticlePubMed
- 52. Vicario DS, Martin JH, Ghez C. Specialized subregions in the cat motor cortex: a single unit analysis in the behaving animal. Exp Brain Res 1983;51:351–367.Article
- 53. Maurer C, Mergner T, Peterka RJ. Multisensory control of human upright stance. Exp Brain Res 2006;171:231–250.ArticlePubMed
- 54. Mergner T, Becker W. A modeling approach to the human spatial orientation system. Ann N Y Acad Sci 2003;1004:303–315.ArticlePubMed
- 55. Horak FB, Shupert CL, Dietz V, Horstmann G. Vestibular and somatosensory contributions to responses to head and body displacements in stance. Exp Brain Res 1994;100:93–106.ArticlePubMed
- 56. Shaikh AG, Meng H, Angelaki DE. Multiple reference frames for motion in the primate cerebellum. J Neurosci 2004;24:4491–4497.ArticlePubMedPMC
- 57. Kravitz DJ, Saleem KS, Baker CI, Mishkin M. A new neural framework for visuospatial processing. Nat Rev Neurosci 2011;12:217–230.ArticlePubMedPMCPDF
- 58. Georgopoulos AP, Grillner S. Visuomotor coordination in reaching and locomotion. Science 1989;245:1209–1210.ArticlePubMed
- 59. Lajoie K, Andujar JE, Pearson K, Drew T. Neurons in area 5 of the posterior parietal cortex in the cat contribute to interlimb coordination during visually guided locomotion: a role in working memory. J Neurophysiol 2010;103:2234–2254.ArticlePubMed
- 60. Marigold DS, Drew T. Contribution of cells in the posterior parietal cortex to the planning of visually guided locomotion in the cat: effects of temporary visual interruption. J Neurophysiol 2011;105:2457–2470.ArticlePubMed
- 61. Massion J. Movement, posture and equilibrium: interaction and coordination. Prog Neurobiol 1992;38:35–56.ArticlePubMed
- 62. Nakajima K, Mori F, Tachibana A, Nambu A, Mori S. Cortical mechanisms for the control of bipedal locomotion in Japanese monkeys: I. Local inactivation of the primary motor cortex (M1). Neurosci Res 2003;46(Suppl 1):S156.
- 63. Mori F, Nakajima K, Tachibana A, Nambu A, Mori S. Cortical mechanisms for the control of bipedal locomotion in Japanese monkeys: II. Local inactivation of the supplementary motor area (SMA). Neurosci Res 2003;46(Suppl 1):S157.
- 64. Matsuyama K, Drew T. Organization of the projections from the pericruciate cortex to the pontomedullary brainstem of the cat: a study using the anterograde tracer Phaseolus vulgaris-leucoagglutinin. J Comp Neurol 1997;389:617–641.ArticlePubMed
- 65. Keizer K, Kuypers HG. Distribution of corticospinal neurons with collaterals to the lower brain stem reticular formation in monkey (Macaca fascicularis). Exp Brain Res 1989;74:311–318.ArticlePubMed
- 66. Aravamuthan BR, McNab JA, Miller KL, Rushworth M, Jenkinson N, Stein JF, et al. Cortical and subcortical connections within the pedunculopontine nucleus of the primate Macaca mulatta determined using probabilistic diffusion tractography. J Clin Neurosci 2009;16:413–420.ArticlePubMed
- 67. Jacobs JV, Lou JS, Kraakevik JA, Horak FB. The supplementary motor area contributes to the timing of the anticipatory postural adjustment during step initiation in participants with and without Parkinson’s disease. Neuroscience 2009;164:877–885.ArticlePubMedPMC
- 68. Hoshi E, Tanji J. Distinctions between dorsal and ventral premotor areas: anatomical connectivity and functional properties. Curr Opin Neurobiol 2007;17:234–242.ArticlePubMed
- 69. Snijders AH, van de Warrenburg BP, Giladi N, Bloem BR. Neurological gait disorders in elderly people: clinical approach and classification. Lancet Neurol 2007;6:63–74.ArticlePubMed
- 70. Cohen RG, Nutt JG, Horak FB. Errors in postural preparation lead to increased choice reaction times for step initiation in older adults. J Gerontol A Biol Sci Med Sci 2011;66:705–713.ArticlePubMedPMCPDF
- 71. Barbieri G, Gissot AS, Fouque F, Casillas JM, Pozzo T, Pérennou D. Does proprioception contribute to the sense of verticality? Exp Brain Res 2008;185:545–552.ArticlePubMed
- 72. Bisdorff AR, Wolsley CJ, Anastasopoulos D, Bronstein AM, Gresty MA. The perception of body verticality (subjective postural vertical) in peripheral and central vestibular disorders. Brain 1996;119(Pt 5):1523–1534.ArticlePubMedPDF
- 73. Merfeld DM, Zupan L, Peterka RJ. Humans use internal models to estimate gravity and linear acceleration. Nature 1999;398:615–618.ArticlePubMedPDF
- 74. Van Beuzekom AD, Van Gisbergen JA. Properties of the internal representation of gravity inferred from spatial-direction and body-tilt estimates. J Neurophysiol 2000;84:11–27.ArticlePubMed
- 75. Pérennou DA, Mazibrada G, Chauvineau V, Greenwood R, Rothwell J, Gresty MA, et al. Lateropulsion, pushing and verticality perception in hemisphere stroke: a causal relationship? Brain 2008;131(Pt 9):2401–2413.ArticlePubMedPDF
- 76. Lopez C, Falconer CJ, Deroualle D, Mast FW. In the presence of others: self-location, balance control and vestibular processing. Neurophysiol Clin 2015;45:241–254.ArticlePubMed
- 77. Akbarian S, Grüsser OJ, Guldin WO. Thalamic connections of the vestibular cortical fields in the squirrel monkey (Saimiri sciureus). J Comp Neurol 1992;326:423–441.ArticlePubMed
- 78. Lopez C, Blanke O. The thalamocortical vestibular system in animals and humans. Brain Res Rev 2011;67:119–146.ArticlePubMed
- 79. Fukushima K. Corticovestibular interactions: anatomy, electrophysiology, and functional considerations. Exp Brain Res 1997;117:1–16.ArticlePubMed
- 80. Faugier-Grimaud S, Ventre J. Anatomic connections of inferior parietal cortex (area 7) with subcortical structures related to vestibulo-ocular function in a monkey (Macaca fascicularis). J Comp Neurol 1989;280:1–14.ArticlePubMed
- 81. Guldin WO, Grüsser OJ. Is there a vestibular cortex? Trends Neurosci 1998;21:254–259.ArticlePubMed
- 82. Brandt T, Dieterich M. The vestibular cortex. Its locations, functions, and disorders. Ann N Y Acad Sci 1999;871:293–312.ArticlePubMed
- 83. Sugiuchi Y, Izawa Y, Ebata S, Shinoda Y. Vestibular cortical area in the periarcuate cortex: its afferent and efferent projections. Ann N Y Acad Sci 2005;1039:111–123.ArticlePubMed
- 84. Barra J, Marquer A, Joassin R, Reymond C, Metge L, Chauvineau V, et al. Humans use internal models to construct and update a sense of verticality. Brain 2010;133(Pt 12):3552–3563.ArticlePubMedPDF
- 85. Maffei V, Mazzarella E, Piras F, Spalletta G, Caltagirone C, Lacquaniti F, et al. Processing of visual gravitational motion in the peri-sylvian cortex: evidence from brain-damaged patients. Cortex 2016;78:55–69.ArticlePubMed
- 86. Zhang LL, Wang JQ, Qi RR, Pan LL, Li M, Cai YL. Motion sickness: current knowledge and recent advance. CNS Neurosci Ther 2016;22:15–24.ArticlePubMed
- 87. Pérennou D, Piscicelli C, Barbieri G, Jaeger M, Marquer A, Barra J. Measuring verticality perception after stroke: why and how? Neurophysiol Clin 2014;44:25–32.ArticlePubMed
- 88. Karnath HO, Dieterich M. Spatial neglect--a vestibular disorder? Brain 2006;129(Pt 2):293–305.ArticlePubMedPDF
- 89. Wilson VJ, Zarzecki P, Schor RH, Isu N, Rose PK, Sato H, et al. Cortical influences on the vestibular nuclei of the cat. Exp Brain Res 1999;125:1–13.ArticlePubMed
- 90. Akbarian S, Grüsser OJ, Guldin WO. Corticofugal connections between the cerebral cortex and brainstem vestibular nuclei in the macaque monkey. J Comp Neurol 1994;339:421–437.ArticlePubMed
- 91. Akbarian S, Grüsser OJ, Guldin WO. Corticofugal projections to the vestibular nuclei in squirrel monkeys: further evidence of multiple cortical vestibular fields. J Comp Neurol 1993;332:89–104.ArticlePubMed
- 92. Ticini LF, Klose U, Nägele T, Karnath HO. Perfusion imaging in pusher syndrome to investigate the neural substrates involved in controlling upright body position. PLoS One 2009;4:e5737. ArticlePubMedPMC
- 93. Stecina K, Fedirchuk B, Hultborn H. Information to cerebellum on spinal motor networks mediated by the dorsal spinocerebellar tract. J Physiol 2013;591:5433–5443.ArticlePubMedPMC
- 94. Büttner U, Glasauer S, Glonti L, Guan Y, Kipiani E, Kleine J, et al. Multimodal signal integration in vestibular neurons of the primate fastigial nucleus. Ann N Y Acad Sci 2003;1004:241–251.ArticlePubMed
- 95. McCall AA, Miller DJ, Catanzaro MF, Cotter LA, Yates BJ. Hindlimb movement modulates the activity of rostral fastigial nucleus neurons that process vestibular input. Exp Brain Res 2015;233:2411–2419.ArticlePubMedPMC
- 96. Cavdar S, Onat FY, Yananli HR, Sehirli US, Tulay C, Saka E, et al. Cerebellar connections to the rostral reticular nucleus of the thalamus in the rat. J Anat 2002;201:485–491.ArticlePubMedPMC
- 97. Asanome M, Matsuyama K, Mori S. Augmentation of postural muscle tone induced by the stimulation of the descending fibers in the midline area of the cerebellar white matter in the acute decerebrate cat. Neurosci Res 1998;30:257–269.ArticlePubMed
- 98. Llinas R. Mechanisms of supraspinal actions upon spinal cord activities. Differences between reticular and cerebellar inhibitory actions upon alpha extensor motoneurons. J Neurophysiol 1964;27:1117–1126.ArticlePubMed
- 99. Eccles JC, Nicoll RA, Schwarz WF, Táboriková H, Willey TJ. Reticulospinal neurons with and without monosynaptic inputs from cerebellar nuclei. J Neurophysiol 1975;38:513–530.ArticlePubMed
- 100. Fukushima K, Peterson BW, Uchino Y, Coulter JD, Wilson VJ. Direct fastigiospinal fibers in the cat. Brain Res 1977;126:538–542.ArticlePubMed
- 101. Bostan AC, Dum RP, Strick PL. Cerebellar networks with the cerebral cortex and basal ganglia. Trends Cogn Sci 2013;17:241–254.ArticlePubMedPMC
- 102. Sasaki K, Kawaguchi S, Oka H, Sakai M, Mizuno N. Electrophysiological studies on the cerebellocerebral projections in monkeys. Exp Brain Res 1976;24:495–507.ArticlePubMed
- 103. Amino Y, Kyuhou S, Matsuzaki R, Gemba H. Cerebellothalamo-cortical projections to the posterior parietal cortex in the macaque monkey. Neurosci Lett 2001;309:29–32.ArticlePubMed
- 104. Kyuhou S, Kawaguchi S. Cerebellocerebral projection from the fastigial nucleus onto the frontal eye field and anterior ectosylvian visual area in the cat. J Comp Neurol 1987;259:571–590.ArticlePubMed
- 105. Middleton FA, Strick PL. Basal ganglia and cerebellar loops: motor and cognitive circuits. Brain Res Rev 2000;31:236–250.ArticlePubMed
- 106. Hikosaka O. GABAergic output of the basal ganglia. Prog Brain Res 2007;160:209–226.ArticlePubMed
- 107. Tarnutzer AA, Marti S, Straumann D. Gravity perception in cerebellar patients. Prog Brain Res 2008;171:369–372.ArticlePubMed
- 108. Timmann D, Horak FB. Perturbed step initiation in cerebellar subjects: 2. Modification of anticipatory postural adjustments. Exp Brain Res 2001;141:110–120.ArticlePubMed
- 109. Bostan AC, Dum RP, Strick PL. The basal ganglia communicate with the cerebellum. Proc Natl Acad Sci U S A 2010;107:8452–8456.ArticlePubMedPMC
- 110. Brown LL, Schneider JS, Lidsky TI. Sensory and cognitive functions of the basal ganglia. Curr Opin Neurobiol 1997;7:157–163.ArticlePubMed
- 111. Bloem BR, Valkenburg VV, Slabbekoorn M, van Dijk JG. The multiple tasks test. Strategies in Parkinson’s disease. Exp Brain Res 2001;137:478–486.ArticlePubMed
- 112. Bhatia KP, Marsden CD. The behavioural and motor consequences of focal lesions of the basal ganglia in man. Brain 1994;117(Pt 4):859–876.ArticlePubMedPDF
- 113. Takakusaki K, Saitoh K, Harada H, Kashiwayanagi M. Role of basal ganglia-brainstem pathways in the control of motor behaviors. Neurosci Res 2004;50:137–151.ArticlePubMed
- 114. Filion M, Tremblay L. Abnormal spontaneous activity of globus pallidus neurons in monkeys with MPTP-induced parkinsonism. Brain Res 1991;547:142–151.ArticlePubMed
- 115. Nambu A. Seven problems on the basal ganglia. Curr Opin Neurobiol 2008;18:595–604.ArticlePubMed
- 116. Bohnen NI, Albin RL. The cholinergic system and Parkinson disease. Behav Brain Res 2011;221:564–573.ArticlePubMedPMC
- 117. Hirsch EC, Graybiel AM, Duyckaerts C, Javoy-Agid F. Neuronal loss in the pedunculopontine tegmental nucleus in Parkinson disease and in progressive supranuclear palsy. Proc Natl Acad Sci U S A 1987;84:5976–5980.ArticlePubMedPMC
- 118. Jellinger K. The pedunculopontine nucleus in Parkinson’s disease, progressive supranuclear palsy and Alzheimer’s disease. J Neurol Neurosurg Psychiatry 1988;51:540–543.ArticlePubMedPMC
- 119. Zweig RM, Jankel WR, Hedreen JC, Mayeux R, Price DL. The pedunculopontine nucleus in Parkinson’s disease. Ann Neurol 1989;26:41–46.ArticlePubMed
- 120. Müller ML, Albin RL, Kotagal V, Koeppe RA, Scott PJ, Frey KA, et al. Thalamic cholinergic innervation and postural sensory integration function in Parkinson’s disease. Brain 2013;136(Pt 11):3282–3289.ArticlePubMedPMCPDF
- 121. Jones BE. From waking to sleeping: neuronal and chemical substrates. Trends Pharmacol Sci 2005;26:578–586.ArticlePubMed
- 122. Steriade M, McCormick DA, Sejnowski TJ. Thalamocortical oscillations in the sleeping and aroused brain. Science 1993;262:679–685.ArticlePubMed
- 123. Winn P. Experimental studies of pedunculopontine functions: are they motor, sensory or integrative? Parkinsonism Relat Disord 2008;14 Suppl 2:S194–S198.ArticlePubMed
- 124. Dautan D, Huerta-Ocampo I, Witten IB, Deisseroth K, Bolam JP, Gerdjikov T, et al. A major external source of cholinergic innervation of the striatum and nucleus accumbens originates in the brainstem. J Neurosci 2014;34:4509–4518.ArticlePubMedPMC
- 125. Mena-Segovia J, Winn P, Bolam JP. Cholinergic modulation of midbrain dopaminergic systems. Brain Res Rev 2008;58:265–271.ArticlePubMed
- 126. Takakusaki K, Shiroyama T, Yamamoto T, Kitai ST. Cholinergic and noncholinergic tegmental pedunculopontine projection neurons in rats revealed by intracellular labeling. J Comp Neurol 1996;371:345–361.ArticlePubMed
- 127. Hasselmo ME, Sarter M. Modes and models of forebrain cholinergic neuromodulation of cognition. Neuropsychopharmacology 2011;36:52–73.ArticlePubMedPMCPDF
- 128. Hanakawa T, Fukuyama H, Katsumi Y, Honda M, Shibasaki H. Enhanced lateral premotor activity during paradoxical gait in Parkinson’s disease. Ann Neurol 1999;45:329–336.ArticlePubMed
- 129. Hanakawa T, Katsumi Y, Fukuyama H, Honda M, Hayashi T, Kimura J, et al. Mechanisms underlying gait disturbance in Parkinson’s disease: a single photon emission computed tomography study. Brain 1999;122(Pt 7):1271–1282.ArticlePubMedPDF
- 130. Bohnen NI, Müller ML, Koeppe RA, Studenski SA, Kilbourn MA, Frey KA, et al. History of falls in Parkinson disease is associated with reduced cholinergic activity. Neurology 2009;73:1670–1676.ArticlePubMedPMC
- 131. Müller ML, Bohnen NI. Cholinergic dysfunction in Parkinson’s disease. Curr Neurol Neurosci Rep 2013;13:377.ArticlePubMedPMC
- 132. Scocco DH, Wagner JN, Racosta J, Chade A, Gershanik OS. Subjective visual vertical in Pisa syndrome. Parkinsonism Relat Disord 2014;20:878–883.ArticlePubMed
- 133. Vitale C, Marcelli V, Furia T, Santangelo G, Cozzolino A, Longo K, et al. Vestibular impairment and adaptive postural imbalance in parkinsonian patients with lateral trunk flexion. Mov Disord 2011;26:1458–1463.ArticlePubMed
- 134. Villarejo A, Camacho A, García-Ramos R, Moreno T, Penas M, Juntas R, et al. Cholinergic-dopaminergic imbalance in Pisa syndrome. Clin Neuropharmacol 2003;26:119–121.ArticlePubMed
- 135. Doherty KM, van de Warrenburg BP, Peralta MC, SilveiraMoriyama L, Azulay JP, Gershanik OS, et al. Postural deformities in Parkinson’s disease. Lancet Neurol 2011;10:538–549.ArticlePubMed
- 136. Vaugoyeau M, Azulay JP. Role of sensory information in the control of postural orientation in Parkinson’s disease. J Neurol Sci 2010;289:66–68.ArticlePubMed
- 137. Vaugoyeau M, Viel S, Assaiante C, Amblard B, Azulay JP. Impaired vertical postural control and proprioceptive integration deficits in Parkinson’s disease. Neuroscience 2007;146:852–863.ArticlePubMed
- 138. Pollak L, Prohorov T, Kushnir M, Rabey M. Vestibulocervical reflexes in idiopathic Parkinson disease. Neurophysiol Clin 2009;39:235–240.ArticlePubMed
- 139. Michel SF, Arias Carrión O, Correa TE, Alejandro PL, Micheli F. Pisa syndrome. Clin Neuropharmacol 2015;38:135–140.ArticlePubMed
- 140. Solla P, Cannas A, Congia S, Floris G, Aste R, Tacconi P, et al. Levodopa/carbidopa/entacapone-induced acute Pisa syndrome in a Parkinson’s disease patient. J Neurol Sci 2008;275:154–156.ArticlePubMed
- 141. van de Warrenburg BP, Bhatia KP, Quinn NP. Pisa syndrome after unilateral pallidotomy in Parkinson’s disease: an unrecognised, delayed adverse event? J Neurol Neurosurg Psychiatry 2007;78:329–330.ArticlePubMedPMC
- 142. Spanaki C, Zafeiris S, Plaitakis A. Levodopa-aggravated lateral flexion of the neck and trunk as a delayed phenomenon of unilateral pallidotomy. Mov Disord 2010;25:655–656.ArticlePubMed
- 143. Mirdamadi JL. Cerebellar role in Parkinson’s disease. J Neurophysiol 2016;116:917–919.ArticlePubMedPMC
- 144. Lewis MM, Galley S, Johnson S, Stevenson J, Huang X, McKeown MJ. The role of the cerebellum in the pathophysiology of Parkinson’s disease. Can J Neurol Sci 2013;40:299–306.ArticlePubMedPMC
- 145. Roseberry TK, Lee AM, Lalive AL, Wilbrecht L, Bonci A, Kreitzer AC. Cell-type-specific control of brainstem locomotor circuits by basal ganglia. Cell 2016;164:526–537.ArticlePubMedPMC
- 146. Castrioto A, Piscicelli C, Perennou D, Krack P, Debu B. The pathogenesis of Pisa syndrome in Parkinson’s disease. Mov Disord 2014;29:1100–1117.ArticlePubMed
- 147. Karachi C, Grabli D, Bernard FA, Tandé D, Wattiez N, Belaid H, et al. Cholinergic mesencephalic neurons are involved in gait and postural disorders in Parkinson disease. J Clin Invest 2010;120:2745–2754.ArticlePubMedPMC
- 148. Rinne JO, Ma SY, Lee MS, Collan Y, Röyttä M. Loss of cholinergic neurons in the pedunculopontine nucleus in Parkinson’s disease is related to disability of the patients. Parkinsonism Relat Disord 2008;14:553–557.ArticlePubMed
- 149. Piallat B, Chabardès S, Torres N, Fraix V, Goetz L, Seigneuret E, et al. Gait is associated with an increase in tonic firing of the sub-cuneiform nucleus neurons. Neuroscience 2009;158:1201–1205.ArticlePubMed
- 150. Thevathasan W, Pogosyan A, Hyam JA, Jenkinson N, Foltynie T, Limousin P, et al. Alpha oscillations in the pedunculopontine nucleus correlate with gait performance in parkinsonism. Brain 2012;135(Pt 1):148–160.ArticlePubMedPMCPDF
- 151. Benarroch EE. Pedunculopontine nucleus: functional organization and clinical implications. Neurology 2013;80:1148–1155.ArticlePubMed
- 152. Hamani C, Moro E, Lozano AM. The pedunculopontine nucleus as a target for deep brain stimulation. J Neural Transm (Vienna) 2011;118:1461–1468.ArticlePubMed
- 153. Pereira EA, Nandi D, Jenkinson N, Stein JF, Green AL, Aziz TZ. Pedunculopontine stimulation from primate to patient. J Neural Transm (Vienna) 2011;118:1453–1460.ArticlePubMed
- 154. Stefani A, Lozano AM, Peppe A, Stanzione P, Galati S, Tropepi D, et al. Bilateral deep brain stimulation of the pedunculopontine and subthalamic nuclei in severe Parkinson’s disease. Brain 2007;130:1596–1607.ArticlePubMedPDF
- 155. Mazzone P, Sposato S, Insola A, Scarnati E. The clinical effects of deep brain stimulation of the pedunculopontine tegmental nucleus in movement disorders may not be related to the anatomical target, leads location, and setup of electrical stimulation. Neurosurgery 2013;73:894–906.discussion 905-906. ArticlePubMedPDF
- 156. Ferraye MU, Debû B, Fraix V, Goetz L, Ardouin C, Yelnik J, et al. Effects of pedunculopontine nucleus area stimulation on gait disorders in Parkinson’s disease. Brain 2010;133(Pt 1):205–214.ArticlePubMedPDF
- 157. Moro E, Hamani C, Poon YY, Al-Khairallah T, Dostrovsky JO, Hutchison WD, et al. Unilateral pedunculopontine stimulation improves falls in Parkinson’s disease. Brain 2010;133(Pt 1):215–224.ArticlePubMedPDF
- 158. Mazzone P, Scarnati E, Garcia-Rill E. Commentary: the pedunculopontine nucleus: clinical experience, basic questions and future directions. J Neural Transm (Vienna) 2011;118:1391–1396.ArticlePubMedPMC
- 159. Mazzone P, Vilela Filho O, Viselli F, Insola A, Sposato S, Vitale F, et al. Our first decade of experience in deep brain stimulation of the brainstem: elucidating the mechanism of action of stimulation of the ventrolateral pontine tegmentum. J Neural Transm (Vienna) 2016;123:751–767.ArticlePubMedPDF
- 160. Alam M, Schwabe K, Krauss JK. The pedunculopontine nucleus area: critical evaluation of interspecies differences relevant for its use as a target for deep brain stimulation. Brain 2011;134(Pt 1):11–23.ArticlePubMedPDF
- 161. Karachi C, André A, Bertasi E, Bardinet E, Lehéricy S, Bernard FA. Functional parcellation of the lateral mesencephalus. J Neurosci 2012;32:9396–9401.ArticlePubMedPMC
- 162. Fling BW, Cohen RG, Mancini M, Carpenter SD, Fair DA, Nutt JG, et al. Functional reorganization of the locomotor network in Parkinson patients with freezing of gait. PLoS One 2014;9:e100291.ArticlePubMedPMC
- 163. Schweder PM, Hansen PC, Green AL, Quaghebeur G, Stein J, Aziz TZ. Connectivity of the pedunculopontine nucleus in parkinsonian freezing of gait. Neuroreport 2010;21:914–916.ArticlePubMed
- 164. Snijders AH, Takakusaki K, Debu B, Lozano AM, Krishna V, Fasano A, et al. Physiology of freezing of gait. Ann Neurol 2016;80:644–659.ArticlePubMed
- 165. Takakusaki K, Takahashi M, Obara K, Chiba R. Neural substrates involved in the control of posture. Adv Robot 2016;Dec. 6. [Epub]. http://dx.doi.org/10.1080/01691864.2016.1252690. ArticlePubMedPMC
REFERENCES
Figure & Data
References
Citations
Citations to this article as recorded by 

- Imagined Timed Up and Go test (iTUG) in people with Parkinson’s Disease: test-retest reliability and validity
Marcos Moreno-Verdú, María del Rosario Ferreira-Sánchez, Patricia Martín-Casas, María Ángeles Atín-Arratibel
Disability and Rehabilitation.2024; 46(5): 1005. CrossRef - Comparison of the effects of rhythmic vibrotactile stimulations and rhythmic auditory stimulations on Parkinson's disease patients’ gait variability: a pilot study
Alexis Lheureux, Thierry Lejeune, Ivan Doncev, Alix Jeanne, Gaëtan Stoquart
Acta Neurologica Belgica.2024; 124(1): 161. CrossRef - Changes in vestibular-related responses to combined noisy galvanic vestibular stimulation and cerebellar transcranial direct current stimulation
Tsubasa Mitsutake, Hisato Nakazono, Tomoyuki Shiozaki, Daisuke Fujita, Maiko Sakamoto
Experimental Brain Research.2024; 242(1): 99. CrossRef - Multisensory Conflict Impairs Cortico-Muscular Network Connectivity and Postural Stability: Insights from Partial Directed Coherence Analysis
Guozheng Wang, Yi Yang, Kangli Dong, Anke Hua, Jian Wang, Jun Liu
Neuroscience Bulletin.2024; 40(1): 79. CrossRef - Correlations between plantar pressure and postural balance in healthy subjects and their comparison according to gender and limb dominance: A cross-sectional descriptive study
Gamze Arin-Bal, Ayse Livanelioglu, Alberto Leardini, Claudio Belvedere
Gait & Posture.2024; 108: 124. CrossRef - The effects of P2 segment of posterior cerebral artery to thalamus blood supply pattern on gait in cerebral small vessel disease: A 7 T MRI based study
Cen Guo, Bei Wang, Yajing Huo, Lili Shan, Tianci Qiao, Zidong Yang, Mingyuan Liu, Yilin Wang, Guanshu Liu, Yajie Liu, He Wang, Yan Han
Neurobiology of Disease.2024; 190: 106372. CrossRef - The relationship between depressive symptoms and physical performance in postmenopausal women: a cross-sectional investigation
Rosely M. Silva, Fábio L. Orsatti, Luana R. Margato, Wellington G. de Sousa, Danyelle C. S. Pelet, Jonathã Luiz Justino da Silva, Anderson D. de Sousa Lino, Cláudio de Oliveira Assumpção, Markus Vinicius C. Souza
Menopause.2024; 31(2): 101. CrossRef - Association between depressive symptoms and dynamic balance among young adults in the community
Jinyoung Park, Cynthia Lee, Ye Eun Nam, Hyunhwa Lee
Heliyon.2024; 10(2): e24093. CrossRef - Cross-Cultural Adaptation and Psychometric Characteristics of the Greek Functional Gait Assessment Scale in Healthy Community-Dwelling Older Adults
Sofia Lampropoulou, Anthi Kellari, Ingrid A. Gedikoglou, Danai Gagara Kozonaki, Polymnia Nika, Vasiliki Sakellari
Applied Sciences.2024; 14(2): 520. CrossRef - Analysis of muscle synergy and gait kinematics during regain of gait function through rehabilitation in a monoplegic patient
Akira Ebihara, Mitsuki Hirota, Yasuhiro Kumakura, Masanori Nagaoka
Frontiers in Human Neuroscience.2024;[Epub] CrossRef - Age and Type of Task-Based Impact of Mental Fatigue on Balance: Systematic Review and Meta-Analysis
Abubakar Tijjani Salihu, Keith D. Hill, Shapour Jaberzadeh
Journal of Motor Behavior.2024; 56(3): 373. CrossRef - Influence of maternal high-fat diet on offspring's locomotor activity during anxiety-related behavioral tests: A systematic review
Carolina Cadete Lucena Cavalcanti, Raul Manhães-de-Castro, Wenicios Ferreira Chaves, Erika Vanesa Cadena-Burbano, José Antonio-Santos, Raquel da Silva Aragão
Behavioural Brain Research.2024; 462: 114869. CrossRef - Using graphic psychology and spatial design to intervene in balance coordination in patients with balance disorders
Weicong Li, Yueling Liu, Huabin Lv, Yue Ma, Wenwen Shi, Yun Qian
Journal of Asian Architecture and Building Engineering.2024; : 1. CrossRef - Biochemical, biomechanical and imaging biomarkers of ischemic stroke: Time for integrative thinking
Meryem Şahin Erdoğan, Esra Sümer Arpak, Cemre Su Kaya Keles, Federico Villagra, Esin Öztürk Işık, Nazire Afşar, Can A. Yucesoy, Luis A. J. Mur, Otar Akanyeti, Hale Saybaşılı
European Journal of Neuroscience.2024; 59(7): 1789. CrossRef - Developing a novel dual-injection FDG-PET imaging methodology to study the functional neuroanatomy of gait
Hilmar P. Sigurdsson, Lisa Alcock, Michael Firbank, Ross Wilson, Philip Brown, Ross Maxwell, Elizabeth Bennett, Nicola Pavese, David J. Brooks, Lynn Rochester
NeuroImage.2024; 288: 120531. CrossRef - Moderate-to-Vigorous Physical Activity and Response Inhibition Predict Balance in Adults with Attention Deficit/Hyperactivity Disorder
Mansour M. Alotaibi, Robert W. Motl, Despina Stavrinos, Scott W. Snyder, Harshvardhan Singh, Donald H. Lein
Journal of Clinical Medicine.2024; 13(4): 968. CrossRef - Assessing Cognitive Workload in Motor Decision-Making through Functional Connectivity Analysis: Towards Early Detection and Monitoring of Neurodegenerative Diseases
Leonardo Ariel Cano, Ana Lía Albarracín, Alvaro Gabriel Pizá, Cecilia Elisabet García-Cena, Eduardo Fernández-Jover, Fernando Daniel Farfán
Sensors.2024; 24(4): 1089. CrossRef - Mesencephalic Locomotor Region and Presynaptic Inhibition during Anticipatory Postural Adjustments in People with Parkinson’s Disease
Carla Silva-Batista, Jumes Lira, Daniel Boari Coelho, Andrea Cristina de Lima-Pardini, Mariana Penteado Nucci, Eugenia Casella Tavares Mattos, Fernando Henrique Magalhaes, Egberto Reis Barbosa, Luis Augusto Teixeira, Edson Amaro Junior, Carlos Ugrinowitsc
Brain Sciences.2024; 14(2): 178. CrossRef - The Role of Cognition in Balance Control
Nazrien M.D Nazrien, Novitri Novitri, Tertiano Prabowo, Farida Arisanti
OBM Neurobiology.2024; 08(01): 1. CrossRef - Development of a hardware CPG model for controlling both legs of a musculoskeletal humanoid robot with gait and gait cycle change by higher center and sensory information
Tatsumi Goto, Rina Okamoto, Takumi Ishihama, Kentaro Yamazaki, Yugo Kokubun, Minami Kaneko, Fumio Uchikoba
Artificial Life and Robotics.2024;[Epub] CrossRef - Vascular smooth muscle cell-specific Igf1r deficiency exacerbates the development of hypertension-induced cerebral microhemorrhages and gait defects
Lauren R. Miller, Marisa A. Bickel, Michaela L. Vance, Hannah Vaden, Domonkos Nagykaldi, Adam Nyul-Toth, Elizabeth C. Bullen, Tripti Gautam, Stefano Tarantini, Andriy Yabluchanskiy, Tamas Kiss, Zoltan Ungvari, Shannon M. Conley
GeroScience.2024; 46(3): 3481. CrossRef - Aging alters functional connectivity of motor theta networks during sensorimotor reactions
Juliana Yordanova, Michael Falkenstein, Vasil Kolev
Clinical Neurophysiology.2024; 159: 97. CrossRef - Gait impairment-related axonal degeneration in Parkinson’s disease by neurite orientation dispersion and density imaging
Xuan Wei, Shiya Wang, Mingkai Zhang, Ying Yan, Zheng Wang, Wei Wei, Houzhen Tuo, Zhenchang Wang
npj Parkinson's Disease.2024;[Epub] CrossRef - Effects of transcranial direct current stimulation alone and in combination with rehabilitation therapies on gait and balance among individuals with Parkinson’s disease: a systematic review and meta-analysis
Thi Xuan Dieu Nguyen, Phuc Thi Mai, Ya-Ju Chang, Tsung-Hsun Hsieh
Journal of NeuroEngineering and Rehabilitation.2024;[Epub] CrossRef - Early detection of diseases causing dementia using digital navigation and gait measures: A systematic review of evidence
Giedrė Čepukaitytė, Coco Newton, Dennis Chan
Alzheimer's & Dementia.2024;[Epub] CrossRef - Working memory load modulates anticipatory postural adjustments during step initiation
Kodai Minami, Eiji Yamanaka, Kohei Okuyama, Michiyuki Kawakami, Kunitsugu Kondo, Riku Ishizaka, Aoki Takahashi, Tatsunori Watanabe
Experimental Brain Research.2024;[Epub] CrossRef - Spatial-temporal parameters during unobstructed walking in people with Parkinson's disease and healthy older people: a public data set
Tiago Penedo, Carlos Augusto Kalva-Filho, Jônatas Augusto Cursiol, Murilo Henrique Faria, Daniel Boari Coelho, Fabio Augusto Barbieri
Frontiers in Aging Neuroscience.2024;[Epub] CrossRef - Influence of oral motor tasks on postural muscle activity during dynamic reactive balance
Daniel Hellmann, Cagla Fadillioglu, Lisa Kanus, Felix Möhler, Hans J. Schindler, Marc Schmitter, Thorsten Stein, Steffen Ringhof
Journal of Oral Rehabilitation.2024;[Epub] CrossRef - Gait-related beta-gamma phase amplitude coupling in the subthalamic nucleus of parkinsonian patients
AmirAli Farokhniaee, Chiara Palmisano, Jasmin Del Vecchio Del Vecchio, Gianni Pezzoli, Jens Volkmann, Ioannis U. Isaias
Scientific Reports.2024;[Epub] CrossRef - Predicting slight freezing of gait in Parkinson's disease with anticipatory postural adjustments and limits of stability
Weijia Hou, Fan Wu, Yue Wang, Weihua Li, Yuanyuan Cheng, Zhizhong Zhu, Siquan Liang, Peipei Liu, Yang Yu, Jialing Wu
Parkinsonism & Related Disorders.2024; 123: 106949. CrossRef - Sound and postural control during stance tasks in abnormal subjective haptic vertical
Kristina Anton, Arne Ernst, Dietmar Basta
Journal of Vestibular Research.2024; : 1. CrossRef - Assessing Postural Control From Birth to Adulthood Among Individuals Born Preterm: A Systematic Review
Soraia Pereira, Augusta Silva, Rubim Santos, Cláudia Costa Silva
Motor Control.2024; : 1. CrossRef - Young adults who improve performance during dual-task walking show more flexible reallocation of cognitive resources: a mobile brain-body imaging (MoBI) study
Eleni Patelaki, John J Foxe, Kevin A Mazurek, Edward G Freedman
Cerebral Cortex.2023; 33(6): 2573. CrossRef - Midfrontal theta dynamics index the monitoring of postural stability
Mitchel Stokkermans, Teodoro Solis-Escalante, Michael X Cohen, Vivian Weerdesteyn
Cerebral Cortex.2023; 33(7): 3454. CrossRef - The Instrumented Stand and Walk (ISAW) test to predict falls in older men
Fay B. Horak, Amy Laird, Patricia Carlson-Kuhta, Melanie Abrahamson, Martina Mancini, Eric S. Orwoll, Jodi A. Lapidus, Vrutangkumar V. Shah
GeroScience.2023; 45(2): 823. CrossRef - Turning and multitask gait unmask gait disturbance in mild‐to‐moderate multiple sclerosis: Underlying specific cortical thinning and connecting fibers damage
Qingmeng Chen, Takaaki Hattori, Hiroshi Tomisato, Masahiro Ohara, Kosei Hirata, Takanori Yokota
Human Brain Mapping.2023; 44(3): 1193. CrossRef - Neurophysiological mechanisms of gait disturbance in advanced Parkinson's disease patients
Kaoru Takakusaki, Mirai Takahashi, Tomohiro Noguchi, Ryosuke Chiba
Neurology and Clinical Neuroscience.2023; 11(4): 201. CrossRef - Altered brain function in patients with vestibular migraine: a study on resting state functional connectivity
Zhengwei Chen, Liangqun Rong, Lijie Xiao, Quan Wang, Yueji Liu, Cunxin Lin, Jianing Wang, Haiyan Liu, Xiu-e Wei
Neuroradiology.2023; 65(3): 579. CrossRef - Assessment of balance in people with mild traumatic brain injury using a balance systems model approach
Kody R. Campbell, Kathleen T. Scanlan, Jennifer L. Wilhelm, Barbara H. Brumbach, Natalie C. Pettigrew, Akira Neilson, Lucy Parrington, Laurie A. King
Gait & Posture.2023; 100: 107. CrossRef - Relationship between lower limb muscle activity and cortical activation among elderly people during walking: Effects of fast speed and cognitive dual task
Jinuk Kim, Jungsoo Lee, Gihyoun Lee, Won Hyuk Chang, Myoung-Hwan Ko, Woo-Kyoung Yoo, Gyu-Ha Ryu, Yun-Hee Kim
Frontiers in Aging Neuroscience.2023;[Epub] CrossRef - Mental states and personality based on real-time physical activity and facial expression recognition
Yating Huang, Dengyue Zhai, Jingze Song, Xuanheng Rao, Xiao Sun, Jin Tang
Frontiers in Psychiatry.2023;[Epub] CrossRef - Sensorimotor recalibration of postural control strategies occurs after whole body vibration
Isotta Rigoni, Giulio Degano, Mahmoud Hassan, Antonio Fratini
Scientific Reports.2023;[Epub] CrossRef - A neuroergonomic approach to assessing motor performance in stroke patients using fNIRS and behavioral data
Lingguo Bu, Jing Qu, Lei Zhao, Yanjie Zhang, Yonghui Wang
Applied Ergonomics.2023; 109: 103979. CrossRef - Case report: A novel approach of closed-loop brain stimulation combined with robot gait training in post-stroke gait disturbance
Atsushi Shima, Tomoaki Miyake, Kazuki Tanaka, Akari Ogawa, Erika Omae, Yui Nagamori, Yusuke Miyata, Koji Ohata, Takakuni Maki, Yumie Ono, Tatsuya Mima, Ryosuke Takahashi, Satoko Koganemaru
Frontiers in Human Neuroscience.2023;[Epub] CrossRef - Cognitive and emotional factors influence specific domains of postural control in individuals with moderate-to-severe Parkinson’s disease
Claudia Eunice Neves de Oliveira, Daniel Boari Coelho, Caroline Ribeiro de Souza, Alana Xavier Batista, Raquel Marquesini, Acácio Moreira-Neto, Carla Silva-Batista, Adriana Tanigawa Bitiati, Luis Augusto Teixeira, Andrea Cristina de Lima-Pardini
Gait & Posture.2023; 101: 66. CrossRef - Developmental changes in straight gait in childhood
Saori Miyagishima, Hiroki Mani, Yui Sato, Takahiro Inoue, Tadayoshi Asaka, Naoki Kozuka, Dimitrios Sokratis Komaris
PLOS ONE.2023; 18(2): e0281037. CrossRef - Case report: Backward gait training combined with gait-synchronized cerebellar transcranial alternating current stimulation in progressive supranuclear palsy
Atsushi Shima, Kazuki Tanaka, Akari Ogawa, Erika Omae, Tomoaki Miyake, Yui Nagamori, Yusuke Miyata, Koji Ohata, Yumie Ono, Tatsuya Mima, Ryosuke Takahashi, Satoko Koganemaru
Frontiers in Human Neuroscience.2023;[Epub] CrossRef - Cerebellar Transcranial Direct Current Stimulation Modulates Anticipatory Postural Adjustments in Healthy Adults
Haian Mao, Wenwu Xiao, Zengming Hao, Shengjun Wen, Huaichun Yang, Fahad Sultan, Chuhuai Wang
The Cerebellum.2023; 23(2): 383. CrossRef - Model of Gait Control in Parkinson’s Disease and Prediction of Robotic Assistance
Clémence Vandamme, Virginie Otlet, Renaud Ronsse, Frédéric Crevecoeur
IEEE Transactions on Neural Systems and Rehabilitation Engineering.2023; 31: 1374. CrossRef - Sex-specific differences in resting-state functional brain activity in pediatric concussion
Bhanu Sharma, Cameron Nowikow, Carol DeMatteo, Michael D. Noseworthy, Brian W. Timmons
Scientific Reports.2023;[Epub] CrossRef - Pain and Parkinson's disease: Current mechanism and management updates
Frederic J. F. Viseux, Arnaud Delval, Martin Simoneau, Luc Defebvre
European Journal of Pain.2023; 27(5): 553. CrossRef - Increased Effective Connectivity of the Left Parietal Lobe During Walking Tasks in Parkinson’s Disease
Yue Wang, Ningbo Yu, Jiewei Lu, Xinyuan Zhang, Jin Wang, Zhilin Shu, Yuanyuan Cheng, Zhizhong Zhu, Yang Yu, Peipei Liu, Jianda Han, Jialing Wu
Journal of Parkinson's Disease.2023; 13(2): 165. CrossRef - Cortical oscillations and interareal synchronization as a preparatory activity for postural response
Kimiya Fujio, Hiroki Obata, Kenta Takeda, Noritaka Kawashima
European Journal of Neuroscience.2023; 57(9): 1516. CrossRef - Effects of wearable visual cueing on gait pattern and stability in patients with Parkinson’s disease
Wei Zhang, Yun Han, Yuanyuan Shi, Shilei Yan, Wenjing Song, Guiyun Cui, Jie Xiang
Frontiers in Neurology.2023;[Epub] CrossRef - Modulating arousal to overcome gait impairments in Parkinson’s disease: how the noradrenergic system may act as a double-edged sword
Anouk Tosserams, Bastiaan R. Bloem, Kaylena A. Ehgoetz Martens, Rick C. Helmich, Roy P. C. Kessels, James M. Shine, Natasha L. Taylor, Gabriel Wainstein, Simon J. G. Lewis, Jorik Nonnekes
Translational Neurodegeneration.2023;[Epub] CrossRef - Contrôle central de l’équilibre et prévention des chutes : impact sur la rééducation
A.P. Yelnik
Bulletin de l'Académie Nationale de Médecine.2023; 207(5): 649. CrossRef - Variability of the lower limb symmetry index associated with the gait parameters in the overweight adult population with flatfoot: a case-control study
Israel Casado-Hernández, Ricardo Becerro-de-Bengoa-Vallejo, Marta Losa-Iglesias, Juan Gómez-Salgado, Daniel López-López, Javier Bayod
Frontiers in Bioengineering and Biotechnology.2023;[Epub] CrossRef - Influence of wearing complete dentures on control of head position during walking in edentulous older adults
Kento Shimizu, Hiroyuki Suzuki, Naoya Isoyama, Yuiko Yanagihara, Shunsuke Minakuchi
Journal of Oral Rehabilitation.2023; 50(10): 1012. CrossRef - Cortical thickness is related to cognitive-motor automaticity and attention allocation in individuals with Alzheimer’s disease: a regions of interest study
Jason K. Longhurst, Karthik R. Sreenivasan, Jemma Kim, Jeffrey L. Cummings, Samantha E. John, Brach Poston, Dietmar Cordes, John V. Rider, Merrill R. Landers
Experimental Brain Research.2023; 241(6): 1489. CrossRef - Structural-Functional Correlates of Response to Pedunculopontine Stimulation in a Randomized Clinical Trial for Axial Symptoms of Parkinson’s Disease
Sorin Breit, Luka Milosevic, Georgios Naros, Idil Cebi, Daniel Weiss, Alireza Gharabaghi
Journal of Parkinson's Disease.2023; 13(4): 563. CrossRef - Interrelation between functional decline and dementia: The potential role of balance assessment
Francesca Biasin, Chiara Ceolin, Silvia Celli, Camilla Terziotti, Cecilia Raffaelli, Cristiano Bontempi, Maria Devita, Marina De Rui, Giuseppe Sergi, Alessandra Coin
Human Movement Science.2023; 89: 103095. CrossRef - Trend Change Analysis as a New Tool to Complement the Evaluation of Human Body Balance in the Time and Frequency Domains
Piotr Wodarski
Journal of Human Kinetics.2023;[Epub] CrossRef - Neural underpinnings of freezing-related dynamic balance control in people with Parkinson's disease
Bauke W. Dijkstra, Moran Gilat, Nicholas D'Cruz, Demi Zoetewei, Alice Nieuwboer
Parkinsonism & Related Disorders.2023; 112: 105444. CrossRef - Electrocortical Dynamics of Usual Walking and the Planning to Step over Obstacles in Parkinson’s Disease
Rodrigo Vitório, Ellen Lirani-Silva, Diego Orcioli-Silva, Victor Spiandor Beretta, Anderson Souza Oliveira, Lilian Teresa Bucken Gobbi
Sensors.2023; 23(10): 4866. CrossRef - Mental fatigue does not affect static balance under both single and dual task conditions in young adults
Abubakar Tijjani Salihu, Jibrin Sammani Usman, Keith D. Hill, Maryam Zoghi, Shapour Jaberzadeh
Experimental Brain Research.2023; 241(7): 1769. CrossRef - Contribution of neural circuits tested by transcranial magnetic stimulation in corticomotor control of low back muscle: a systematic review
Mikaël Desmons, Michael Theberge, Catherine Mercier, Hugo Massé-Alarie
Frontiers in Neuroscience.2023;[Epub] CrossRef - Predicting the fatigue in Parkinson's disease using inertial sensor gait data and clinical characteristics
Hui Wang, Binbin Hu, Juan Huang, Lin Chen, Min Yuan, Xingfu Tian, Ting Shi, Jiahao Zhao, Wei Huang
Frontiers in Neurology.2023;[Epub] CrossRef - Basal Ganglia and Brainstem Located Cerebral Microbleeds Contributed to Gait Impairment in Patients with Cerebral Small Vessel Disease
He-Jiao Mao, Jiang-Xia Zhang, Wen-Cheng Zhu, Hao Zhang, Xiang-Min Fan, Fei Han, Jun Ni, Li-Xin Zhou, Ming Yao, Feng Tian, Ning Su, Yi-Cheng Zhu
Journal of Alzheimer's Disease.2023; 94(3): 1005. CrossRef - Facilitating or disturbing? An investigation about the effects of auditory frequencies on prefrontal cortex activation and postural sway
Valeria Belluscio, Giulia Cartocci, Tommaso Terbojevich, Paolo Di Feo, Bianca Maria Serena Inguscio, Marco Ferrari, Valentina Quaresima, Giuseppe Vannozzi
Frontiers in Neuroscience.2023;[Epub] CrossRef - Functional connectome predicting individual gait function and its relationship with molecular architecture in Parkinson's disease
Haoting Wu, Chenqing Wu, Jianmei Qin, Cheng Zhou, Sijia Tan, Xiaojie DuanMu, Xiaojun Guan, Xueqin Bai, Tao Guo, Jingjing Wu, Jingwen Chen, Jiaqi Wen, Zhengye Cao, Ting Gao, Luyan Gu, Peiyu Huang, Baorong Zhang, Xiaojun Xu, Minming Zhang
Neurobiology of Disease.2023; 184: 106216. CrossRef - Behavioral decomposition reveals rich encoding structure employed across neocortex in rats
Bartul Mimica, Tuçe Tombaz, Claudia Battistin, Jingyi Guo Fuglstad, Benjamin A. Dunn, Jonathan R. Whitlock
Nature Communications.2023;[Epub] CrossRef - Efficacy of Dual‐Task Training in Patients with Parkinson's Disease: A Systematic Review with Meta‐Analysis
Héctor García‐López, María de los Ángeles Castillo‐Pintor, Adelaida María Castro‐Sánchez, Inmaculada Carmen Lara‐Palomo, Esteban Obrero‐Gaitán, Irene Cortés‐Pérez
Movement Disorders Clinical Practice.2023; 10(9): 1268. CrossRef - Artificial Intelligence Distinguishes Pathological Gait: The Analysis of Markerless Motion Capture Gait Data Acquired by an iOS Application (TDPT-GT)
Chifumi Iseki, Tatsuya Hayasaka, Hyota Yanagawa, Yuta Komoriya, Toshiyuki Kondo, Masayuki Hoshi, Tadanori Fukami, Yoshiyuki Kobayashi, Shigeo Ueda, Kaneyuki Kawamae, Masatsune Ishikawa, Shigeki Yamada, Yukihiko Aoyagi, Yasuyuki Ohta
Sensors.2023; 23(13): 6217. CrossRef - Transcranial direct current stimulation suggests not improving postural control during adapted tandem position in people with Parkinson's disease: A pilot study
Beatriz Regina Legutke, Lilian Teresa Bucken Gobbi, Diego Orcioli-Silva, Paulo Cezar Rocha dos Santos, Gabriel Antonio Gazziero Moraca, Rodrigo Vitório, Victor Spiandor Beretta
Behavioural Brain Research.2023; 452: 114581. CrossRef - Postural stability and plantar pressure parameters in healthy subjects: variability, correlation analysis and differences under open and closed eye conditions
P. De Blasiis, P. Caravaggi, A. Fullin, A. Leardini, A. Lucariello, A. Perna, G. Guerra, A. De Luca
Frontiers in Bioengineering and Biotechnology.2023;[Epub] CrossRef - The temporal neurovascular coupling response remains intact during sinusoidal hypotensive and hypertensive challenges
Joel S Burma, Selina Rattana, Ibukunoluwa K Oni, Andrew P Lapointe, Jeff F Dunn, Jonathan D Smirl
Physiological Measurement.2023; 44(7): 074002. CrossRef - The generation mechanisms of gait impairment in Parkinson’s disease and corresponding animal models
Yue Wu, Yezhong Tang, Guangzhan Fang
Chinese Science Bulletin.2023; 68(23): 3043. CrossRef - Human upper extremity motor cortex activity shows distinct oscillatory signatures for stereotyped arm and leg movements
Clara Kwon Starkweather, Melanie A. Morrison, Maria Yaroshinsky, Kenneth Louie, Jannine Balakid, Kara Presbrey, Philip A. Starr, Doris D. Wang
Frontiers in Human Neuroscience.2023;[Epub] CrossRef - Discrete cortical control during quiet stance revealed by desynchronization and rebound of beta oscillations
Akihiro Nakamura, Ryota Miura, Yasuyuki Suzuki, Pietro Morasso, Taishin Nomura
Neuroscience Letters.2023; 814: 137443. CrossRef - Soy sauce and vanilla odors improve balance ability in children
Junichi Inatomi, Kiwa Yasuoka, Hideaki Takebayashi, Yutaka Yano, Satoko Kataoka, Masahiro Yamaguchi
Journal of Sensory Studies.2023;[Epub] CrossRef - Brain activities for Open Intelligence : Application of Brain Science for Behavior Adaptation
Shingo Shimoda
Journal of the Robotics Society of Japan.2023; 41(7): 591. CrossRef - In Silico Neuroprotective Effects of Specific Rheum palmatum Metabolites on Parkinson’s Disease Targets
Patrick Jay B. Garcia, Steven Kuan-Hua Huang, Kathlia A. De Castro-Cruz, Rhoda B. Leron, Po-Wei Tsai
International Journal of Molecular Sciences.2023; 24(18): 13929. CrossRef - Effect of COVID-19 on Maintaining Balance in Highly Skilled Handball Players
Emilio Fernández-Rodríguez, Tomasz Niźnikowski, Oscar Romero Ramos, Logan Markwell
Polish Journal of Sport and Tourism.2023; 30(3): 18. CrossRef - Injury of the Vestibulocerebellar Tract and Signs of Ataxia in Patients with Cerebellar Stroke
Sang-Seok Yeo, Seung-Min Nam, In-Hee Cho
Journal of Clinical Medicine.2023; 12(21): 6877. CrossRef - Analysis of abnormal posture in patients with Parkinson's disease using a computational model considering muscle tones
Yuichiro Omura, Hiroki Togo, Kohei Kaminishi, Tetsuya Hasegawa, Ryosuke Chiba, Arito Yozu, Kaoru Takakusaki, Mitsunari Abe, Yuji Takahashi, Takashi Hanakawa, Jun Ota
Frontiers in Computational Neuroscience.2023;[Epub] CrossRef - Functional near-infrared spectroscopy in the assessment of rehabilitation efficacy of virtual reality products for people with cognitive disorders
Jing Qu, Yanjie Zhang, Lingguo Bu
International Journal of Industrial Ergonomics.2023; 97: 103500. CrossRef - Effect of dual-task interaction combining postural and visual perturbations on cortical activity and postural control ability
Ryoki Nishimoto, Sayaka Fujiwara, Yumiko Kutoku, Toru Ogata, Masahito Mihara
NeuroImage.2023; 280: 120352. CrossRef - Tango-therapy vs physical exercise in older people with dementia; a randomized controlled trial
Lucía Bracco, Arrate Pinto-Carral, Linda Hillaert, France Mourey
BMC Geriatrics.2023;[Epub] CrossRef - Circuits and Biomarkers of the Central Nervous System Relating to Astronaut Performance: Summary Report for a NASA-Sponsored Technical Interchange Meeting
Joshua S. Alwood, Ajitkumar P. Mulavara, Janani Iyer, Siddhita D. Mhatre, Susanna Rosi, Mark Shelhamer, Catherine Davis, Christopher W. Jones, Xiao Wen Mao, Rajeev I. Desai, Alexandra M. Whitmire, Thomas J. Williams
Life.2023; 13(9): 1852. CrossRef - Objective Physical Function in the Alzheimer’s Disease Continuum: Association with Cerebrospinal Fluid Biomarkers in the ALBION Study
Stefanos N. Sampatakakis, Eirini Mamalaki, Eva Ntanasi, Faidra Kalligerou, Ioannis Liampas, Mary Yannakoulia, Antonios N. Gargalionis, Nikolaos Scarmeas
International Journal of Molecular Sciences.2023; 24(18): 14079. CrossRef - Differential spatiotemporal gait effects with frequency and dopaminergic modulation in STN-DBS
Ritesh A. Ramdhani, Jeremy Watts, Myriam Kline, Toni Fitzpatrick, Martin Niethammer, Anahita Khojandi
Frontiers in Aging Neuroscience.2023;[Epub] CrossRef - Physical activity and sport practice to improve balance control of visually impaired individuals: a narrative review with future perspectives
Giuditta Carretti, Mirko Manetti, Mirca Marini
Frontiers in Sports and Active Living.2023;[Epub] CrossRef - Regional activity and effective connectivity within the frontoparietal network during precision walking with visual cueing: an fNIRS study
Duc Trung Le, Masato Tsuyuhara, Hiroki Kuwamura, Kento Kitano, Thu Dang Nguyen, Thuan Duc Nguyen, Naoto Fujita, Tatsunori Watanabe, Hisao Nishijo, Masahito Mihara, Susumu Urakawa
Cerebral Cortex.2023; 33(22): 11157. CrossRef - Effects of dual-task training on chronic ankle instability: a systematic review and meta-analysis
Lu Wang, Ge Yu, Yaping Chen
BMC Musculoskeletal Disorders.2023;[Epub] CrossRef - Fluctuations in Upper and Lower Body Movement during Walking in Normal Pressure Hydrocephalus and Parkinson’s Disease Assessed by Motion Capture with a Smartphone Application, TDPT-GT
Chifumi Iseki, Shou Suzuki, Tadanori Fukami, Shigeki Yamada, Tatsuya Hayasaka, Toshiyuki Kondo, Masayuki Hoshi, Shigeo Ueda, Yoshiyuki Kobayashi, Masatsune Ishikawa, Shigenori Kanno, Kyoko Suzuki, Yukihiko Aoyagi, Yasuyuki Ohta
Sensors.2023; 23(22): 9263. CrossRef - Does Impaired Plantar Cutaneous Vibration Perception Contribute to Axial Motor Symptoms in Parkinson’s Disease? Effects of Medication and Subthalamic Nucleus Deep Brain Stimulation
Tobias Heß, Peter Themann, Christian Oehlwein, Thomas L. Milani
Brain Sciences.2023; 13(12): 1681. CrossRef - Cortico-muscular connectivity is modulated by passive and active Lokomat-assisted Gait
Fiorenzo Artoni, Andrea Cometa, Stefania Dalise, Valentina Azzollini, Silvestro Micera, Carmelo Chisari
Scientific Reports.2023;[Epub] CrossRef - Neuroplasticity as the basis of motor rehabilitation
A. E. Amamchyan, G. Sh. Gafiyatullina
Medical Herald of the South of Russia.2023; 14(4): 122. CrossRef - Hyperglycemia affects axial signs in patients with Parkinson’s disease through mechanisms of insulin resistance or non-insulin resistance
Ruidan Wang, Zhaohui Jin, Qiaoxia Zhen, Lin Qi, Cui Liu, Ping Wang, Yonghong Liu, Jinping Fang, Yanjun Liu, Yuan Su, Yixuan Wang, Detao Meng, Hongjiao Yan, Yi Zhen, Zhenzhen Li, Boyan Fang
Neurological Sciences.2023;[Epub] CrossRef - Changes of neural coupling between cognitive and motor networks associated with dual-task performance in Parkinson’s disease
Eunkyung Kim, Seo Jung Yun, Byung-Mo Oh, Han Gil Seo
Neurological Sciences.2023;[Epub] CrossRef - Effect of hardware balance trainings on postural control, risk of falls and cognitive function on elderly people with chronic cerebral ischemia. (Randomized controlled trial)
L.D. Litvina, S.M. Konev, E.S. Koneva, D.U. Butko, K.V. Lyadov, E.N. Zhumanova, L.A. Gridin, N.B. Korchazhkina, K.V. Kotenko
Voprosy kurortologii, fizioterapii i lechebnoi fizicheskoi kul'tury.2023; 100(6): 31. CrossRef - Separated center-of-pressure measurements reveal new characteristics of reduced anticipatory postural adjustments during gait initiation in patients with Parkinson’s disease
Ryo Onuma, Tadashi Masuda, Fumihiko Hoshi, Tadamitsu Matsuda, Tomoko Sakai, Atsushi Okawa, Tetsuya Jinno
Physiotherapy Theory and Practice.2022; 38(13): 2544. CrossRef - Kyphosis and bizarre patterns impair spontaneous gait performance in end-of-life mice with Alzheimer's disease pathology while gait is preserved in normal aging
Lidia Castillo-Mariqueo, Lydia Giménez-Llort
Neuroscience Letters.2022; 767: 136280. CrossRef - Brain network topology and future development of freezing of gait in Parkinson’s disease: a longitudinal study
Nannan Li, Du Lei, Jiaxin Peng, Xueling Suo, Junying Li, Liren Duan, Chaolan Chen, Qiyong Gong, Rong Peng
Journal of Neurology.2022; 269(5): 2503. CrossRef - Changes of cerebral network activity after invasive stimulation of the mesencephalic locomotor region in a rat stroke model
Stefanie D. Krämer, Michael K. Schuhmann, Fabian Schadt, Ina Israel, Samuel Samnick, Jens Volkmann, Felix Fluri
Experimental Neurology.2022; 347: 113884. CrossRef - Neuroticism and Extraversion Are Related to Changes in Postural Stability During Anatomically-Related Cognitive Tasks
Christopher I. Higginson, Marisa Valenti, Karlie Ibrahim, Brian A. Knarr, Rob Ryan, Jill S. Higginson
Journal of Motor Behavior.2022; 54(4): 401. CrossRef - Wide-Field Calcium Imaging of Dynamic Cortical Networks during Locomotion
Sarah L West, Justin D Aronson, Laurentiu S Popa, Kathryn D Feller, Russell E Carter, William M Chiesl, Morgan L Gerhart, Aditya C Shekhar, Leila Ghanbari, Suhasa B Kodandaramaiah, Timothy J Ebner
Cerebral Cortex.2022; 32(12): 2668. CrossRef - Longitudinal Relation Between Structural Network Efficiency, Cognition, and Gait in Cerebral Small Vessel Disease
Mengfei Cai, Mina A Jacob, David G Norris, Frank-Erik de Leeuw, Anil M Tuladhar, Lewis A Lipsitz
The Journals of Gerontology: Series A.2022; 77(3): 554. CrossRef - Presence Hallucinations during Locomotion in Patients with Parkinson's Disease
Jevita Potheegadoo, Herberto Dhanis, Judit Horvath, Pierre R. Burkhard, Olaf Blanke
Movement Disorders Clinical Practice.2022; 9(1): 127. CrossRef - Whole-body vibration exercise and training increase regional CBF in mild cognitive impairment with enhanced cognitive function
Ikuo Odano, Fumio Maeyatsu, Mami Asari, Sayaka Yamaguchi, Tsukasa Miura, Yasuyuki Taki
Annals of Nuclear Medicine.2022; 36(1): 82. CrossRef - Visuomotor errors drive step length and step time adaptation during ‘virtual’ split-belt walking: the effects of reinforcement feedback
Sumire Sato, Ashley Cui, Julia T. Choi
Experimental Brain Research.2022; 240(2): 511. CrossRef - Influence of controlled masticatory muscle activity on dynamic reactive balance
Cagla Fadillioglu, Lisa Kanus, Felix Möhler, Steffen Ringhof, Hans J. Schindler, Thorsten Stein, Daniel Hellmann
Journal of Oral Rehabilitation.2022; 49(3): 327. CrossRef - Discussion of Research Priorities for Gait Disorders in Parkinson's Disease
Nicolaas I. Bohnen, Rui M. Costa, William T. Dauer, Stewart A. Factor, Nir Giladi, Mark Hallett, Simon J.G. Lewis, Alice Nieuwboer, John G. Nutt, Kaoru Takakusaki, Un Jung Kang, Serge Przedborski, Stella M. Papa
Movement Disorders.2022; 37(2): 253. CrossRef - Analysis of the Stability of the Body in a Standing Position When Shooting at a Stationary Target―A Randomized Controlled Trial
Marlena Krawczyk-Suszek, Blanka Martowska, Rafał Sapuła
Sensors.2022; 22(1): 368. CrossRef - Gaps and roadmap of novel neuromodulation targets for treatment of gait in Parkinson’s disease
Rubens Gisbert Cury, Nicola Pavese, Tipu Z. Aziz, Joachim K. Krauss, Elena Moro
npj Parkinson's Disease.2022;[Epub] CrossRef - The Influence of Aging on the Functional Connectivity of the Human Basal Ganglia
Clara Rodriguez-Sabate, Ingrid Morales, Manuel Rodriguez
Frontiers in Aging Neuroscience.2022;[Epub] CrossRef - Effects of task prioritization on a postural-motor task in early-stage Parkinson’s disease: EEG connectivity and clinical implication
Cheng-Ya Huang, Liang-Chi Chen, Ruey-Meei Wu, Ing-Shiou Hwang
GeroScience.2022; 44(4): 2061. CrossRef - Effect of cognitive task complexity on dual task postural stability: a systematic review and meta-analysis
Abubakar Tijjani Salihu, Keith D. Hill, Shapour Jaberzadeh
Experimental Brain Research.2022; 240(3): 703. CrossRef - Targeting Sensory and Motor Integration for Recovery of Movement After CNS Injury
Ahmet S. Asan, James R. McIntosh, Jason B. Carmel
Frontiers in Neuroscience.2022;[Epub] CrossRef - Increased prefrontal activity during usual walking in aging
I. Hoang, L. Paire-Ficout, R. Derollepot, S. Perrey, H. Devos, M. Ranchet
International Journal of Psychophysiology.2022; 174: 9. CrossRef - Effects of sling exercise therapy on post-stroke walking impairment: a systematic review and meta-analysis
Junzi Long, Yasu Zhang, Xiaomin Liu, Xiaoqin Jin
International Journal of Rehabilitation Research.2022; 45(1): 12. CrossRef - Electromyographic responses to unexpected Achilles tendon vibration-induced perturbations during standing in young and older people
Anastasia Papavasileiou, Lida Mademli, Vassilia Hatzitaki, Dimitrios A. Patikas
Experimental Brain Research.2022; 240(4): 1017. CrossRef - Do physical activity, cardiorespiratory fitness, and subcortical brain structures explain reduced walking performance in older adults with multiple sclerosis?
Jessica F. Baird, Gary R. Cutter, Robert W. Motl
Multiple Sclerosis and Related Disorders.2022; 60: 103702. CrossRef - Gait and posture are correlated domains in Parkinson’s disease
Thaisy Moraes Costa, Lucas Simieli, Felipe Marrese Bersotti, Luis Mochizuki, Fabio Augusto Barbieri, Daniel Boari Coelho
Neuroscience Letters.2022; 775: 136537. CrossRef - Why Should Constant Stimulation of Saccular Afferents Modify the Posture and Gait of Patients with Bilateral Vestibular Dysfunction? The Saccular Substitution Hypothesis
Ian S. Curthoys, Paul F. Smith, Angel Ramos de Miguel
Journal of Clinical Medicine.2022; 11(4): 1132. CrossRef - A Neural Controller Model Considering the Vestibulospinal Tract in Human Postural Control
Yuichiro Omura, Kohei Kaminishi, Ryosuke Chiba, Kaoru Takakusaki, Jun Ota
Frontiers in Computational Neuroscience.2022;[Epub] CrossRef - Fear of Falling Does Not Influence Dual-Task Gait Costs in People with Parkinson’s Disease: A Cross-Sectional Study
Tino Prell, Manuela Uhlig, Steffen Derlien, Walter Maetzler, Hannah M. Zipprich
Sensors.2022; 22(5): 2029. CrossRef - Dynamics of cortical and corticomuscular connectivity during planning and execution of visually guided steps in humans
Meaghan E Spedden, Mikkel M Beck, Timothy O West, Simon F Farmer, Jens B Nielsen, Jesper Lundbye-Jensen
Cerebral Cortex.2022; 33(2): 258. CrossRef - Combined Subthalamic and Nigral Stimulation Modulates Temporal Gait Coordination and Cortical Gait-Network Activity in Parkinson’s Disease
Jonas R. Wagner, Miriam Schaper, Wolfgang Hamel, Manfred Westphal, Christian Gerloff, Andreas K. Engel, Christian K. E. Moll, Alessandro Gulberti, Monika Pötter-Nerger
Frontiers in Human Neuroscience.2022;[Epub] CrossRef - Variability of Postural Stability and Plantar Pressure Parameters in Healthy Subjects Evaluated by a Novel Pressure Plate
Allegra Fullin, Paolo Caravaggi, Pietro Picerno, Massimiliano Mosca, Silvio Caravelli, Antonio De Luca, Angela Lucariello, Paolo De Blasiis
International Journal of Environmental Research and Public Health.2022; 19(5): 2913. CrossRef - How Does the Central Nervous System for Posture and Locomotion Cope With Damage-Induced Neural Asymmetry?
Didier Le Ray, Mathias Guayasamin
Frontiers in Systems Neuroscience.2022;[Epub] CrossRef - Rodent models for gait network disorders in Parkinson's disease – a translational perspective
Nikolaus Wenger, Arend Vogt, Matej Skrobot, Elisa L. Garulli, Burce Kabaoglu, Christina Salchow-Hömmen, Thomas Schauer, Daniel Kroneberg, Michael K. Schuhmann, Chi Wang Ip, Christoph Harms, Matthias Endres, Ioannis U. Isaias, Philip Tovote, Robert Blum
Experimental Neurology.2022; 352: 114011. CrossRef - Transcutaneous spinal stimulation alters cortical and subcortical activation patterns during mimicked-standing: A proof-of-concept fMRI study
Gerome A. Manson, Darryn A. Atkinson, Zhaoyue Shi, Jony Sheynin, Christof Karmonik, Rachel L. Markley, Dimitry G. Sayenko
Neuroimage: Reports.2022; 2(2): 100090. CrossRef - Differential Relationships Between Brain Structure and Dual Task Walking in Young and Older Adults
Kathleen E. Hupfeld, Justin M. Geraghty, Heather R. McGregor, C. J. Hass, Ofer Pasternak, Rachael D. Seidler
Frontiers in Aging Neuroscience.2022;[Epub] CrossRef - What Is the Brainstem Control of Locomotion?
Farwa Ali, Eduardo Benarroch
Neurology.2022; 98(11): 446. CrossRef - The emerging postural instability phenotype in idiopathic Parkinson disease
Frank M. Skidmore, William S. Monroe, Christopher P. Hurt, Anthony P. Nicholas, Adam Gerstenecker, Thomas Anthony, Leon Jololian, Gary Cutter, Adil Bashir, Thomas Denny, David Standaert, Elizabeth A. Disbrow
npj Parkinson's Disease.2022;[Epub] CrossRef - Brain Activation During Active Balancing and Its Behavioral Relevance in Younger and Older Adults: A Functional Near-Infrared Spectroscopy (fNIRS) Study
Nico Lehmann, Yves-Alain Kuhn, Martin Keller, Norman Aye, Fabian Herold, Bogdan Draganski, Wolfgang Taube, Marco Taubert
Frontiers in Aging Neuroscience.2022;[Epub] CrossRef - Influence of Controlled Stomatognathic Motor Activity on Sway, Control and Stability of the Center of Mass During Dynamic Steady-State Balance—An Uncontrolled Manifold Analysis
Cagla Fadillioglu, Lisa Kanus, Felix Möhler, Steffen Ringhof, Daniel Hellmann, Thorsten Stein
Frontiers in Human Neuroscience.2022;[Epub] CrossRef - Frequency‐dependent modulation of neural oscillations across the gait cycle
Mingqi Zhao, Gaia Bonassi, Jessica Samogin, Gaia Amaranta Taberna, Elisa Pelosin, Alice Nieuwboer, Laura Avanzino, Dante Mantini
Human Brain Mapping.2022; 43(11): 3404. CrossRef - Gait subgroups among older adults with chronic pain differ in cerebellum and basal ganglia gray matter volumes
Ania L. Lipat, David J. Clark, Chris J. Hass, Yenisel Cruz-Almeida
Experimental Gerontology.2022; 163: 111773. CrossRef - Restorative Action of Vitamin D3 on Motor Dysfunction Through Enhancement of Neurotrophins and Antioxidant Expression in the Striatum
S.K.V. Manjari, Shuvadeep Maity, R. Poornima, Suk-Yu Yau, K. Vaishali, David Stellwagen, Pragya Komal
Neuroscience.2022; 492: 67. CrossRef - Consensus Paper: Ataxic Gait
Pierre Cabaraux, Sunil K. Agrawal, Huaying Cai, Rocco Salvatore Calabro, Carlo Casali, Loic Damm, Sarah Doss, Christophe Habas, Anja K. E. Horn, Winfried Ilg, Elan D. Louis, Hiroshi Mitoma, Vito Monaco, Maria Petracca, Alberto Ranavolo, Ashwini K. Rao, Se
The Cerebellum.2022; 22(3): 394. CrossRef - Body Representation in Patients with Severe Spinal Cord Injury: A Pilot Study on the Promising Role of Powered Exoskeleton for Gait Training
Maria Grazia Maggio, Antonino Naro, Rosaria De Luca, Desiree Latella, Tina Balletta, Lory Caccamo, Giovanni Pioggia, Daniele Bruschetta, Rocco Salvatore Calabrò
Journal of Personalized Medicine.2022; 12(4): 619. CrossRef - Parkinson’s Disease Symptoms Associated with Developing On-State Axial Symptoms Early after Subthalamic Deep Brain Stimulation
Gustavo Fernández-Pajarín, Ángel Sesar, José Luis Relova, Begoña Ares, Isabel Jiménez, Miguel Gelabert-González, Eduardo Arán, Alfonso Castro
Diagnostics.2022; 12(4): 1001. CrossRef - Glycation modulates glutamatergic signaling and exacerbates Parkinson’s disease-like phenotypes
Ana Chegão, Mariana Guarda, Bruno M. Alexandre, Liana Shvachiy, Mariana Temido-Ferreira, Inês Marques-Morgado, Bárbara Fernandes Gomes, Rune Matthiesen, Luísa V. Lopes, Pedro R. Florindo, Ricardo A. Gomes, Patrícia Gomes-Alves, Joana E. Coelho, Tiago Flem
npj Parkinson's Disease.2022;[Epub] CrossRef - The Association Between Gait Speed and Sleep Problems Among Chinese Adults Aged 50 and Greater
Lili Wang, Benxu Zou
Frontiers in Neuroscience.2022;[Epub] CrossRef - Effect of optokinetic virtual reality scenes on a sitting-to-stand movement
Akkradate Siriphorn, Chachane Jarudej, Nuttapong Ochaklin, Sarunyou Nuttawanlop, Theerapan Prasertteerapong
Human Movement Science.2022; 83: 102956. CrossRef - Neurophysiological mechanisms of gait disturbance in advanced Parkinson's disease patients
Kaoru Takakusaki, Mirai Takahashi, Tomohiro Noguchi, Ryosuke Chiba
Neurology and Clinical Neuroscience.2022;[Epub] CrossRef - Is Balance Control Affected by Sleep Deprivation? A Systematic Review of the Impact of Sleep on the Control of Balance
Guilherme Silva Umemura, Fabianne Furtado, Fabia Camile dos Santos, Bruno da Silva Brandão Gonçalves, Arturo Forner-Cordero
Frontiers in Neuroscience.2022;[Epub] CrossRef - Troubleshooting Gait Disturbances in Parkinson’s Disease With Deep Brain Stimulation
Nicoló G. Pozzi, Chiara Palmisano, Martin M. Reich, Philip Capetian, Claudio Pacchetti, Jens Volkmann, Ioannis U. Isaias
Frontiers in Human Neuroscience.2022;[Epub] CrossRef - The functional and structural neural correlates of dynamic balance impairment and recovery in persons with acquired brain injury
Katherin Joubran, Simona Bar-Haim, Lior Shmuelof
Scientific Reports.2022;[Epub] CrossRef - Motor imagery in amyotrophic lateral Sclerosis: An fMRI study of postural control
Malek Abidi, Pierre-Francois Pradat, Nicolas Termoz, Annabelle Couillandre, Peter Bede, Giovanni de Marco
NeuroImage: Clinical.2022; 35: 103051. CrossRef - Brain and muscle activation patterns during postural control affect static postural control
Ken Kumai, Yumi Ikeda, Katsuya Sakai, Keisuke Goto, Kenji Morikawa, Keiichirou Shibata
Gait & Posture.2022; 96: 102. CrossRef - Decoding neural activity preceding balance loss during standing with a lower-limb exoskeleton using an interpretable deep learning model
Akshay Sujatha Ravindran, Christopher A Malaya, Isaac John, Gerard E Francisco, Charles Layne, Jose L Contreras-Vidal
Journal of Neural Engineering.2022; 19(3): 036015. CrossRef - Free-living gait does not differentiate chronic mTBI patients compared to healthy controls
Dylan Powell, Alan Godfrey, Lucy Parrington, Kody R. Campbell, Laurie A. King, Sam Stuart
Journal of NeuroEngineering and Rehabilitation.2022;[Epub] CrossRef - The “Hand as Foot” teaching method in anatomy of the inner ear
Meng Xu, Feifei Zhao, Yanlin Chen, Xiaobo Zhu
Asian Journal of Surgery.2022; 45(11): 2451. CrossRef - Assessing Neurokinematic and Neuromuscular Connectivity During Walking Using Mobile Brain-Body Imaging
Mingqi Zhao, Gaia Bonassi, Jessica Samogin, Gaia Amaranta Taberna, Camillo Porcaro, Elisa Pelosin, Laura Avanzino, Dante Mantini
Frontiers in Neuroscience.2022;[Epub] CrossRef - Stabilometric Biofeedback Training in Cognitive and Affective Function Improvement. Contribution of the Russian Scientific School. Part II
O. M. Bazanova, A. V. Kovaleva
Human Physiology.2022; 48(3): 271. CrossRef - Gait analysis under the lens of statistical physics
Massimiliano Zanin, Felipe Olivares, Irene Pulido-Valdeolivas, Estrella Rausell, David Gomez-Andres
Computational and Structural Biotechnology Journal.2022; 20: 3257. CrossRef - Role of primary motor cortex in gait: automatic-voluntary dissociation seen in paretic leg of a patient who had a stroke
Masanori Nagaoka, Yasuhiro Kumakura, Katsuyuki Inaba, Akira Ebihara, Miyu Usui
BMJ Neurology Open.2022; 4(1): e000275. CrossRef - Reliability and Validity of Mini-Balance Evaluation System Test in Type 2 Diabetic Patients with Peripheral Neuropathy
Sitt Nyein Phyu, Punnee Peungsuwan, Rungthip Puntumetakul, Uraiwan Chatchawan
International Journal of Environmental Research and Public Health.2022; 19(11): 6944. CrossRef - Gray Matter Abnormalities in Myotonic Dystrophy Type 1: A Voxel-Wise Meta-Analysis
Qirui Jiang, Junyu Lin, Chunyu Li, Yanbing Hou, Huifang Shang
Frontiers in Neurology.2022;[Epub] CrossRef - Association of Cervical Vestibular-Evoked Myogenic Potentials (VEMP) and Postural Instability in Patients with Parkinson’s Disease: A Cross-Sectional Study
Archana Gaur, Ravichandran Rajamanickam, Chennappan Chinnathambi, Divya Selvaraju, Varatharajan Sakthivadivel
Annals of Neurosciences.2022; 29(2-3): 137. CrossRef - Neurodegenerative and Cerebrovascular Brain Pathologies Are Differentially Associated With Declining Grip Strength and Gait In Older Adults
Shahram Oveisgharan, Lei Yu, Tianhao Wang, Julie A Schneider, David A Bennett, Aron S Buchman, Lewis A Lipsitz
The Journals of Gerontology: Series A.2022;[Epub] CrossRef - Resting state functional networks predict different aspects of postural control in Parkinson’s disease
Anjanibhargavi Ragothaman, Martina Mancini, John G. Nutt, Damien A. Fair, Oscar Miranda-Dominguez, Fay B. Horak
Gait & Posture.2022; 97: 122. CrossRef - Cumulative additional information does not improve the neuromuscular control during postural responses to perturbations in postural instability/gait disorders subtype of Parkinson's disease
Victor Spiandor Beretta, Paulo Cezar Rocha Santos, Diego Orcioli-Silva, Diego Alejandro Rojas Jaimes, Marcelo Pinto Pereira, Fabio Augusto Barbieri, Lilian Teresa Bucken Gobbi
Experimental Gerontology.2022; 166: 111892. CrossRef - Relation between Cortical Activation and Effort during Robot-Mediated Walking in Healthy People: A Functional Near-Infrared Spectroscopy Neuroimaging Study (fNIRS)
Julien Bonnal, Fanny Monnet, Ba-Thien Le, Ophélie Pila, Anne-Gaëlle Grosmaire, Canan Ozsancak, Christophe Duret, Pascal Auzou
Sensors.2022; 22(15): 5542. CrossRef - Association of Genetic Variant Linked to Hemochromatosis With Brain Magnetic Resonance Imaging Measures of Iron and Movement Disorders
Robert Loughnan, Jonathan Ahern, Cherisse Tompkins, Clare E. Palmer, John Iversen, Wesley K. Thompson, Ole Andreassen, Terry Jernigan, Leo Sugrue, Anders Dale, Mary E. T. Boyle, Chun Chieh Fan
JAMA Neurology.2022; 79(9): 919. CrossRef - Muscle Strength and Balance as Mediators in the Association between Physical Activity and Health-Related Quality of Life in Community-Dwelling Older Adults
Marcelo de Maio Nascimento, Bruna R. Gouveia, Élvio Rúbio Gouveia, Pedro Campos, Adilson Marques, Andreas Ihle
Journal of Clinical Medicine.2022; 11(16): 4857. CrossRef - Spinal cord stimulation therapy for patients with Parkinson’s disease and gait problems (STEP-PD): study protocol for an exploratory, double-blind, randomised, placebo-controlled feasibility trial
Victor S Hvingelby, Miriam Højholt Terkelsen, Erik L Johnsen, Mette Møller, Erik Hvid Danielsen, Tove Henriksen, Andreas Nørgaard Glud, Yen Tai, Anne Sofie Møller Andersen, Kaare Meier, Per Borghammer, Elena Moro, Jens Christian Hedemann Sørensen, Nicola
BMJ Neurology Open.2022; 4(2): e000333. CrossRef - Adaptations of postural sway dynamics and cortical response to unstable stance with stroboscopic vision in older adults
Yi-Ying Tsai, Yi-Ching Chen, Chen-Guang Zhao, Ing-Shiou Hwang
Frontiers in Physiology.2022;[Epub] CrossRef - Blood pressure changes impact corticospinal integrity and downstream gait and balance control
Elizabeth A. Coon, Anna M. Castillo, Timothy G. Lesnick, Sheelakumari Raghavan, Michelle M. Mielke, Robert I. Reid, B. Gwen Windham, Ronald C. Petersen, Clifford R. Jack, Jonathan Graff-Radford, Prashanthi Vemuri
Neurobiology of Aging.2022; 120: 60. CrossRef - Mobile Brain Imaging to Examine Task-Related Cortical Correlates of Reactive Balance: A Systematic Review
Rudri Purohit, Tanvi Bhatt
Brain Sciences.2022; 12(11): 1487. CrossRef - Transcranial direct current stimulation for balance rehabilitation in neurological disorders: A systematic review and meta-analysis
Victor Spiandor Beretta, Paulo Cezar Rocha Santos, Diego Orcioli-Silva, Vinicius Cavassano Zampier, Rodrigo Vitório, Lilian Teresa Bucken Gobbi
Ageing Research Reviews.2022; 81: 101736. CrossRef - Freezing of gait, gait initiation, and gait automaticity share a similar neural substrate in Parkinson's disease
Acácio Moreira-Neto, Carlos Ugrinowitsch, Daniel Boari Coelho, Andrea Cristina de Lima-Pardini, Egberto Reis Barbosa, Luis Augusto Teixeira, Edson Amaro, Fay B. Horak, Martina Mancini, Mariana Penteado Nucci, Carla Silva-Batista
Human Movement Science.2022; 86: 103018. CrossRef - Association between Backward Walking and Cognition in Parkinson Disease: A Systematic Review
Mathias Baptiste Correno, Clint Hansen, Matthias Chardon, Tracy Milane, Edoardo Bianchini, Nicolas Vuillerme
International Journal of Environmental Research and Public Health.2022; 19(19): 12810. CrossRef - The Effect of Dual-Task Motor-Cognitive Training in Adults with Neurological Diseases Who Are at Risk of Falling
Barbara Spanò, Massimo De Tollis, Sara Taglieri, Alice Manzo, Claudia Ricci, Maria G. Lombardi, Lorenzo Polidori, Ivo A. Griffini, Marta Aloisi, Vincenzo Vinicola, Rita Formisano, Carlo Caltagirone, Roberta Annicchiarico
Brain Sciences.2022; 12(9): 1207. CrossRef - Connecting real-world digital mobility assessment to clinical outcomes for regulatory and clinical endorsement–the Mobilise-D study protocol
A. Stefanie Mikolaizak, Lynn Rochester, Walter Maetzler, Basil Sharrack, Heleen Demeyer, Claudia Mazzà, Brian Caulfield, Judith Garcia-Aymerich, Beatrix Vereijken, Valdo Arnera, Ram Miller, Paolo Piraino, Nadir Ammour, Mark Forrest Gordon, Thierry Trooste
PLOS ONE.2022; 17(10): e0269615. CrossRef - Toxicity evaluation induced by single and 28-days repeated exposure of withametelin and daturaolone in Sprague Dawley rats
Muhammad Waleed Baig, Muhammad Majid, Bakht Nasir, Syed Shams ul Hassan, Simona Bungau, Ihsan-ul Haq
Frontiers in Pharmacology.2022;[Epub] CrossRef - Cognitive Function and Postural Control Strategies in Relation to Disease Progression in Patients with Parkinson’s Disease
Monika Zawadka-Kunikowska, Jacek J. Klawe, Małgorzata Tafil-Klawe, Monika Bejtka, Łukasz Rzepiński, Mirosława Cieślicka
International Journal of Environmental Research and Public Health.2022; 19(19): 12694. CrossRef - Effect of sports background on the visual and vestibular signal processing abilities of athletes
F. Zsarnoczky-Dulhazi, A. Hegedus, P. Soldos, L. Trzaskoma, B. Kopper
Science & Sports.2022; 37(8): 798.e1. CrossRef - Abnormal neural oscillations during gait and dual-task in Parkinson’s disease
Rachel O. Nwogo, Stefan Kammermeier, Arun Singh
Frontiers in Systems Neuroscience.2022;[Epub] CrossRef - Effects of different virtual reality technology driven dual-tasking paradigms on posture and saccadic eye movements in healthy older adults
Yu Imaoka, Andri Flury, Laura Hauri, Eling D. de Bruin
Scientific Reports.2022;[Epub] CrossRef - Principles of gait encoding in the subthalamic nucleus of people with Parkinson’s disease
Yohann Thenaisie, Kyuhwa Lee, Charlotte Moerman, Stefano Scafa, Andrea Gálvez, Elvira Pirondini, Morgane Burri, Jimmy Ravier, Alessandro Puiatti, Ettore Accolla, Benoit Wicki, André Zacharia, Mayte Castro Jiménez, Julien F. Bally, Grégoire Courtine, Jocel
Science Translational Medicine.2022;[Epub] CrossRef - Glutamatergic pedunculopontine tegmental neurons control wakefulness and locomotion via distinct axonal projections
Daniel Kroeger, Jack Thundercliffe, Alex Phung, Roberto De Luca, Carolyn Geraci, Samuel Bragg, Kayleen J McCafferty, Sathyajit S Bandaru, Elda Arrigoni, Thomas E Scammell
Sleep.2022;[Epub] CrossRef - Exploring Mediation Effects of Gait Speed, Body Balance, and Falls in the Relationship between Physical Activity and Health-Related Quality of Life in Vulnerable Older Adults
Marcelo de Maio Nascimento, Élvio Rúbio Gouveia, Bruna R. Gouveia, Adilson Marques, Cíntia França, Duarte L. Freitas, Pedro Campos, Andreas Ihle
International Journal of Environmental Research and Public Health.2022; 19(21): 14135. CrossRef - Fronto-parietal cortex activation during walking in patients with Parkinson's disease adopting different postural strategies
Xinyuan Zhang, Yue Wang, Jiewei Lu, Jin Wang, Zhilin Shu, Yuanyuan Cheng, Zhizhong Zhu, PeiPei Liu, Yang Yu, Ningbo Yu, Jianda Han, Jialing Wu
Frontiers in Neurology.2022;[Epub] CrossRef - Perioperative Brain Function Monitoring with Electroencephalography in Horses Anesthetized with Multimodal Balanced Anesthetic Protocol Subjected to Surgeries
Carla Murillo, Hsin-Yi Weng, Ann B. Weil, Matthias Kreuzer, Jeff C. Ko
Animals.2022; 12(20): 2851. CrossRef - Prediction of the gait function using the nigrostriatal and corticoreticulospinal tracts of the affected hemisphere in a cerebral infarct: A diffusion tensor imaging study
Sung Ho Jang, Sang Seok Yeo, Min Jye Cho
Medicine.2022; 101(39): e30788. CrossRef - The Romberg's sign: from walking in the dark to tests on the force plate
Anastasia I. Mezenchuk, Oleg V. Kubryak
Almanac of Clinical Medicine.2022; 50(5): 335. CrossRef - Role of Cervical Spinal Magnetic Stimulation in Improving Posture and Functional Ambulation of Patients with Relapsing Remitting Multiple Sclerosis
Shereen I. Fawaz, Shin-Ichi Izumi, Soha M. Hamada, Abir A. Omara, Ghada O. Wassef, Heba Gamal Saber, Sherihan M. Salama, Cristina Fonte
Rehabilitation Research and Practice.2022; 2022: 1. CrossRef - The brain-body disconnect: A somatic sensory basis for trauma-related disorders
Breanne E. Kearney, Ruth A. Lanius
Frontiers in Neuroscience.2022;[Epub] CrossRef - Dual-task walking improvement with enhanced kinesthetic awareness in Parkinson’s disease with mild gait impairment: EEG connectivity and clinical implication
Cheng-Ya Huang, Yu-An Chen, Ruey-Meei Wu, Ing-Shiou Hwang
Frontiers in Aging Neuroscience.2022;[Epub] CrossRef - Trends in vestibular pathology: persistent postural perceptual dizziness (PPPD)
Daniela Cîrpaciu, Vlad Andrei Budu, Cristina-Maria Goanţă
ORL.ro.2022; 4(57): 14. CrossRef - Biomechanical and Sensory Feedback Regularize the Behavior of Different Locomotor Central Pattern Generators
Kaiyu Deng, Alexander J. Hunt, Nicholas S. Szczecinski, Matthew C. Tresch, Hillel J. Chiel, C. J. Heckman, Roger D. Quinn
Biomimetics.2022; 7(4): 226. CrossRef - Phase-dependent modulation of the vestibular–cerebellar network via combined alternating current stimulation influences human locomotion and posture
Hisato Nakazono, Takanori Taniguchi, Tsubasa Mitsutake, Akinori Takeda, Emi Yamada, Katsuya Ogata
Frontiers in Neuroscience.2022;[Epub] CrossRef - Gait Recovery Characteristic According to the Injury Aspect of Descending Motor Pathway in a Chronic Stroke Patient: a Case Study
Sang Seok Yeo
The Journal of Korean Physical Therapy.2022; 34(6): 326. CrossRef - Cortico-Subthalamic Field Potentials Support Classification of the Natural Gait Cycle in Parkinson’s Disease and Reveal Individualized Spectral Signatures
Kenneth H. Louie, Ro’ee Gilron, Maria S. Yaroshinsky, Melanie A. Morrison, Julia Choi, Coralie de Hemptinne, Simon Little, Philip A. Starr, Doris D. Wang
eneuro.2022; 9(6): ENEURO.0325-22.2022. CrossRef - Test-retest reliability and minimal detectable change of the computerized dynamic posturography PROPRIO for adults with chronic traumatic brain injury
Guilherme M. Cesar, Thad W. Buster, Judith M. Burnfield
Disability and Rehabilitation.2021; 43(14): 2038. CrossRef - Joint-Position Sense Accuracy Is Equally Affected by Vision among Children with and without Cerebral Palsy
Flávia de Andrade e Souza Mazuchi, Luis Mochizuki, Joseph Hamill, Patricia Martins Franciulli, Aline Bigongiari, Isabella Tábata de Almeida Martins, Ulysses Fernandes Ervilha
Journal of Motor Behavior.2021; 53(2): 209. CrossRef - Cortical responses to whole‐body balance perturbations index perturbation magnitude and predict reactive stepping behavior
Teodoro Solis‐Escalante, Mitchel Stokkermans, Michael X. Cohen, Vivian Weerdesteyn
European Journal of Neuroscience.2021; 54(12): 8120. CrossRef - White Matter Microstructure of the Cerebellar Peduncles Is Associated with Balance Performance during Sensory Re-Weighting in People with Multiple Sclerosis
Arianna D. Odom, Sutton B. Richmond, Brett W. Fling
The Cerebellum.2021; 20(1): 92. CrossRef - Highly challenging balance and gait training for individuals with Parkinson’s disease improves pace, rhythm and variability domains of gait – A secondary analysis from a randomized controlled trial
Linda Rennie, Arve Opheim, Espen Dietrichs, Niklas Löfgren, Erika Franzén
Clinical Rehabilitation.2021; 35(2): 200. CrossRef - Traumatic brain injury-induced cerebral microbleeds in the elderly
Luca Toth, Andras Czigler, Peter Horvath, Balint Kornyei, Nikolett Szarka, Attila Schwarcz, Zoltan Ungvari, Andras Buki, Peter Toth
GeroScience.2021; 43(1): 125. CrossRef - Altered prefrontal cortex responses in older adults with subjective memory complaints and dementia during dual‐task gait: An fNIRS study
Wei‐Peng Teo, Timo Rantalainen, Nathan Nuzum, Leah Valente, Helen Macpherson
European Journal of Neuroscience.2021; 53(4): 1324. CrossRef - Connectivity of the Cingulate Sulcus Visual Area (CSv) in Macaque Monkeys
V De Castro, A T Smith, A L Beer, C Leguen, N Vayssière, Y Héjja-Brichard, P Audurier, B R Cottereau, J B Durand
Cerebral Cortex.2021; 31(2): 1347. CrossRef - Efficient Actor-Critic Reinforcement Learning With Embodiment of Muscle Tone for Posture Stabilization of the Human Arm
Masami Iwamoto, Daichi Kato
Neural Computation.2021; 33(1): 129. CrossRef - Single-Session Cerebellar Transcranial Direct Current Stimulation Affects Postural Control Learning and Cerebellar Brain Inhibition in Healthy Individuals
Natsuki Katagiri, Saki Kawakami, Sayuri Okuyama, Tadaki Koseki, Daisuke Kudo, Shigehiro Namba, Shigeo Tanabe, Tomofumi Yamaguchi
The Cerebellum.2021; 20(2): 203. CrossRef - Is freezing of gait correlated with postural control in patients with moderate‐to‐severe Parkinson’s disease?
Daniel Boari Coelho, Caroline Ribeiro de Souza, Andrea Cristina de Lima‐Pardini, Renata de Castro Treza, Thiago Kenzo Fujioka Shida, Carla Silva‐Batista, Luis Augusto Teixeira, Yoland Smith
European Journal of Neuroscience.2021; 53(4): 1189. CrossRef - The use of Motor and Cognitive Dual-Task quantitative assessment on subjects with mild cognitive impairment: A systematic review
Gianmaria Mancioppi, Laura Fiorini, Erika Rovini, Filippo Cavallo
Mechanisms of Ageing and Development.2021; 193: 111393. CrossRef - Functional Near-infrared Spectroscopy Reveals the Compensatory Potential of Pre-frontal Cortical Activity for Standing Balance in Young and Older Adults
Rebecca J. St George, Mark R. Hinder, Rohan Puri, Eliza Walker, Michele L. Callisaya
Neuroscience.2021; 452: 208. CrossRef - Effects of Cognitive Over Postural Demands on Upright Standing Among Young Adults
Kell Grandjean da Costa, Erika K. Hussey, Eduardo Bodnariuc Fontes, Alekya Menta, John W. Ramsay, Clifford L. Hancock, Kari L. Loverro, Elizabeth Marfeo, Nathan Ward
Perceptual and Motor Skills.2021; 128(1): 80. CrossRef - Asymmetry and Variability Should Be Included in the Assessment of Gait Function in Poststroke Hemiplegia With Independent Ambulation During Early Rehabilitation
Woo-Sub Kim, Hanboram Choi, Jung-Woo Jung, Joon Sik Yoon, Ju Hyong Jeoung
Archives of Physical Medicine and Rehabilitation.2021; 102(4): 611. CrossRef - Electroencephalographic Study on Sensory Integration in Visually Induced Postural Sway
Takahiro Kagawa, Scott Makeig, Makoto Miyakoshi
Journal of Cognitive Neuroscience.2021; 33(3): 482. CrossRef - Benefits of nonlinear analysis indices of walking stride interval in the evaluation of neurodegenerative diseases
Frédéric Dierick, Charlotte Vandevoorde, Frédéric Chantraine, Olivier White, Fabien Buisseret
Human Movement Science.2021; 75: 102741. CrossRef - Freezing of Gait in Parkinson’s Disease: Invasive and Noninvasive Neuromodulation
Shervin Rahimpour, Wendy Gaztanaga, Amol P. Yadav, Stephano J. Chang, Max O. Krucoff, Iahn Cajigas, Dennis A. Turner, Doris D. Wang
Neuromodulation: Technology at the Neural Interface.2021; 24(5): 829. CrossRef - Response Inhibition Deficits in Women with the FMR1 Premutation are Associated with Age and Fall Risk
Carly Moser, Lyndsay Schmitt, Joseph Schmidt, Amanda Fairchild, Jessica Klusek
Brain and Cognition.2021; 148: 105675. CrossRef - The potential of noisy galvanic vestibular stimulation for optimizing and assisting human performance
Kim Lajoie, Daniel S. Marigold, Bulmaro A. Valdés, Carlo Menon
Neuropsychologia.2021; 152: 107751. CrossRef - Evaluation of vestibular functions and balance in Edirne Band musicians
Halit Selçuk, Sevgi Özdinç, Menekşe Karahan, Ayşe Zeynep Yılmazer Kayatekin, Enis Uluçam
Work.2021; 68(2): 415. CrossRef - A review of movement disorders in chemotherapy-induced neurotoxicity
Allison B. Wang, Stephen N. Housley, Ann Marie Flores, Sheetal M. Kircher, Eric J. Perreault, Timothy C. Cope
Journal of NeuroEngineering and Rehabilitation.2021;[Epub] CrossRef - REM Sleep Without Atonia and Gait Impairment in People with Mild-to-Moderate Parkinson’s Disease
Sommer L. Amundsen-Huffmaster, Matthew N. Petrucci, Maria E. Linn-Evans, Jae Woo Chung, Michael J. Howell, Aleksandar Videnovic, Paul J. Tuite, Scott E. Cooper, Colum D. MacKinnon
Journal of Parkinson's Disease.2021; 11(2): 767. CrossRef - Age-Related Changes in Presynaptic Inhibition During Gait Initiation
Solival Santos Filho, Daniel Boari Coelho, Carlos Ugrinowitsch, Caroline Ribeiro de Souza, Fernando Henrique Magalhães, Andrea Cristina de Lima-Pardini, Éden Marcos Braga de Oliveira, Eugenia Mattos, Luis Augusto Teixeira, Carla Silva-Batista, David Le Co
The Journals of Gerontology: Series A.2021; 76(4): 568. CrossRef - A synthetic kinematic index of trunk displacement conveying the overall motor condition in Parkinson’s disease
Emahnuel Troisi Lopez, Roberta Minino, Pierpaolo Sorrentino, Rosaria Rucco, Anna Carotenuto, Valeria Agosti, Domenico Tafuri, Valentino Manzo, Marianna Liparoti, Giuseppe Sorrentino
Scientific Reports.2021;[Epub] CrossRef - Gait in Benign Paroxysmal Positional Vertigo
Yong-Hyun Lim, Kyunghun Kang, Ho-Won Lee, Ji-Soo Kim, Sung-Hee Kim
Frontiers in Neurology.2021;[Epub] CrossRef - Pigeon as a model to study peripheral projections from the horizontal semicircular canal vestibular apparatus to a brainstem target immunoreactive for AMPA
João Roberto Rocha, Maria de Fátima Passetto, Julianne dos Santos Maldonado-Menetti, Ana Lúcia Beirão Cabral, Claudio Antônio Barbosa de Toledo, Marcia Koike
Acta Cirúrgica Brasileira.2021;[Epub] CrossRef - Center of pressure responses to unpredictable external perturbations indicate low accuracy in predicting fall risk in people with Parkinson’s disease
Gabriel Antonio Gazziero Moraca, Victor Spiandor Beretta, Paulo Cezar Rocha dos Santos, Priscila Nóbrega‐Sousa, Diego Orcioli‐Silva, Rodrigo Vitório, Lilian Teresa Bucken Gobbi
European Journal of Neuroscience.2021; 53(8): 2901. CrossRef - Gait analysis in a rat model of traumatic brain injury
John Reed, Antigone Grillakis, Alyssa Kline, Anwar E. Ahmed, Kimberly R. Byrnes
Behavioural Brain Research.2021; 405: 113210. CrossRef - Vitamin D, Falls and Balance Capacity Impacts in Older Adults: Update
Ray Marks
Journal of Aging Research and Healthcare.2021; 3(4): 22. CrossRef - Coordinated features in jaw and neck muscle activities induced by chewing of soft and hard gum in healthy subjects
Tomohiro Ishii, Noriyuki Narita, Hiroshi Endo, Masanobu Wakami, Masakazu Okubo, Takeshi Uchida, Ikuo Kantake, Koh Shibutani
Clinical and Experimental Dental Research.2021; 7(5): 868. CrossRef - Cortical activity and gait parameter characteristics in people with multiple sclerosis during unobstructed gait and obstacle avoidance
Felipe Balistieri Santinelli, Emerson Sebastião, Marina Hiromi Kuroda, Vinicius Christianini Moreno, Julia Pilon, Luiz Henrique Palucci Vieira, Fabio Augusto Barbieri
Gait & Posture.2021; 86: 226. CrossRef - Distinct cortical thickness patterns link disparate cerebral cortex regions to select mobility domains
Inbal Maidan, Anat Mirelman, Jeffrey M. Hausdorff, Yaakov Stern, Christian G. Habeck
Scientific Reports.2021;[Epub] CrossRef - Brain connectivity during simulated balance in older adults with and without Parkinson's disease
Elizabeth P. Pasman, Martin J. McKeown, Saurabh Garg, Taylor W. Cleworth, Bastiaan R. Bloem, J. Timothy Inglis, Mark G. Carpenter
NeuroImage: Clinical.2021; 30: 102676. CrossRef - Innovative motor and cognitive dual-task approaches combining upper and lower limbs may improve dementia early detection
Gianmaria Mancioppi, Laura Fiorini, Erika Rovini, Radia Zeghari, Auriane Gros, Valeria Manera, Philippe Robert, Filippo Cavallo
Scientific Reports.2021;[Epub] CrossRef - Cognition mediates the relation between structural network efficiency and gait in small vessel disease
Mengfei Cai, Mina A. Jacob, David G. Norris, Marco Duering, Frank-Erik de Leeuw, Anil M. Tuladhar
NeuroImage: Clinical.2021; 30: 102667. CrossRef - Does the impaired postural control in Parkinson's disease affect the habituation to non-sequential external perturbation trials?
Victor Spiandor Beretta, Mark Gregory Carpenter, Fabio Augusto Barbieri, Paulo Cezar Rocha Santos, Diego Orcioli-Silva, Marcelo Pinto Pereira, Lilian Teresa Bucken Gobbi
Clinical Biomechanics.2021; 85: 105363. CrossRef - A Neural Model of Intrinsic and Extrinsic Hippocampal Theta Rhythms: Anatomy, Neurophysiology, and Function
Stephen Grossberg
Frontiers in Systems Neuroscience.2021;[Epub] CrossRef - A randomized sham-controlled trial on the effect of continuous positive airway pressure treatment on gait control in severe obstructive sleep apnea patients
Sébastien Baillieul, Bernard Wuyam, Dominic Pérennou, Renaud Tamisier, Sébastien Bailly, Meriem Benmerad, Céline Piscicelli, Thibault Le Roux-Mallouf, Samuel Vergès, Jean-Louis Pépin
Scientific Reports.2021;[Epub] CrossRef - Detecting Depression Through Gait Data: Examining the Contribution of Gait Features in Recognizing Depression
Yameng Wang, Jingying Wang, Xiaoqian Liu, Tingshao Zhu
Frontiers in Psychiatry.2021;[Epub] CrossRef - Effects of prolonged-release fampridine on multiple sclerosis-related gait impairments. A crossover, double-blinded, placebo-controlled study
Maxime Valet, Souraya El Sankari, Vincent Van Pesch, Christine Detrembleur, Thierry Lejeune, Gaëtan Stoquart
Clinical Biomechanics.2021; 86: 105382. CrossRef - Gait Impairment and Upper Extremity Disturbance Are Associated With Total Magnetic Resonance Imaging Cerebral Small Vessel Disease Burden
Yutong Hou, Yue Li, Shuna Yang, Wei Qin, Lei Yang, Wenli Hu
Frontiers in Aging Neuroscience.2021;[Epub] CrossRef - Long-Lasting Event-Related Beta Synchronizations of Electroencephalographic Activity in Response to Support-Surface Perturbations During Upright Stance: A Pilot Study Associating Beta Rebound and Active Monitoring in the Intermittent Postural Control
Akihiro Nakamura, Yasuyuki Suzuki, Matija Milosevic, Taishin Nomura
Frontiers in Systems Neuroscience.2021;[Epub] CrossRef - Dual-task walking and automaticity after Stroke: Insights from a secondary analysis and imaging sub-study of a randomised controlled trial
Johnny Collett, Melanie K Fleming, Daan Meester, Emad Al-Yahya, Derick T Wade, Andrea Dennis, Piergiorgio Salvan, Andrew Meaney, Janet Cockburn, Joanna Dawes, Heidi Johansen-Berg, Helen Dawes
Clinical Rehabilitation.2021; 35(11): 1599. CrossRef - Aerobic Exercise Combined With Transcranial Direct Current Stimulation Over the Prefrontal Cortex in Parkinson Disease: Effects on Cortical Activity, Gait, and Cognition
Núbia Ribeiro Conceição, Lilian Teresa Bucken Gobbi, Priscila Nóbrega-Sousa, Diego Orcioli-Silva, Victor Spiandor Beretta, Ellen Lirani-Silva, Alexandre Hideki Okano, Rodrigo Vitório
Neurorehabilitation and Neural Repair.2021; 35(8): 717. CrossRef - Non-ambulatory measures of lower extremity sensorimotor function are associated with walking function in Multiple Sclerosis
Sumire Sato, John Buonaccorsi, Jules D. Miehm, Jongil Lim, Caitlin Rajala, Farnaz Khalighinejad, Carolina Ionete, Jane A. Kent, Richard E.A. van Emmerik
Multiple Sclerosis and Related Disorders.2021; 53: 103051. CrossRef - Self-Perception of the Knee Is Associated with Joint Motion during the Loading Response in Individuals with Knee Osteoarthritis: A Pilot Cross-Sectional Study
Haruki Toda, Tsubasa Maruyama, Koji Fujita, Yuki Yamauchi, Mitsunori Tada
Sensors.2021; 21(12): 4009. CrossRef - Fields of Forel Brain Stimulation Improves Levodopa-Unresponsive Gait and Balance Disorders in Parkinson's Disease
Maria Sheila Guimarães Rocha, Julian Leticia de Freitas, Carlos Daniel Miranda Costa, Maira Okada de Oliveira, Paulo Roberto Terzian, João Welberthon Matos Queiroz, Jamana Barbosa Ferraz, João Fellipe Santos Tatsch, Diogo Coutinho Soriano, Clement Hamani,
Neurosurgery.2021; 89(3): 450. CrossRef - The effects of transcranial direct current stimulation on gait in patients with Parkinson’s disease: a systematic review
Fateme Pol, Mohammad Ali Salehinejad, Hamzeh Baharlouei, Michael A. Nitsche
Translational Neurodegeneration.2021;[Epub] CrossRef - Cerebral Hemodynamic Responses to the Difficulty Level of Ambulatory Tasks in Patients With Parkinson’s Disease: A Systematic Review and Meta-Analysis
Jin P. Lin, Hong S. Feng, Hua Zhai, Xia Shen
Neurorehabilitation and Neural Repair.2021; 35(9): 755. CrossRef - The Dendrite Arbor of Purkinje Cells Is Altered Following to Tail Regeneration in the Leopard Gecko
Stefanie S Bradley, Erika Howe, Craig D C Bailey, Matthew K Vickaryous
Integrative and Comparative Biology.2021; 61(2): 370. CrossRef - The diagnosis of central vestibular disorder based on the complementary examination of the vestibulospinal reflex
Stefani Maihoub, András Molnár, László Tamás, Ágnes Szirmai
Journal of Otology.2021;[Epub] CrossRef - Compromised Brain Activity With Age During a Game-Like Dynamic Balance Task: Single- vs. Dual-Task Performance
Veerle de Rond, Diego Orcioli-Silva, Bauke Wybren Dijkstra, Jean-Jacques Orban de Xivry, Annette Pantall, Alice Nieuwboer
Frontiers in Aging Neuroscience.2021;[Epub] CrossRef - Cognitive and Motor Cortical Activity During Cognitively Demanding Stepping Tasks in Older People at Low and High Risk of Falling
Paulo H. S. Pelicioni, Stephen R. Lord, Daina L. Sturnieks, Bethany Halmy, Jasmine C. Menant
Frontiers in Medicine.2021;[Epub] CrossRef - Uniqueness of gait kinematics in a cohort study
Gunwoo Park, Kyoung Min Lee, Seungbum Koo
Scientific Reports.2021;[Epub] CrossRef - Visual Effect on Brain Connectome That Scales Feedforward and Feedback Processes of Aged Postural System During Unstable Stance
Yi-Ching Chen, Chien-Chun Huang, Chen-Guang Zhao, Ing-Shiou Hwang
Frontiers in Aging Neuroscience.2021;[Epub] CrossRef - The Biology and Pathobiology of Glutamatergic, Cholinergic, and Dopaminergic Signaling in the Aging Brain
Anna Gasiorowska, Malgorzata Wydrych, Patrycja Drapich, Maciej Zadrozny, Marta Steczkowska, Wiktor Niewiadomski, Grazyna Niewiadomska
Frontiers in Aging Neuroscience.2021;[Epub] CrossRef - Dual-Task Training Effect on Cognitive and Body Function, β-amyloid Levels in Alzheimer’s Dementia Patients: A Randomized Controlled Trial
Seung-Min Nam, Seong-gil Kim
The Journal of Korean Physical Therapy.2021; 33(3): 136. CrossRef - Graph‐theoretical analysis of EEG functional connectivity during balance perturbation in traumatic brain injury: A pilot study
Vikram Shenoy Handiru, Alaleh Alivar, Armand Hoxha, Soha Saleh, Easter S. Suviseshamuthu, Guang H. Yue, Didier Allexandre
Human Brain Mapping.2021; 42(14): 4427. CrossRef - Adapting Footfall Rhythmicity to Auditory Perturbations Affects Resilience of Locomotor Behavior: A Proof-of-Concept Study
Deepak K. Ravi, Caroline C. Heimhofer, William R. Taylor, Navrag B. Singh
Frontiers in Neuroscience.2021;[Epub] CrossRef - Towards real-world generalizability of a circuit for action-stopping
Ricci Hannah, Adam R. Aron
Nature Reviews Neuroscience.2021; 22(9): 538. CrossRef - Physiological and cognitive determinants of gait in middle-aged, older-aged and oldest-aged Asian adults – The Yishun study
Lay Khoon Lau, Shiou Liang Wee, Jagadish Ullal Mallya, Philip Lin Kiat Yap, Wei Jun Benedict Pang, Kexun Kenneth Chen, Khalid Abdul Jabbar, Wei Ting Seah, Tze Pin Ng
Aging and Health Research.2021; 1(3): 100030. CrossRef - Amble Gait EEG Points at Complementary Cortical Networks Underlying Stereotypic Multi-Limb Co-ordination
Joyce B. Weersink, Natasha M. Maurits, Bauke M. de Jong
Frontiers in Human Neuroscience.2021;[Epub] CrossRef - Age-Related Differences in Corticospinal Excitability and Anticipatory Postural Adjustments of the Trunk
Rebecca S. Rowland, Ned Jenkinson, Shin-Yi Chiou
Frontiers in Aging Neuroscience.2021;[Epub] CrossRef - Postural sway in the moving room scenario: New evidence for functional dissociation between self-motion perception and postural control
Kentaro Horiuchi, Kuniyasu Imanaka, Masami Ishihara, Manabu Sakakibara
PLOS ONE.2021; 16(9): e0257212. CrossRef - Altered Cortical Cholinergic Network in Parkinson’s Disease at Different Stage: A Resting-State fMRI Study
Wenshuang Sheng, Tao Guo, Cheng Zhou, Jingjing Wu, Ting Gao, Jiali Pu, Baorong Zhang, Minming Zhang, Yunjun Yang, Xiaojun Guan, Xiaojun Xu
Frontiers in Aging Neuroscience.2021;[Epub] CrossRef - Gait phenotype in batten disease: A marker of disease progression
John R. Ostergaard
European Journal of Paediatric Neurology.2021;[Epub] CrossRef - High Sensory Responsiveness in Older Adults is Associated with Walking Outside but Not Inside: Proof of Concept Study
Maayan Agmon, Tami Bar-Shalita, Rachel Kizony
Clinical Interventions in Aging.2021; Volume 16: 1651. CrossRef - Association of Posture Instability with Dopamine Drop of Nigrostriatal System and Hypometabolism of Cerebral Cortex in Parkinson’s Disease
Hongyan Wang, Hong-Yu Li, Xiuhai Guo, Yongtao Zhou
Current Neurovascular Research.2021; 18(2): 244. CrossRef - Does Subthalamic Deep Brain Stimulation Impact Asymmetry and Dyscoordination of Gait in Parkinson’s Disease?
Deepak K. Ravi, Christian R. Baumann, Elena Bernasconi, Michelle Gwerder, Niklas K. Ignasiak, Mechtild Uhl, Lennart Stieglitz, William R. Taylor, Navrag B. Singh
Neurorehabilitation and Neural Repair.2021; 35(11): 1020. CrossRef - Any Behavioral Change May Have Physiological Significance: Benign Neglect In Tier I Neurotoxicity Testing
David V. Gauvin, Zachary J. Zimmermann
Current Opinion in Toxicology.2021;[Epub] CrossRef - Impact of regional white matter hyperintensities on specific gait function in Alzheimer's disease and mild cognitive impairment
Noriko Ogama, Hidetoshi Endo, Shosuke Satake, Shumpei Niida, Hidenori Arai, Takashi Sakurai
Journal of Cachexia, Sarcopenia and Muscle.2021; 12(6): 2045. CrossRef - The effects of different frequencies of rhythmic acoustic stimulation on gait stability in healthy elderly individuals: a pilot study
Roberta Minino, Emahnuel Troisi Lopez, Pierpaolo Sorrentino, Rosaria Rucco, Anna Lardone, Matteo Pesoli, Domenico Tafuri, Laura Mandolesi, Giuseppe Sorrentino, Marianna Liparoti
Scientific Reports.2021;[Epub] CrossRef - Feasibility and tolerance of a robotic postural training to improve standing in a person with ambulatory spinal cord injury
V. Santamaria, T. D. Luna, S. K. Agrawal
Spinal Cord Series and Cases.2021;[Epub] CrossRef - A Novel Viewpoint on the Anticipatory Postural Adjustments During Gait Initiation
Veronica Farinelli, Francesco Bolzoni, Silvia Maria Marchese, Roberto Esposti, Paolo Cavallari
Frontiers in Human Neuroscience.2021;[Epub] CrossRef - Tai Chi and Yoga for Improving Balance on One Leg: A Neuroimaging and Biomechanics Study
Xin-Peng Chen, Le-Jun Wang, Xiao-Qian Chang, Kuan Wang, Hui-Fang Wang, Ming Ni, Wen-Xin Niu, Ming Zhang
Frontiers in Neurology.2021;[Epub] CrossRef - Sleep deprivation affects gait control
Guilherme S. Umemura, João Pedro Pinho, Jacques Duysens, Hermano Igo Krebs, Arturo Forner-Cordero
Scientific Reports.2021;[Epub] CrossRef - Postural Control Strategies and Balance-Related Factors in Individuals with Traumatic Transtibial Amputations
Barbora Kolářová, Miroslav Janura, Zdeněk Svoboda, Petr Kolář, Dagmar Tečová, Milan Elfmark
Sensors.2021; 21(21): 7284. CrossRef - Walking While Talking and Prefrontal Oxygenation in Motoric Cognitive Risk Syndrome: Clinical and Pathophysiological Aspects
Cristina Udina, Emmeline Ayers, Marco Inzitari, Joe Verghese, Marco Canevelli
Journal of Alzheimer's Disease.2021; 84(4): 1585. CrossRef - Cortical and Cerebellar Oscillatory Responses to Postural Instability in Parkinson's Disease
Taylor J. Bosch, Stefan Kammermeier, Christopher Groth, Matt Leedom, Elizabeth K. Hanson, Patti Berg-Poppe, Arun Singh
Frontiers in Neurology.2021;[Epub] CrossRef - Circuits for State-Dependent Modulation of Locomotion
Alejandro J. Pernía-Andrade, Nikolaus Wenger, Maria S. Esposito, Philip Tovote
Frontiers in Human Neuroscience.2021;[Epub] CrossRef - Postural and gait symptoms in de novo Parkinson's disease patients correlate with cholinergic white matter pathology
Muhammad Nazmuddin, Jan-Willem van Dalen, Ronald J.H. Borra, Gilles N. Stormezand, Harm Jan van der Horn, Sygrid van der Zee, Jeffrey Boertien, Teus van Laar
Parkinsonism & Related Disorders.2021; 93: 43. CrossRef - Neurophysiological Support of Motor Control in “Hybrid” Postures. Literature Review
N. D. Babanov, E. A. Biryukova
Neuroscience and Behavioral Physiology.2021; 51(8): 1162. CrossRef - A Narrative Review of Alternate Gait Training Using Knee-ankle-foot Orthosis in Stroke Patients with Severe Hemiparesis
Hiroaki ABE, Kei KADOWAKI, Naohide TSUJIMOTO, Toru OKANUKA
Physical Therapy Research.2021; 24(3): 195. CrossRef - Intrinsic brainstem circuits comprised of Chx10-expressing neurons contribute to reticulospinal output in mice
Jeremy W. Chopek, Ying Zhang, Robert M. Brownstone
Journal of Neurophysiology.2021; 126(6): 1978. CrossRef - Spinal Cord Circuits: Models and Reality
Uwe Windhorst
Neurophysiology.2021; 53(3-6): 142. CrossRef - Cerebellar transcranial direct current stimulation modulates the effect of cerebellar transcranial magnetic stimulation on the excitability of spinal reflex
Akiyoshi Matsugi, Yohei Okada
Neuroscience Research.2020; 150: 37. CrossRef - Dynamic running hexapod robot based on high-performance computing
Xiaokun Leng, Songhao Piao, Lin Chang, Zhicheng He, Zheng Zhu
The Journal of Supercomputing.2020; 76(2): 844. CrossRef - Optic flow and attention alter locomotion differently in the young and old
Kenneth S. Pechtl, J. Richard Jennings, Mark S. Redfern
Gait & Posture.2020; 76: 1. CrossRef - Axonal regeneration and functional recovery driven by endogenous Nogo receptor antagonist LOTUS in a rat model of unilateral pyramidotomy
Ryu Ueno, Hajime Takase, Jun Suenaga, Masao Kishimoto, Yuji Kurihara, Kohtaro Takei, Nobutaka Kawahara, Tetsuya Yamamoto
Experimental Neurology.2020; 323: 113068. CrossRef - Local dynamic stability during long-fatiguing walks in people with multiple sclerosis
I. Arpan, P.C. Fino, B.W. Fling, F. Horak
Gait & Posture.2020; 76: 122. CrossRef - Instrumental assessment of balance and gait in depression: A systematic review
Martino Belvederi Murri, Federico Triolo, Alice Coni, Carlo Tacconi, Erika Nerozzi, Andrea Escelsior, Matteo Respino, Francesca Neviani, Marco Bertolotti, Klea Bertakis, Lorenzo Chiari, Stamatula Zanetidou, Mario Amore
Psychiatry Research.2020; 284: 112687. CrossRef - Baropodometric evaluation of foot load distribution during gait in the group of professionally active nurses
Anna Kołcz, Natalia Główka, Mateusz Kowal, Małgorzata Paprocka-Borowicz
Journal of Occupational Health.2020;[Epub] CrossRef - Effect of aging on H-reflex response to fatigue
Andrew Philip Lavender, Sadik Balkozak, Mustafa Görkem Özyurt, Betilay Topkara, İlhan Karacan, İdil Bilici, Anne-Marie Hill, Kemal Sitki Türker
Experimental Brain Research.2020; 238(2): 273. CrossRef - The repeatability of the instrumented timed Up & Go test: The performance of older adults and parkinson’s disease patients under different conditions
Slavka Viteckova, Radim Krupicka, Petr Dusek, Vaclav Cejka, Patrik Kutilek, Jan Novak, Zoltan Szabo, Evžen Růžička
Biocybernetics and Biomedical Engineering.2020; 40(1): 363. CrossRef - Cerebral Cortical Activity Following Non-invasive Cerebellar Stimulation—a Systematic Review of Combined TMS and EEG Studies
Lara Fernandez, Nigel C. Rogasch, Michael Do, Gillian Clark, Brendan P. Major, Wei-Peng Teo, Linda K. Byrne, Peter G. Enticott
The Cerebellum.2020; 19(2): 309. CrossRef - Resting-state fMRI reveals increased functional connectivity in the cerebellum but decreased functional connectivity of the caudate nucleus in Parkinson’s disease
Oliver Kaut, Clemens Mielacher, René Hurlemann, Ullrich Wüllner
Neurological Research.2020; 42(1): 62. CrossRef - Gait Generation and Its Energy Efficiency Based on Rat Neuromusculoskeletal Model
Misaki Toeda, Shinya Aoi, Soichiro Fujiki, Tetsuro Funato, Kazuo Tsuchiya, Dai Yanagihara
Frontiers in Neuroscience.2020;[Epub] CrossRef - Cognitive and physical markers of prodromal dementia: A 12‐year‐long population study
Giulia Grande, Debora Rizzuto, Davide L. Vetrano, Anna Marseglia, Nicola Vanacore, Erika J. Laukka, Anna‐Karin Welmer, Laura Fratiglioni
Alzheimer's & Dementia.2020; 16(1): 153. CrossRef - The sensory role of the sole of the foot: Review and update on clinical perspectives
Frederic J.F. Viseux
Neurophysiologie Clinique.2020; 50(1): 55. CrossRef - Overactivity in Chronic Pain, the Role of Pain-related Endurance and Neuromuscular Activity
Monika I. Hasenbring, Nicole E. Andrews, Gerold Ebenbichler
The Clinical Journal of Pain.2020; 36(3): 162. CrossRef - Motor tract integrity predicts walking recovery
Julie Soulard, Coline Huber, Sebastien Baillieul, Antoine Thuriot, Felix Renard, Bérengère Aubert Broche, Alexandre Krainik, Nicolas Vuillerme, Assia Jaillard
Neurology.2020;[Epub] CrossRef - Altered intra- and inter-network functional connectivity in patients with persistent postural-perceptual dizziness
Kangzhi Li, Lihong Si, Bin Cui, Xia Ling, Bo Shen, Xu Yang
NeuroImage: Clinical.2020; 26: 102216. CrossRef - The role of physical exercise and rehabilitation in delirium
N. Gual, M. García-Salmones, L. Brítez, N. Crespo, C. Udina, L. M. Pérez, M. Inzitari
European Geriatric Medicine.2020; 11(1): 83. CrossRef - Neural circuits of idiopathic Normal Pressure Hydrocephalus: A perspective review of brain connectivity and symptoms meta-analysis
Alessandra Griffa, Dimitri Van De Ville, François R. Herrmann, Gilles Allali
Neuroscience & Biobehavioral Reviews.2020; 112: 452. CrossRef - Orbitrap mass spectrometry for monitoring the ganglioside pattern in human cerebellum development and aging
Raluca Ica, Alina Petrut, Cristian V.A. Munteanu, Mirela Sarbu, Željka Vukelić, Ligia Petrica, Alina D. Zamfir
Journal of Mass Spectrometry.2020;[Epub] CrossRef - What are the association patterns between handgrip strength and adverse health conditions? A topical review
Ryan McGrath, Nathaniel Johnson, Lukus Klawitter, Sean Mahoney, Kara Trautman, Caroline Carlson, Ella Rockstad, Kyle J Hackney
SAGE Open Medicine.2020; 8: 205031212091035. CrossRef - Cortical Oscillations during Gait: Wouldn’t Walking Be So Automatic?
Arnaud Delval, Madli Bayot, Luc Defebvre, Kathy Dujardin
Brain Sciences.2020; 10(2): 90. CrossRef - Loss of presynaptic inhibition for step initiation in parkinsonian individuals with freezing of gait
Jumes Leopoldino Oliveira Lira, Carlos Ugrinowitsch, Daniel Boari Coelho, Luis Augusto Teixeira, Andrea Cristina de Lima‐Pardini, Fernando Henrique Magalhães, Egberto Reis Barbosa, Fay B. Horak, Carla Silva‐Batista
The Journal of Physiology.2020; 598(8): 1611. CrossRef - Which Effects on Neuroanatomy and Path-Integration Survive? Results of a Randomized Controlled Study on Intensive Balance Training
Milos Dordevic, Marco Taubert, Patrick Müller, Martin Riemer, Jörn Kaufmann, Anita Hökelmann, Notger G. Müller
Brain Sciences.2020; 10(4): 210. CrossRef - Freezing of gait in Parkinson’s disease: pathophysiology, risk factors and treatments
Chao Gao, Jun Liu, Yuyan Tan, Shengdi Chen
Translational Neurodegeneration.2020;[Epub] CrossRef - Clinical experience of comprehensive treatment on the balance function of Parkinson's disease
Haitao Li, Siquan Liang, Yang Yu, Yue Wang, Yuanyuan Cheng, Hechao Yang, Xiaoguang Tong
Medicine.2020; 99(19): e20154. CrossRef - Digital natives and dual task: Handling it but not immune against cognitive-locomotor interferences
Frédéric Dierick, Fabien Buisseret, Mathieu Renson, Adèle Mae Luta, Slavko Rogan
PLOS ONE.2020; 15(5): e0232328. CrossRef - Functional neuroimaging of human postural control: A systematic review with meta-analysis
Bauke W. Dijkstra, Esther M.J. Bekkers, Moran Gilat, Veerle de Rond, Robert M. Hardwick, Alice Nieuwboer
Neuroscience & Biobehavioral Reviews.2020; 115: 351. CrossRef - The neural correlates of falls: Alterations in large-scale resting-state networks in elderly fallers
Inbal Maidan, Amgad Droby, Yael Jacob, Nir Giladi, Jeffrey M Hausdorff, Anat Mirelman
Gait & Posture.2020; 80: 56. CrossRef - Short rehabilitation training program may improve postural control in children with autism spectrum disorders: preliminary evidences
Simona Caldani, Paola Atzori, Hugo Peyre, Richard Delorme, Maria Pia Bucci
Scientific Reports.2020;[Epub] CrossRef - Effects of injuries to descending motor pathways on restoration of gait in patients with pontine hemorrhage
Sang Seok Yeo, Sung Ho Jang, Ga Young Park, Seunghue Oh
Journal of Stroke and Cerebrovascular Diseases.2020; 29(7): 104857. CrossRef - Handgrip strength accuracy as discriminator of functional independence in centenarian women
Artur Rodrigues Fortunato, Raquel Ester Lima da Silva, Giovana Zarpellon Mazo
Revista Brasileira de Cineantropometria & Desempenho Humano.2020;[Epub] CrossRef - Assessing the effect of current steering on the total electrical energy delivered and ambulation in Parkinson’s disease
Daphne Hui, Aditya A. Murgai, Greydon Gilmore, Shabna I. Mohideen, Andrew G. Parrent, Mandar S. Jog
Scientific Reports.2020;[Epub] CrossRef - Speech disorders and anxiety: interaction mechanisms and therapy potential
E.E. Vasenina, O.S. Levin
Zhurnal nevrologii i psikhiatrii im. S.S. Korsakova.2020; 120(4): 136. CrossRef - Progression of motor disability in cerebral palsy: The role of concomitant epilepsy
Tomoyuki Takano, Anri Hayashi, Yuki Harada
Seizure.2020; 80: 81. CrossRef - Turning Characteristics of the More-Affected Side in Parkinson’s Disease Patients with Freezing of Gait
Hwayoung Park, Changhong Youm, Myeounggon Lee, Byungjoo Noh, Sang-Myung Cheon
Sensors.2020; 20(11): 3098. CrossRef - Fifteen Years of Wireless Sensors for Balance Assessment in Neurological Disorders
Alessandro Zampogna, Ilaria Mileti, Eduardo Palermo, Claudia Celletti, Marco Paoloni, Alessandro Manoni, Ivan Mazzetta, Gloria Dalla Costa, Carlos Pérez-López, Filippo Camerota, Letizia Leocani, Joan Cabestany, Fernanda Irrera, Antonio Suppa
Sensors.2020; 20(11): 3247. CrossRef - Muscle Synergies in Parkinson’s Disease
Ilaria Mileti, Alessandro Zampogna, Alessandro Santuz, Francesco Asci, Zaccaria Del Prete, Adamantios Arampatzis, Eduardo Palermo, Antonio Suppa
Sensors.2020; 20(11): 3209. CrossRef - Transcranial direct current stimulation combined with physical or cognitive training in people with Parkinson’s disease: a systematic review
Victor Spiandor Beretta, Núbia Ribeiro Conceição, Priscila Nóbrega-Sousa, Diego Orcioli-Silva, Luana Karla Braz Fonseca Dantas, Lilian Teresa Bucken Gobbi, Rodrigo Vitório
Journal of NeuroEngineering and Rehabilitation.2020;[Epub] CrossRef - Assessing the Temporal Organization of Walking Variability: A Systematic Review and Consensus Guidelines on Detrended Fluctuation Analysis
Deepak K. Ravi, Vivien Marmelat, William R. Taylor, Karl M. Newell, Nick Stergiou, Navrag B. Singh
Frontiers in Physiology.2020;[Epub] CrossRef - Backward locomotor treadmill training combined with transcutaneous spinal direct current stimulation in stroke: a randomized pilot feasibility and safety study
Oluwole O Awosika, Saira Matthews, Emily J Staggs, Pierce Boyne, Xiao Song, Bridget A Rizik, Heidi J Sucharew, Christina Zhang, Gabrielle Mungcal, Rohitha Moudgal, Amit Bhattacharya, Kari Dunning, Daniel Woo, Brett M Kissela
Brain Communications.2020;[Epub] CrossRef - Behavioural manifestations and associated non-motor features of freezing of gait: A narrative review and theoretical framework
Kaylena A. Ehgoetz Martens, Daniel S. Peterson, Quincy J. Almeida, Simon J.G. Lewis, Jeffrey M. Hausdorff, Alice Nieuwboer
Neuroscience & Biobehavioral Reviews.2020; 116: 350. CrossRef - New insight into Parkinson’s disease‐related impairment of the automatic control of upright stance
Cédrick T. Bonnet, Arnaud Delval, Tarkeshwar Singh, Yann‐Romain Kechabia, Luc Defebvre
European Journal of Neuroscience.2020; 52(12): 4851. CrossRef - Firing activity of locus coeruleus noradrenergic neurons decreases in necdin-deficient mice, an animal model of Prader–Willi syndrome
Rui-Ni Wu, Wei-Chen Hung, Ching-Tsuey Chen, Li-Ping Tsai, Wen-Sung Lai, Ming-Yuan Min, Shi-Bing Wong
Journal of Neurodevelopmental Disorders.2020;[Epub] CrossRef - A functional observational battery for evaluation of neurological outcomes in a rat model of acute bacterial meningitis
Jane Fisher, Chiara Pavan, Luisa S. Ohlmeier, Bo Nilson, Iben Lundgaard, Adam Linder, Peter Bentzer
Intensive Care Medicine Experimental.2020;[Epub] CrossRef - Impact of Extremity Manipulation on Postural Sway Characteristics: A Preliminary, Randomized Crossover Study
Christopher A. Malaya, Joshua Haworth, Katherine A. Pohlman, Cody Powell, Dean L. Smith
Journal of Manipulative and Physiological Therapeutics.2020; 43(5): 457. CrossRef - Dissociated motor learning and de-adaptation in patients with functional gait disorders
Denise Lin, Patricia Castro, Amy Edwards, Akila Sekar, Mark J Edwards, Jan Coebergh, Adolfo M Bronstein, Diego Kaski
Brain.2020; 143(8): 2594. CrossRef - Multicenter Validation of Metabolic Abnormalities Related to PSP According to the MDS‐PSP Criteria
Gloria Martí‐Andrés, Liza van Bommel, Sanne K. Meles, Mario Riverol, Rafael Valentí, Rosalie V. Kogan, Remco J. Renken, Vita Gurvits, Teus van Laar, Marco Pagani, Elena Prieto, M. Rosario Luquin, Klaus L. Leenders, Javier Arbizu
Movement Disorders.2020; 35(11): 2009. CrossRef - Gait initiation in progressive supranuclear palsy: brain metabolic correlates
Chiara Palmisano, Massimiliano Todisco, Giorgio Marotta, Jens Volkmann, Claudio Pacchetti, Carlo A. Frigo, Gianni Pezzoli, Ioannis U. Isaias
NeuroImage: Clinical.2020; 28: 102408. CrossRef - Altered Brain Function in Cerebral Small Vessel Disease Patients With Gait Disorders: A Resting-State Functional MRI Study
Xia Zhou, Chao Zhang, Linlin Li, Yimei Zhang, Wei Zhang, Wenwen Yin, Xianfeng Yu, Xiaoqun Zhu, Yinfeng Qian, Zhongwu Sun
Frontiers in Aging Neuroscience.2020;[Epub] CrossRef - Lower Thalamic Blood Flow Is Associated With Slower Stride Velocity in Older Adults
Noah D. Koblinsky, Sarah Atwi, Ellen Cohen, Nicole D. Anderson, Carol E. Greenwood, Bradley J. MacIntosh, Andrew D. Robertson
Frontiers in Aging Neuroscience.2020;[Epub] CrossRef - Gait-related frequency modulation of beta oscillatory activity in the subthalamic nucleus of parkinsonian patients
Andrea Canessa, Chiara Palmisano, Ioannis U. Isaias, Alberto Mazzoni
Brain Stimulation.2020;[Epub] CrossRef - Immersive Virtual Reality to Restore Natural Long-Range Autocorrelations in Parkinson’s Disease Patients’ Gait During Treadmill Walking
Alexis Lheureux, Julien Lebleu, Caroline Frisque, Corentin Sion, Gaëtan Stoquart, Thibault Warlop, Christine Detrembleur, Thierry Lejeune
Frontiers in Physiology.2020;[Epub] CrossRef - Non-pharmacological Treatment Challenges in Early Parkinson's Disease for Axial and Cognitive Symptoms: A Mini Review
Gabriella Sharpe, Antonella Macerollo, Margherita Fabbri, Elina Tripoliti
Frontiers in Neurology.2020;[Epub] CrossRef - Genetic basis of falling risk susceptibility in the UK Biobank Study
Katerina Trajanoska, Lotta J. Seppala, Carolina Medina-Gomez, Yi-Hsiang Hsu, Sirui Zhou, Natasja M. van Schoor, Lisette C. P. G. M. de Groot, David Karasik, J. Brent Richards, Douglas P. Kiel, Andre G. Uitterlinden, John R. B. Perry, Nathalie van der Veld
Communications Biology.2020;[Epub] CrossRef - Effect of Different Intensities of Transcranial Direct Current Stimulation on Postural Response to External Perturbation in Patients With Parkinson’s Disease
Victor Spiandor Beretta, Rodrigo Vitório, Priscila Nóbrega-Sousa, Núbia Ribeiro Conceição, Diego Orcioli-Silva, Marcelo Pinto Pereira, Lilian Teresa Bucken Gobbi
Neurorehabilitation and Neural Repair.2020; 34(11): 1009. CrossRef - Adapting to the Mechanical Properties and Active Force of an Exoskeleton by Altering Muscle Synergies in Chronic Stroke Survivors
Linda Rinaldi, Ling-Fung Yeung, Patrick Chi-Hong Lam, Marco Y. C. Pang, Raymond Kai-Yu Tong, Vincent C. K. Cheung
IEEE Transactions on Neural Systems and Rehabilitation Engineering.2020; 28(10): 2203. CrossRef - Brain networks associated with anticipatory postural adjustments in Parkinson’s disease patients with freezing of gait
Andrea C. de Lima-Pardini, Daniel B. Coelho, Mariana P. Nucci, Catarina C. Boffino, Alana X. Batista, Raymundo M. de Azevedo Neto, Carla Silva-Batista, Egberto R. Barbosa, Rajal G. Cohen, Fay B. Horak, Luis A. Teixeira, Edson Amaro Jr
NeuroImage: Clinical.2020; 28: 102461. CrossRef - Rhythmic neural activity is comodulated with short-term gait modifications during first-time use of a dummy prosthesis: a pilot study
Vera G. M. Kooiman, Helco G. van Keeken, Natasha M. Maurits, Vivian Weerdesteyn, Teodoro Solis-Escalante
Journal of NeuroEngineering and Rehabilitation.2020;[Epub] CrossRef - Deep brain stimulation: a review of the open neural engineering challenges
Matteo Vissani, Ioannis U Isaias, Alberto Mazzoni
Journal of Neural Engineering.2020; 17(5): 051002. CrossRef - History of and Insights Into Spinal Cord Stimulation in Parkinson Disease
Bruno Braz Garcia, Edson Ricardo Junior, Mariana Ferreira Pereira de Araújo, Hougelle Simplício
Neurorehabilitation and Neural Repair.2020; 34(11): 967. CrossRef - Influence of Robotic Mechanotherapy in Various Combinations with Non-Invasive Electrostimulation of Muscles and Spinal Cord on the Postural Balance in Children with Severe Forms of Cerebral Palsy
I.E. Nikityuk, E.L. Kononova, G.A. Ikoeva, I.Yu Solokhina
Bulletin of Restorative Medicine.2020; 98(4): 26. CrossRef - Toward a Regulatory Qualification of Real-World Mobility Performance Biomarkers in Parkinson’s Patients Using Digital Mobility Outcomes
Marco Viceconti, Sabina Hernandez Penna, Wilhelmus Dartee, Claudia Mazzà, Brian Caulfield, Clemens Becker, Walter Maetzler, Judith Garcia-Aymerich, Giorgio Davico, Lynn Rochester
Sensors.2020; 20(20): 5920. CrossRef - Brain Network Oscillations During Gait in Parkinson’s Disease
Doris D. Wang, Julia T. Choi
Frontiers in Human Neuroscience.2020;[Epub] CrossRef - Disrupted white matter integrity and network connectivity are related to poor motor performance
Feifei Zhai, Jie Liu, Ning Su, Fei Han, Lixin Zhou, Jun Ni, Ming Yao, Shuyang Zhang, Zhengyu Jin, Liying Cui, Feng Tian, Yicheng Zhu
Scientific Reports.2020;[Epub] CrossRef - Serotonergic Modulation of Locomotor Activity From Basal Vertebrates to Mammals
Aurélie Flaive, Maxime Fougère, Cornelis Immanuel van der Zouwen, Dimitri Ryczko
Frontiers in Neural Circuits.2020;[Epub] CrossRef - Classification of Rhythmic Cortical Activity Elicited by Whole-Body Balance Perturbations Suggests the Cortical Representation of Direction-Specific Changes in Postural Stability
Teodoro Solis-Escalante, Digna De Kam, Vivian Weerdesteyn
IEEE Transactions on Neural Systems and Rehabilitation Engineering.2020; 28(11): 2566. CrossRef - Executive Control of Walking in People With Parkinson’s Disease With Freezing of Gait
Rodrigo Vitorio, Samuel Stuart, Martina Mancini
Neurorehabilitation and Neural Repair.2020; 34(12): 1138. CrossRef - Postural instability in Parkinson’s disease: Review and bottom-up rehabilitative approaches
Frederic J.F. Viseux, Arnaud Delval, Luc Defebvre, Martin Simoneau
Neurophysiologie Clinique.2020; 50(6): 479. CrossRef - Can dual-task paradigms predict Falls better than single task? – A systematic literature review
Madli Bayot, Kathy Dujardin, Lucile Dissaux, Céline Tard, Luc Defebvre, Cédrick T. Bonnet, Etienne Allart, Gilles Allali, Arnaud Delval
Neurophysiologie Clinique.2020; 50(6): 401. CrossRef - Neural Correlates of Freezing of Gait in Parkinson's Disease: An Electrophysiology Mini-Review
J. Sebastian Marquez, S. M. Shafiul Hasan, Masudur R. Siddiquee, Corneliu C. Luca, Virendra R. Mishra, Zoltan Mari, Ou Bai
Frontiers in Neurology.2020;[Epub] CrossRef - Association of Postural Instability with Autonomic Dysfunction in Early Parkinson’s Disease
Sooyeoun You, Hyun Ah Kim, Hyung Lee
Journal of Clinical Medicine.2020; 9(11): 3786. CrossRef - Neural Mechanisms for Locomotor and Postural Control in Man and Its Application to Neurorehabilitation
Ichiro Miyai
The Japanese Journal of Rehabilitation Medicine.2020; 57(10): 965. CrossRef - A case of generalized tonic seizures related to acute myocarditis
Jeong-Hwan Yang, Jae Yoon Na, Jinseok Lee, Da-Eun Kim, Jinyoung Song, I-Seok Kang, Hyeryun Yeh
Pediatric Emergency Medicine Journal.2020; 7(2): 127. CrossRef - Rapid and robust patterns of spontaneous locomotor deficits in mouse models of Huntington’s disease
Taneli Heikkinen, Timo Bragge, Niina Bhattarai, Teija Parkkari, Jukka Puoliväli, Outi Kontkanen, Patrick Sweeney, Larry C. Park, Ignacio Munoz-Sanjuan, Yuqing Li
PLOS ONE.2020; 15(12): e0243052. CrossRef - The Relationship of Rapid Eye Movement Sleep Behavior Disorder and Freezing of Gait in Parkinson’s Disease
Chelsea Mae N Nobleza, Mariah Siddiqui, Parth V Shah, Prachi Balani, Angel R Lopez, Safeera Khan
Cureus.2020;[Epub] CrossRef - Influence of Autocorrelated Rhythmic Auditory Stimulations on Parkinson’s Disease Gait Variability: Comparison With Other Auditory Rhythm Variabilities and Perspectives
Alexis Lheureux, Thibault Warlop, Charline Cambier, Baptiste Chemin, Gaëtan Stoquart, Christine Detrembleur, Thierry Lejeune
Frontiers in Physiology.2020;[Epub] CrossRef - A Roadmap to Inform Development, Validation and Approval of Digital Mobility Outcomes: The Mobilise-D Approach
Lynn Rochester, Claudia Mazzà, Arne Mueller, Brian Caulfield, Marie McCarthy, Clemens Becker, Ram Miller, Paolo Piraino, Marco Viceconti, Wilhelmus P. Dartee, Judith Garcia-Aymerich, Aida A. Aydemir, Beatrix Vereijken, Valdo Arnera, Nadir Ammour, Michael
Digital Biomarkers.2020; 4(Suppl. 1): 13. CrossRef - Cerebellar contribution to locomotor behavior: A neurodevelopmental perspective
Aaron Sathyanesan, Vittorio Gallo
Neurobiology of Learning and Memory.2019; 165: 106861. CrossRef - A meta-analysis: Parkinson's disease and dual-task walking
Tiphanie E. Raffegeau, Lisa M. Krehbiel, Nyeonju Kang, Frency J. Thijs, Lori J.P. Altmann, James H. Cauraugh, Chris J. Hass
Parkinsonism & Related Disorders.2019; 62: 28. CrossRef - Treadmill exercise intervention improves gait and postural control in alpha-synuclein mouse models without inducing cerebral autophagy
Georgia Minakaki, Fabio Canneva, Frédéric Chevessier, Frederik Bode, Stefanie Menges, Ivanna K. Timotius, Liubov S. Kalinichenko, Holger Meixner, Christian P. Müller, Bjoern M. Eskofier, Nicolas Casadei, Olaf Riess, Rolf Schröder, Jürgen Winkler, Wei Xian
Behavioural Brain Research.2019; 363: 199. CrossRef - Acupuncture Enhances Communication between Cortices with Damaged White Matters in Poststroke Motor Impairment
Xiao Han, Lijun Bai, Chuanzhu Sun, Xuan Niu, Yanzhe Ning, Zhen Chen, Yingying Li, Kuangshi Li, Diyang Lyu, Caihong Fu, Fangyuan Cui, Zhengguang Chen, Zhongjian Tan, Lixin Tang, Yihuai Zou
Evidence-Based Complementary and Alternative Medicine.2019; 2019: 1. CrossRef - Improvement of gait and balance by non-invasive brain stimulation: its use in rehabilitation
Soumya Ghosh
Expert Review of Neurotherapeutics.2019; 19(2): 133. CrossRef - Neural Substrates of Cognitive Motor Interference During Walking; Peripheral and Central Mechanisms
Emad Al-Yahya, Wala’ Mahmoud, Daan Meester, Patrick Esser, Helen Dawes
Frontiers in Human Neuroscience.2019;[Epub] CrossRef - Efficiency of Sensorimotor Networks: Posture and Gait in Young and Older Adults
G. Di Scala, M. Dupuy, E. Guillaud, E. Doat, E. Barse, B. Dillhareguy, F.A.M. Jean, M. Audiffren, J.R. Cazalets, S. Chanraud
Experimental Aging Research.2019; 45(1): 41. CrossRef - Central pattern generators in the brainstem and spinal cord: an overview of basic principles, similarities and differences
Inge Steuer, Pierre A. Guertin
Reviews in the Neurosciences.2019; 30(2): 107. CrossRef - The Effects of Dual‐Task Cognitive Interference and Environmental Challenges on Balance in Huntington's Disease
Nicollette L. Purcell, Jennifer G. Goldman, Bichun Ouyang, Bryan Bernard, Joan A. O'Keefe
Movement Disorders Clinical Practice.2019; 6(3): 202. CrossRef - Descending dopaminergic control of brainstem locomotor circuits
Maxime Fougère, Aurélie Flaive, Alain Frigon, Dimitri Ryczko
Current Opinion in Physiology.2019; 8: 30. CrossRef - Visual Fixation and Continuous Head Rotations Have Minimal Effect on Set-Point Adaptation to Magnetic Vestibular Stimulation
Bryan K. Ward, David S. Zee, Dale C. Roberts, Michael C. Schubert, Nicolas Pérez-Fernández, Jorge Otero-Millan
Frontiers in Neurology.2019;[Epub] CrossRef - Computational modeling of brainstem circuits controlling locomotor frequency and gait
Jessica Ausborn, Natalia A Shevtsova, Vittorio Caggiano, Simon M Danner, Ilya A Rybak
eLife.2019;[Epub] CrossRef - Structure and function of the mesencephalic locomotor region in normal and parkinsonian primates
Brian Lau, Chantal François, Carine Karachi
Current Opinion in Physiology.2019; 8: 121. CrossRef - Ambulation Patterns Post-Discharge in Older Adults Identified as Fall Risk: A Descriptive Pilot Study
Barbara J. King, Roger Brown, Linsey Steege, Haocen Wang, Fang-lin Kuo, Cynthia Brown
Research in Gerontological Nursing.2019; 12(3): 113. CrossRef - Quantification of trunk segmental coordination and head stability in laterally unstable sitting identifies aging and cerebellar ataxia
Koshiro Haruyama, Kenji Kasai, Ryohei Makino, Fumihiko Hoshi, Ken Nishihara
Clinical Biomechanics.2019; 63: 127. CrossRef - Deterioration of Brain Neural Tracts in Elderly Women with Sarcopenia
So Young Kwak, Sang Gyu Kwak, Tae Sang Yoon, Eun Jung Kong, Min Cheol Chang
The American Journal of Geriatric Psychiatry.2019; 27(8): 774. CrossRef - “Special K” Drug on Adolescent Rats: Oxidative Damage and Neurobehavioral Impairments
Sabrina de Carvalho Cartágenes, Luanna Melo Pereira Fernandes, Taiana Cristina Vilhena Sarmento Carvalheiro, Thais Miranda de Sousa, Antônio Rafael Quadros Gomes, Marta Chagas Monteiro, Ricardo Sousa de Oliveira Paraense, Maria Elena Crespo-López, Rafael
Oxidative Medicine and Cellular Longevity.2019; 2019: 1. CrossRef - Current models of speech motor control: A control-theoretic overview of architectures and properties
Benjamin Parrell, Adam C. Lammert, Gregory Ciccarelli, Thomas F. Quatieri
The Journal of the Acoustical Society of America.2019; 145(3): 1456. CrossRef - Falls and Fractures in Diabetes—More than Bone Fragility
Nicklas Højgaard Rasmussen, Jakob Dal
Current Osteoporosis Reports.2019; 17(3): 147. CrossRef - Expression of aggressiveness modulates mesencephalic c-fos activation during a social interaction test in Japanese quail (Coturnix japonica)
Jorge Martin Caliva, Fernando Falkenburger Melleu, Jose Marino-Neto, Raul Hector Marin, Jackelyn Melissa Kembro
Behavioural Brain Research.2019; 367: 221. CrossRef - Positive verbal suggestion optimizes postural control
Bernardo Villa-Sánchez, Mehran Emadi Andani, Giulia Menegaldo, Michele Tinazzi, Mirta Fiorio
Scientific Reports.2019;[Epub] CrossRef - Neuromodulation to improve gait and balance function using a sensory neuroprosthesis in people who report insensate feet – A randomized control cross-over study
Sara R. Koehler-McNicholas, Lori Danzl, Alana Y. Cataldo, Lars I. E. Oddsson, Alfonso Fasano
PLOS ONE.2019; 14(4): e0216212. CrossRef - Selective suppression of the vestibulo-ocular reflex during human locomotion
Haike Dietrich, Max Wuehr
Journal of Neurology.2019; 266(S1): 101. CrossRef - Gait stride-to-stride variability and foot clearance pattern analysis in Idiopathic Parkinson’s Disease and Vascular Parkinsonism
Flora Ferreira, Miguel F. Gago, Estela Bicho, Catarina Carvalho, Nafiseh Mollaei, Lurdes Rodrigues, Nuno Sousa, Pedro Pereira Rodrigues, Carlos Ferreira, João Gama
Journal of Biomechanics.2019; 92: 98. CrossRef - Von der Biomechanik zur Bioinformatik
Philip Eckardt
Osteopathische Medizin.2019; 20(2): 27. CrossRef - Measuring gait speed to better identify prodromal dementia
Giulia Grande, Federico Triolo, Arturo Nuara, Anna-Karin Welmer, Laura Fratiglioni, Davide L. Vetrano
Experimental Gerontology.2019; 124: 110625. CrossRef - Effects of transcranial direct current stimulation (tDCS) on balance improvement: a systematic review and meta-analysis
Maria Clara D. Soares de Moura, Fuad A. Hazime, Luana V. Marotti Aparicio, Luanda A. C. Grecco, André R. Brunoni, Renata Hydeé Hasue
Somatosensory & Motor Research.2019; 36(2): 122. CrossRef - Long-Term Effects of Whole-Body Vibration on Human Gait: A Systematic Review and Meta-Analysis
Matthieu Fischer, Thomas Vialleron, Guillaume Laffaye, Paul Fourcade, Tarek Hussein, Laurence Chèze, Paul-André Deleu, Jean-Louis Honeine, Eric Yiou, Arnaud Delafontaine
Frontiers in Neurology.2019;[Epub] CrossRef - Effective Parameters for Gait Analysis in Experimental Models for Evaluating Peripheral Nerve Injuries in Rats
Ivair Matias Júnior, Priscila Medeiros, Renato Leonardo de Freita, Hilton Vicente-César, José Raniery Ferreira Junior, Hélio Rubens Machado, Rafael Menezes-Reis
Neurospine.2019; 16(2): 305. CrossRef - Freezing of gait in Parkinson’s disease reflects a sudden derangement of locomotor network dynamics
Nicoló G Pozzi, Andrea Canessa, Chiara Palmisano, Joachim Brumberg, Frank Steigerwald, Martin M Reich, Brigida Minafra, Claudio Pacchetti, Gianni Pezzoli, Jens Volkmann, Ioannis U Isaias
Brain.2019; 142(7): 2037. CrossRef - Usual walking and obstacle avoidance are influenced by depressive and anxiety symptoms in patients with Parkinson's disease
Priscila Nóbrega‐Sousa, Diego Orcioli‐Silva, Ellen Lirani‐Silva, Victor Spiandor Beretta, Rodrigo Vitório, Lilian Teresa Bucken Gobbi
Geriatrics & Gerontology International.2019; 19(9): 868. CrossRef - Complexity based measures of postural stability provide novel evidence of functional decline in fragile X premutation carriers
Clodagh O’Keeffe, Laura P. Taboada, Niamh Feerick, Louise Gallagher, Timothy Lynch, Richard B. Reilly
Journal of NeuroEngineering and Rehabilitation.2019;[Epub] CrossRef - Motor-Cognitive Neural Network Communication Underlies Walking Speed in Community-Dwelling Older Adults
Victoria N. Poole, On-Yee Lo, Thomas Wooten, Ikechukwu Iloputaife, Lewis A. Lipsitz, Michael Esterman
Frontiers in Aging Neuroscience.2019;[Epub] CrossRef - Programmable coupled oscillators for synchronized locomotion
Sourav Dutta, Abhinav Parihar, Abhishek Khanna, Jorge Gomez, Wriddhi Chakraborty, Matthew Jerry, Benjamin Grisafe, Arijit Raychowdhury, Suman Datta
Nature Communications.2019;[Epub] CrossRef - Immediate effects of rhythmic auditory stimulation on gait kinematics in Parkinson’s disease ON/OFF medication
Carmen Erra, Ilaria Mileti, Marco Germanotta, Martina Petracca, Isabella Imbimbo, Alessandro De Biase, Stefano Rossi, Diego Ricciardi, Alessandra Pacilli, Enrica Di Sipio, Eduardo Palermo, Anna Rita Bentivoglio, Luca Padua
Clinical Neurophysiology.2019; 130(10): 1789. CrossRef - Patterns of co‐altered brain structure and function underlying neurological soft signs in schizophrenia spectrum disorders
Dusan Hirjak, Mahmoud Rashidi, Stefan Fritze, Alina L. Bertolino, Lena S. Geiger, Zhenxiang Zang, Katharina M. Kubera, Mike M. Schmitgen, Fabio Sambataro, Vince D. Calhoun, Matthias Weisbrod, Heike Tost, Robert C. Wolf
Human Brain Mapping.2019; 40(17): 5029. CrossRef - Spinal Cord Stimulation for Freezing of Gait: From Bench to Bedside
Erich Talamoni Fonoff, Andrea C. de Lima-Pardini, Daniel Boari Coelho, Bernardo Assumpção Monaco, Birajara Machado, Carolina Pinto de Souza, Maria Gabriela dos Santos Ghilardi, Clement Hamani
Frontiers in Neurology.2019;[Epub] CrossRef - Head Anticipation During Locomotion With Auditory Instruction in the Presence and Absence of Visual Input
Felix Dollack, Monica Perusquía-Hernández, Hideki Kadone, Kenji Suzuki
Frontiers in Human Neuroscience.2019;[Epub] CrossRef - Neuroplasticity Modifications Following a Lower‐Limb Amputation: A Systematic Review
Francisco Molina‐Rueda, Cristian Navarro‐Fernández, Alicia Cuesta‐Gómez, Isabel M Alguacil‐Diego, Alberto Molero‐Sánchez, María Carratalá‐Tejada
PM&R.2019; 11(12): 1326. CrossRef - Correlation between selected gait variables and emotion using virtual reality
Young Kim, JunYoung Moon, Nak-Jun Sung, Min Hong
Journal of Ambient Intelligence and Humanized Computing.2019;[Epub] CrossRef - Neural substrates of reduced walking activity after supratentorial stroke: A voxel-based lesion symptom mapping study
Sébastien Baillieul, Charlotte Elsworth-Edelsten, Arnaud Saj, Gilles Allali
Human Movement Science.2019; 67: 102517. CrossRef - Olfactory Stimulation with Japanese Soy Sauce Improves Upper Limb Performance
Yutaka Yano, Yoshihiro Murata, Mutsuo Taniguchi, Fumino Okutani, Masahiro Yamaguchi, Hideto Kaba
Occupational Therapy International.2019; 2019: 1. CrossRef - Botulinum toxin A injection into the entopeduncular nucleus improves dynamic locomotory parameters in hemiparkinsonian rats
Adrianna R. Tsang, Nagalingam Rajakumar, Mandar S. Jog, David I. Finkelstein
PLOS ONE.2019; 14(10): e0223450. CrossRef - Functional and Brain Activation Changes Following Specialized Upper-Limb Exercise in Parkinson’s Disease
Luca Valerio Messa, Federica Ginanneschi, Davide Momi, Lucia Monti, Carla Battisti, David Cioncoloni, Barbara Pucci, Emiliano Santarnecchi, Alessandro Rossi
Frontiers in Human Neuroscience.2019;[Epub] CrossRef - Neuroimaging advances in Parkinson's disease with freezing of gait: A systematic review
Komal Bharti, Antonio Suppa, Silvia Tommasin, Alessandro Zampogna, Sara Pietracupa, Alfredo Berardelli, Patrizia Pantano
NeuroImage: Clinical.2019; 24: 102059. CrossRef - Effects of supraspinal feedback on human gait: rhythmic auditory distortion
Arturo Forner-Cordero, João Pedro Pinho, Guilherme Umemura, João Carlos Lourenço, Bruno Mezêncio, Cinthia Itiki, Hermano Igo Krebs
Journal of NeuroEngineering and Rehabilitation.2019;[Epub] CrossRef - Cortical disconnection of the ipsilesional primary motor cortex is associated with gait speed and upper extremity motor impairment in chronic left hemispheric stroke
Denise M. Peters, Julius Fridriksson, Jill C. Stewart, Jessica D. Richardson, Chris Rorden, Leonardo Bonilha, Addie Middleton, Ezequiel Gleichgerrcht, Stacy L. Fritz
Human Brain Mapping.2018; 39(1): 120. CrossRef - Effects of transcranial direct current stimulation over the supplementary motor area body weight-supported treadmill gait training in hemiparetic patients after stroke
Atsushi Manji, Kazu Amimoto, Tadamitsu Matsuda, Yoshiaki Wada, Akira Inaba, Sangkyun Ko
Neuroscience Letters.2018; 662: 302. CrossRef - Gender-related differences in neural responses to gaming cues before and after gaming: implications for gender-specific vulnerabilities to Internet gaming disorder
Guangheng Dong, Lingxiao Wang, Xiaoxia Du, Marc N Potenza
Social Cognitive and Affective Neuroscience.2018; 13(11): 1203. CrossRef - Stem Cell Transplantation and Physical Exercise in Parkinson’s Disease, a Literature Review of Human and Animal Studies
Jaison Daniel Cucarián Hurtado, Jenny Paola Berrío Sánchez, Ramiro Barcos Nunes, Alcyr Alves de Oliveira
Stem Cell Reviews and Reports.2018; 14(2): 166. CrossRef - Postural control and the relation with cervical sensorimotor control in patients with idiopathic adult-onset cervical dystonia
J. De Pauw, R. Mercelis, A. Hallemans, G. Van Gils, S. Truijen, P. Cras, W. De Hertogh
Experimental Brain Research.2018; 236(3): 803. CrossRef - Efecto de un programa de rehabilitación mediante entrenamiento en tapiz rodante con tareas duales en las alteraciones del equilibrio y la marcha en el daño cerebral adquirido
Á. Aguilera-Rubio, P. Fernández-González, F. Molina-Rueda, A. Cuesta-Gómez
Rehabilitación.2018; 52(2): 107. CrossRef - Noisy vestibular stimulation increases gait speed in normals and in bilateral vestibulopathy
Shinichi Iwasaki, Chisato Fujimoto, Naoya Egami, Makoto Kinoshita, Fumiharu Togo, Yoshiharu Yamamoto, Tatsuya Yamasoba
Brain Stimulation.2018; 11(4): 709. CrossRef - Paradoxical kinesia induced by appetitive 50-kHz ultrasonic vocalizations in rats depends on glutamatergic mechanisms in the inferior colliculus
Luan Castro Tonelli, Markus Wöhr, Rainer Schwarting, Liana Melo-Thomas
Neuropharmacology.2018; 135: 172. CrossRef - Managing Gait, Balance, and Posture in Parkinson’s Disease
Bettina Debû, Clecio De Oliveira Godeiro, Jarbas Correa Lino, Elena Moro
Current Neurology and Neuroscience Reports.2018;[Epub] CrossRef - Vestibular signals in primate cortex for self-motion perception
Yong Gu
Current Opinion in Neurobiology.2018; 52: 10. CrossRef - What If Low Back Pain Is the Most Prevalent Parkinsonism in the World?
Jesse V. Jacobs, Sharon M. Henry, Fay B. Horak
Frontiers in Neurology.2018;[Epub] CrossRef - The Dorsal Column Lesion Model of Spinal Cord Injury and Its Use in Deciphering the Neuron‐Intrinsic Injury Response
Callan L. Attwell, Mike van Zwieten, Joost Verhaagen, Matthew R. J. Mason
Developmental Neurobiology.2018; 78(10): 926. CrossRef - Exergames Inherently Contain Cognitive Elements as Indicated by Cortical Processing
Phillipp Anders, Tim Lehmann, Helen Müller, Karoline B. Grønvik, Nina Skjæret-Maroni, Jochen Baumeister, Beatrix Vereijken
Frontiers in Behavioral Neuroscience.2018;[Epub] CrossRef - Phase matters: A role for the subthalamic network during gait
Gabriele Arnulfo, Nicolò Gabriele Pozzi, Chiara Palmisano, Alice Leporini, Andrea Canessa, Joachim Brumberg, Gianni Pezzoli, Cordula Matthies, Jens Volkmann, Ioannis Ugo Isaias, Nader Pouratian
PLOS ONE.2018; 13(6): e0198691. CrossRef - Social jetlag impairs balance control
Guilherme Silva Umemura, João Pedro Pinho, Bruno da Silva Brandão Gonçalves, Fabianne Furtado, Arturo Forner-Cordero
Scientific Reports.2018;[Epub] CrossRef - The consequence of cerebral small vessel disease: Linking brain atrophy to motor impairment in the elderly
Ning Su, Xinyu Liang, Fei‐Fei Zhai, Li‐Xin Zhou, Jun Ni, Ming Yao, Feng Tian, Shu‐Yang Zhang, Zheng‐Yu Jin, Li‐Ying Cui, Gaolang Gong, Yi‐Cheng Zhu
Human Brain Mapping.2018; 39(11): 4452. CrossRef - Extrapyramidal deficits in ALS: a combined biomechanical and neuroimaging study
Maryse Feron, Annabelle Couillandre, Eya Mseddi, Nicolas Termoz, Malek Abidi, Eric Bardinet, Daniel Delgadillo, Timothée Lenglet, Giorgia Querin, Marie-Laure Welter, Nadine Le Forestier, François Salachas, Gaelle Bruneteau, Maria del Mar Amador, Rabab Deb
Journal of Neurology.2018; 265(9): 2125. CrossRef - Brain responses during strategic online gaming of varying proficiencies: Implications for better gaming
Min Wang, Guangheng Dong, Lingxiao Wang, Hui Zheng, Marc N. Potenza
Brain and Behavior.2018;[Epub] CrossRef - Adaptations to Postural Perturbations in Patients With Freezing of Gait
Esther M. J. Bekkers, Sam Van Rossom, Elke Heremans, Kim Dockx, Surendar Devan, Sabine M. P. Verschueren, Alice Nieuwboer
Frontiers in Neurology.2018;[Epub] CrossRef - Influence of the intensity of galvanic vestibular stimulation and cutaneous stimulation on the soleus H-reflex in healthy individuals
Yohei Okada, Tomoyuki Shiozaki, Junji Nakamura, Yuji Azumi, Miyo Inazato, Moe Ono, Honoka Kondo, Miyuki Sugitani, Akiyoshi Matsugi
NeuroReport.2018; 29(13): 1135. CrossRef - Role of medical ozone in attenuating age-related changes in the rat cerebellum
Wael B Elkholy, Marwa A Al-Gholam
Microscopy.2018; 67(4): 214. CrossRef - A bio-inspired self-responding emotional behavior system for virtual creatures
Diana G. Gómez-Martínez, Jonathan-Hernando Rosales, Vianney Muñoz-Jiménez, Félix Ramos, Marco Ramos
Biologically Inspired Cognitive Architectures.2018; 26: 26. CrossRef - Balancing between the two: Are freezing of gait and postural instability in Parkinson’s disease connected?
E.M.J. Bekkers, B.W. Dijkstra, E. Heremans, S.M.P. Verschueren, B.R. Bloem, A. Nieuwboer
Neuroscience & Biobehavioral Reviews.2018; 94: 113. CrossRef - Effects of spinal cord stimulation on postural control in Parkinson's disease patients with freezing of gait
Andrea Cristina de Lima-Pardini, Daniel Boari Coelho, Carolina Pinto Souza, Carolina Oliveira Souza, Maria Gabriela dos Santos Ghilardi, Tiago Garcia, Mariana Voos, Matija Milosevic, Clement Hamani, Luis Augusto Teixeira, Erich Talamoni Fonoff
eLife.2018;[Epub] CrossRef - The Balance of Sleep: Role of the Vestibular Sensory System
Stephane Besnard, Brahim Tighilet, Christian Chabbert, Martin Hitier, Joseph Toulouse, Anne Legall, Marie-Laure Machado, Paul F. Smith
Sleep Medicine Reviews.2018;[Epub] CrossRef - Neural Mechanism of Human Gait and Postural Control
Hiroaki Fujimoto, Ichiro Miyai
The Japanese Journal of Rehabilitation Medicine.2018; 55(9): 724. CrossRef - Targeting the pedunculopontine nucleus in Parkinson's disease: Time to go back to the drawing board
Roger L. Albin, D. James Surmeier, Cecilia Tubert, Martin Sarter, Martijn L.T.M. Müller, Nicolaas I. Bohnen, William T. Dauer
Movement Disorders.2018; 33(12): 1871. CrossRef - Vestibular System and Self-Motion
Zhixian Cheng, Yong Gu
Frontiers in Cellular Neuroscience.2018;[Epub] CrossRef - Physical exercise enhances adult cortical plasticity in a neonatal rat model of hypoxic-ischemic injury: Evidence from BOLD-fMRI and electrophysiological recordings
Sun Young Chae, Jun Ho Jang, Geun Ho Im, Ji-Hyun Jeong, Won-Beom Jung, Sukjin Ko, Hyesoo Jie, Ji Hye Kim, Yun Sil Chang, Seungsoo Chung, Ki-Soo Kim, Jung Hee Lee
NeuroImage.2018;[Epub] CrossRef - Detection and Diagnosis of Paralysis Agitans
Xiao Fan, Wanrong Sun, Aifeng Ren, Dou Fan, Nan Zhao, Daniyal Haider, Xiaodong Yang, Qammer H. Abbasi
IEEE Access.2018; 6: 73023. CrossRef - Effect of Treadmill Training With Eyes Open and Closed on Knee Proprioception, Functional Balance and Mobility in Children With Spastic Diplegia
Samah Attia El Shemy
Annals of Rehabilitation Medicine.2018; 42(6): 854. CrossRef - Magnetic Vestibular Stimulation (MVS) As a Technique for Understanding the Normal and Diseased Labyrinth
Bryan K. Ward, Jorge Otero-Millan, Prem Jareonsettasin, Michael C. Schubert, Dale C. Roberts, David S. Zee
Frontiers in Neurology.2017;[Epub] CrossRef - Postural Control in Bilateral Vestibular Failure: Its Relation to Visual, Proprioceptive, Vestibular, and Cognitive Input
Andreas Sprenger, Jann F. Wojak, Nico M. Jandl, Christoph Helmchen
Frontiers in Neurology.2017;[Epub] CrossRef - Diabetic Neuropathy and Gait: A Review
Uazman Alam, David R. Riley, Ravinder S. Jugdey, Shazli Azmi, Satyan Rajbhandari, Kristiaan D’Août, Rayaz A. Malik
Diabetes Therapy.2017; 8(6): 1253. CrossRef - Freezing of gait is associated with cortical thinning in mesial frontal cortex
Miroslav Vastik, Pavel Hok, Jan Valosek, Petr Hlustik, Katerina Mensikova, Petr Kanovsky
Biomedical Papers.2017; 161(4): 389. CrossRef - Cerebral Small Vessel Disease Burden Is Associated with Motor Performance of Lower and Upper Extremities in Community-Dwelling Populations
Ning Su, Fei-Fei Zhai, Li-Xin Zhou, Jun Ni, Ming Yao, Ming-Li Li, Zheng-Yu Jin, Gao-Lang Gong, Shu-Yang Zhang, Li-Ying Cui, Feng Tian, Yi-Cheng Zhu
Frontiers in Aging Neuroscience.2017;[Epub] CrossRef - Cough-Anal Reflex May Be the Expression of a Pre-Programmed Postural Action
Paolo Cavallari, Francesco Bolzoni, Roberto Esposti, Carlo Bruttini
Frontiers in Human Neuroscience.2017;[Epub] CrossRef - The effect of visual information on gait parameters with induced ankle muscle fatigue
Hye Rim Suh, Jin-Hee Hwang, Su-Young Lee
Physical Therapy Rehabilitation Science.2017; 6(3): 140. CrossRef - Construct Validity and Reliability of the SARA Gait and Posture Sub-scale in Early Onset Ataxia
Tjitske F. Lawerman, Rick Brandsma, Renate J. Verbeek, Johannes H. van der Hoeven, Roelineke J. Lunsing, Hubertus P. H. Kremer, Deborah A. Sival
Frontiers in Human Neuroscience.2017;[Epub] CrossRef - Striatal But Not Extrastriatal Dopamine Receptors Are Critical to Dopaminergic Motor Stimulation
Yuhan Wang, Fu-Ming Zhou
Frontiers in Pharmacology.2017;[Epub] CrossRef
Comments on this article
 KMDS
KMDS
 E-submission
E-submission
 PubReader
PubReader ePub Link
ePub Link Cite
Cite