Articles
- Page Path
- HOME > J Mov Disord > Volume 8(3); 2015 > Article
-
Review Article
What Is Wrong with Balance in Parkinson’s Disease? - Jeong-Ho Park1, Yeo-Jeong Kang1, Fay Bahling Horak2,3
-
Journal of Movement Disorders 2015;8(3):109-114.
DOI: https://doi.org/10.14802/jmd.15018
Published online: September 10, 2015
1Department of Neurology, College of Medicine, Soonchunhyang University Bucheon Hospital, Bucheon, Korea
2Department of Neurology, School of Medicine, Oregon Health & Science University, Portland, OR, USA
3Portland VA Medical Center, Portland, OR, USA
- Corresponding author: Fay Bahling Horak, PT, PhD, Department of Neurology, School of Medicine, Oregon Health & Science University, 3181 SW Sam Jackson Park Rd, Portland, OR 97239-3098, USA Tel: +1-593-418-2600 Fax: +1-503-418-2501 E-mail: horakf@ohsu.edu
Copyright © 2015 The Korean Movement Disorder Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
- Postural instability and resulting falls are major factors determining quality of life, morbidity, and mortality in individuals with Parkinson’s disease (PD). A better understanding of balance impairments would improve management of balance dysfunction and prevent falls in patients with PD. The effects of bradykinesia, rigidity, impaired proprioception, freezing of gait and attention on postural stability in patients with idiopathic PD have been well characterized in laboratory studies. The purpose of this review is to systematically summarize the types of balance impairments contributing to postural instability in people with PD.
- Postural instability, a key component of functional mobility, is often overlooked by both clinicians and their patients with Parkinson’s disease (PD), who focus on gait impairments. However, balance problems and resulting falls are major factors determining quality of life, morbidity, and mortality in individuals with PD [1-4]. According to a retrospective fall study of 489 in-patients admitted to a department of neurology, approximately 60% of PD patients had a history of at least one fall over the previous twelve months [4]. A recent meta-analysis and prospective studies also report considerably high fall incidences, even in optimally-medicated, early-stage PD (40–70%) [1,5,6]. From the results of epidemiologic studies, it is evident that balance dysfunction and related falls are common in individuals with PD, and the risk of falls increases gradually with disease progression without vigorous intervention to reduce the risk.
- Balance can be defined as control of the body’s center of mass (CoM) over its base of support in order to achieve postural equilibrium and orientation [7]. Balance control requires active brain processes that integrate information from all levels of the nervous and musculoskeletal system, not only while moving (dynamic balance) but also while standing still (static balance). The basalganglia, a key pathologic structure in PD, is involved in control of balance via the thalamic-cortical-spinal loops and via the brainstem pedunculopontine nucleus and the reticulospinal system [8]. The basal ganglia is involved in controlling the flexibility of postural tone, scaling up the magnitude of postural movements, selecting postural strategies for environmental context (central set), and automatizing postural responses and gait [8-10]. Although subtle balance problems, such as increased postural sway jerkiness and slow turning, can be measured in PD even at the time of diagnosis, the severity and type of balance problems increases with disease progression [11,12].
- Although dopamine-sensitive bradykinesia and rigidity progressively affect balance and gait early in the disease, dopamine insensitive-balance problems increase later in the disease: impaired kinesthesia, inflexible postural set, lack of automaticity, and executive dysfunction. Each patient with PD eventually gets postural instability but the severity of each type of balance impairments may be very different. For example, Figure 1 shows declines in different types of balance control at different stages of disease for two different patients until each patient reaches a threshold of postural instability and frequent falls. The purpose of this review is to systematically summarize the types and underlying causes of balance impairments contributing to postural instability in people with PD. A better understanding of balance impairments would improve management of balance dysfunction and prevent falls in people with PD [13-17].
INTRODUCTION
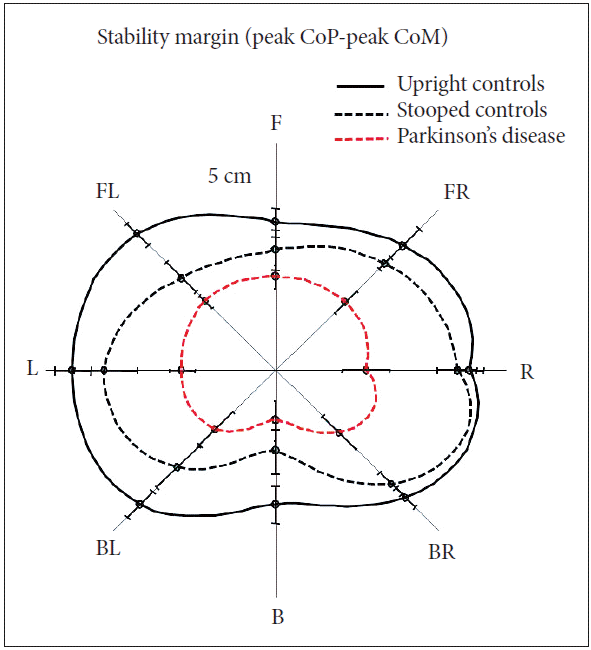
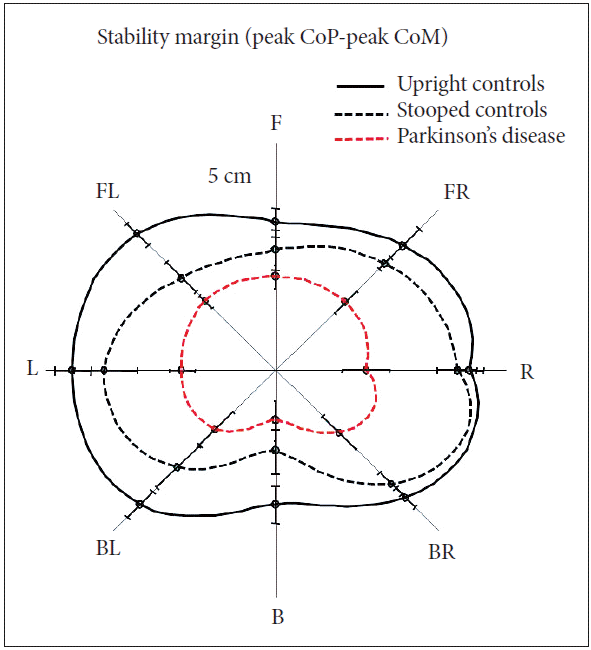
- Excessive, inflexible axial postural tone, called rigidity, results in many biomechanical impairments affecting postural control such as abnormally stooped (flexed) postural alignment, forward head, Pisa syndrome, reduced joint range of motion, and low back pain. Stooped posture, due to excessive, static flexor muscle activity, is associated with increased velocity and jerkiness of postural sway in stance [18,19]. Automatic postural responses to external displacements are also impaired by a stooped postural alignment. The postural stability margin, which is defined by the difference in displacement of the center of pressure (CoP) under the feet and displacement of the body CoM in response to external perturbations, is reduced in healthy subjects who assume a stooped posture like PD (Figure 2) [19]. This observation suggests that a stooped posture, per se, has a destabilizing effect on postural control and is not an attempt to compensate for postural instability. Responses to backward body perturbations are especially compromised by a stooped posture, perhaps due to a shortening of the tibialis anterior muscle, which is critical for bringing the backward falling body back to equilibrium.
- Axial rigidity measured during standing is most excessive at the hip joints than at the trunk or neck [20]. The severity of axial rigidity has been shown to relate to impairments in turning and rolling [21]. An inflexible pattern of excessive postural tone and consequent reduced range of motion mechanically impedes the ability to execute postural reactions effectively. For example, rigidity is associated with cocontraction of muscles around joints in response to postural perturbations, rather than efficient postural synergies up one side of the body, making patients vulnerable to loss of balance and falls [9].
RIGIDITY RESULTS IN BIOMECHANICAL IMPAIRMENTS
- Automatic postural responses to unexpected external perturbations of the body’s CoM can be classified into feet-in-place and change-in-support strategies [22]. In patients with PD, both types of postural response strategies are smaller and slower (bradykinetic) than normal. Feet-in-place postural responses are slow to develop force with reduced scaling up for larger perturbations, often leading to displacement of the body CoM within base of support [9]. Interestingly, postural responses are even weaker with levodopa medication [9]. Patients with PD are also unable to quickly change postural muscle synergies with changes in initial support conditions, called inflexible “central set”, for example, standing versus-sitting posture, holding versus-free stance, surface translation versus-rotation, and voluntary resist versus-relax [23,24]. The difficultly adapting the pattern of muscle activation for changes in conditions may explain why it is difficult for patients with PD to benefit from assistive devices, such as canes and walkers, to improve stability.
- Change-in-support strategies (for example, compensatory stepping or reaching) are also slowed and smaller due to bradykinesia [25]. Small compensatory stepping responses in response to external perturbations are larger when patients are provided a visual cue and can see their stepping leg [14]. However, when the stepping leg is not visible, the step to a visual target falls short, consistent with an increased visual dependence to overcome bradykinetic compensatory steps [14].
- Patients who have freezing of gait also show freezing of their compensatory stepping responses to recover equilibrium, suggesting a common pathological neural pathway that leads to falls [23]. In addition, whereas healthy subjects quickly reach out with an arm to grab stationary objects to stabilize equilibrium in response to an external perturbation, patients with PD often bring their arms closer to their bodies [25].
- Unfortunately deep brain stimulation (DBS) does not improve bradykinetic postural responses. Six months after DBS in the subthalamic nucleus (STN), both feet-in-place and compensatory stepping responses are weaker than before surgery whether the stimulators are turned on or off, although turning the stimulator on improves postural responses slightly (Figure 3) [26]. DBS in the globus pallidus interna (GPi) did not improve or worsen postural responses [26]. A recent meta-analysis also showed that postural instability and gait difficulty subscores of the Unified Parkinson’s disease rating scale were not maintained over time after surgery, particularly for STN DBS compared to GPi [27].
- Like postural responses, anticipatory (or preparatory) postural adjustments (APAs) are bradykinetic. APAs are essential in maintaining postural equilibrium during self-initiated, voluntary movements such as: step-initiation, rise-to-toes, arm raising, or leg lifting. For example, normal step initiation is associated with a preparatory lateral shift of the body CoM toward the stance leg due to active displacement of the CoP toward the swing leg prior to lifting a foot off the ground. Patients with PD have smaller than normal APAs and are unable to scale up the size of APAs as needed with wider stance (Figure 4) [28]. The small size of the lateral APA weight shift prior to step initiation, results in start hesitation or akinetic freezing of gait, especially when initial stance width is wide. Bradykinetic APAs may explain why patients with PD tend to keep their feet close together as a narrow stance requires smaller APAs than a wide stance to initiate gait. Levodopa and external cues increase the size of APAs prior to step initiation, whereas DBS surgery, especially in the STN, reduces the size of APAs [29].
BRADYKINESIA IMPAIRS POSTURAL RESPONSES AND ANTICIPATORY POSTURAL ADJUSTMENTS
- Central sensory integration involves active interpretation of visual, vestibular and somatosensory inputs for orientation of the body in space. When sensory information is unavailable or conflicting, a process of sensory reweighting occurs so the nervous system ignores ambiguous, unhelpful information and relies more on useful sensory information. For example, somatosensory input normally contributes 70% to postural stability when standing on a firm surface with eyes open but, but vestibular input contributes 100% to postural stability when standing on an unstable surface with eyes closed [30]. Patients with advanced PD are often unable to stand on an unstable surface with eye closed, although their vestibular system is usually normal [31]. This finding suggests that patients with PD may take more time for the sensory reweighting process than healthy people. Another problem in sensory orientation is that the patients with PD have impaired use of proprioception for a kinesthetic body map. For example, patients with PD have difficulty perceiving small changes in surface inclination, i.e., impaired kinesthesia [32]. Consequently, a poor sense of kinesthesia in PD is compensated by an over-reliance on vision for postural orientation.
IMPAIRED KINESTHESIA AFFECTS SENSORY INTEGRATION
- Dynamic balance control involves controlling the body CoM with respect to, but not necessarily over a moving base of support. Walking becomes less automatic in PD so it requires more attention, particularly for challenging tasks such as turning, walking between obstacles, and dual-tasking. Newly diagnosed patients with PD show signs of bradykinetic gait features, such as reduced trunk rotation, decreased arm swing, and slow turns, even when walking speed is normal [11]. Extent of arm swing on the most involved side may, in fact, be the most sensitive early sign of gait affected by the disease. Bradykinesia results in slow gait velocity and shuffling of the feet that progresses with the disease. Freezing of gait is not usually only lack of stepping but often includes high frequency “trembling of the legs” as the patients attempt to get their feet moving. These quick motions may represent repeated, multiple APAs that get larger and larger until a step is finally triggered [33].
BRADYKINESIA AND FREEZING IMPAIR GAIT
- Many aspects of cognitive impairment in PD, such as altered attention, narrow cognitive focus, comorbid dementia, and fear of falls, could affect balance control. Normally, humans can walk efficiently with perfect balancing while thinking or carrying objects simultaneously, but people with PD often fail this dual-task walking. In fact, cognitive loading (i.e.; subtraction by Serial 7s) during gait affect most temporal and spatial gait measures in PD even more than age-matched control subjects [34,35]. The underlying mechanism is still unknown but it is assumed that basal ganglia may be responsible for making postural control “automatic” as it shifts from the cortical loop to brainstem loop. This automaticity of postural control, as measured by minimal change in balance and/or gait with a dual, cognitive task, may be a good measure of successful rehabilitation of imbalance in patients with PD.
COGNITION-IMPAIRED ATTENTION
- Patients with PD show impairments in many aspects of postural control including: 1) rigidity affecting biomechanics, 2) bradykinesia of postural responses and anticipatory postural adjustments, 3) impaired kinesthesia for sensory integration, 4) bradykinetic gait with freezing, and 5) less automaticity of gait and balance. Unfortunately, levodopa and DBS often do not improve these postural impairments, so patients with PD need rehabilitation to improve balance and gait and prevent falls.
CONCLUSION
-
Conflicts of Interest
OHSU and Dr. Horak have a significant financial interest in APDM, a company that may have a commercial interest in the results of this research and technology. This potential institutional and individual conflict has been reviewed and managed by OHSU.
Notes
- This publication was made possible with support from a grant from the National Institutes on Aging (AG006457) a component of the National Institutes of Health (NIH), and NIH Roadmap for Medical Research.
Acknowledgments



- 1. Kerr GK, Worringham CJ, Cole MH, Lacherez PF, Wood JM, Silburn PA. Predictors of future falls in Parkinson disease. Neurology 2010;75:116–124.ArticlePubMed
- 2. Rahman S, Griffin HJ, Quinn NP, Jahanshahi M. Quality of life in Parkinson’s disease: the relative importance of the symptoms. Mov Disord 2008;23:1428–1434.ArticlePubMed
- 3. Williams DR, Watt HC, Lees AJ. Predictors of falls and fractures in bradykinetic rigid syndromes: a retrospective study. J Neurol Neurosurg Psychiatry 2006;77:468–473.ArticlePubMedPMC
- 4. Stolze H, Klebe S, Zechlin C, Baecker C, Friege L, Deuschl G. Falls in frequent neurological diseases--prevalence, risk factors and aetiology. J Neurol 2004;251:79–84.ArticlePubMed
- 5. Pickering RM, Grimbergen YA, Rigney U, Ashburn A, Mazibrada G, Wood B, et al. A meta-analysis of six prospective studies of falling in Parkinson’s disease. Mov Disord 2007;22:1892–1900.ArticlePubMed
- 6. Bloem BR, Grimbergen YA, Cramer M, Willemsen M, Zwinderman AH. Prospective assessment of falls in Parkinson’s disease. J Neurol 2001;248:950–958.ArticlePubMed
- 7. MacPherson JM, Horak FB. Posture. In: Kandel ER, Schwartz JH, Jessell TM, Siegelbaum SA, Hudspeth AJ, editors. Principles of neural science. 5th ed. New York: McGraw-Hill; 2000:935–959.
- 8. Takakusaki K, Habaguchi T, Ohtinata-Sugimoto J, Saitoh K, Sakamoto T. Basal ganglia efferents to the brainstem centers controlling postural muscle tone and locomotion: a new concept for understanding motor disorders in basal ganglia dysfunction. Neuroscience 2003;119:293–308.ArticlePubMed
- 9. Horak FB, Frank J, Nutt J. Effects of dopamine on postural control in parkinsonian subjects: scaling, set, and tone. J Neurophysiol 1996;75:2380–2396.ArticlePubMed
- 10. Horak FB, Dimitrova D, Nutt JG. Direction-specific postural instability in subjects with Parkinson’s disease. Exp Neurol 2005;193:504–521.ArticlePubMed
- 11. Zampieri C, Salarian A, Carlson-Kuhta P, Aminian K, Nutt JG, Horak FB. The instrumented timed up and go test: potential outcome measure for disease modifying therapies in Parkinson’s disease. J Neurol Neurosurg Psychiatry 2010;81:171–176.ArticlePubMedPMC
- 12. Mancini M, Horak FB, Zampieri C, Carlson-Kuhta P, Nutt JG, Chiari L. Trunk accelerometry reveals postural instability in untreated Parkinson’s disease. Parkinsonism Relat Disord 2011;17:557–562.ArticlePubMedPMC
- 13. Robinson K, Dennison A, Roalf D, Noorigian J, Cianci H, Bunting-Perry L, et al. Falling risk factors in Parkinson’s disease. NeuroRehabilitation 2005;20:169–182.ArticlePubMed
- 14. Jacobs JV, Horak FB. Abnormal proprioceptive-motor integration contributes to hypometric postural responses of subjects with Parkinson’s disease. Neuroscience 2006;141:999–1009.ArticlePubMed
- 15. Maki BE, McIlroy WE. Control of rapid limb movements for balance recovery: age-related changes and implications for fall prevention. Age Ageing 2006;35 Suppl 2:ii12–ii18.ArticlePDF
- 16. King LA, Horak FB. Lateral stepping for postural correction in Parkinson’s disease. Arch Phys Med Rehabil 2008;89:492–499.ArticlePubMedPMC
- 17. Horak FB. Postural orientation and equilibrium: what do we need to know about neural control of balance to prevent falls? Age Ageing 2006;35 Suppl 2:ii7–ii11.ArticlePDF
- 18. Bloem BR, Beckley DJ, van Dijk JG. Are automatic postural responses in patients with Parkinson’s disease abnormal due to their stooped posture? Exp Brain Res 1999;124:481–488.ArticlePubMed
- 19. Jacobs JV, Dimitrova DM, Nutt JG, Horak FB. Can stooped posture explain multidirectional postural instability in patients with Parkinson’s disease? Exp Brain Res 2005;166:78–88.ArticlePubMedPMC
- 20. Wright WG, Gurfinkel VS, Nutt J, Horak FB, Cordo PJ. Axial hypertonicity in Parkinson’s disease: direct measurements of trunk and hip torque. Exp Neurol 2007;208:38–46.ArticlePubMedPMC
- 21. Franzén E, Paquette C, Gurfinkel VS, Cordo PJ, Nutt JG, Horak FB. Reduced performance in balance, walking and turning tasks is associated with increased neck tone in Parkinson’s disease. Exp Neurol 2009;219:430–438.ArticlePubMedPMC
- 22. Horak FB, Nashner LM. Central programming of postural movements: adaptation to altered support-surface configurations. J Neurophysiol 1986;55:1369–1381.ArticlePubMed
- 23. Chong RK, Horak FB, Woollacott MH. Parkinson’s disease impairs the ability to change set quickly. J Neurol Sci 2000;175:57–70.ArticlePubMed
- 24. Horak FB, Nutt JG, Nashner LM. Postural inflexibility in parkinsonian subjects. J Neurol Sci 1992;111:46–58.ArticlePubMed
- 25. Carpenter MG, Allum JH, Honegger F, Adkin AL, Bloem BR. Postural abnormalities to multidirectional stance perturbations in Parkinson’s disease. J Neurol Neurosurg Psychiatry 2004;75:1245–1254.ArticlePubMedPMC
- 26. St George RJ, Carlson-Kuhta P, Nutt JG, Hogarth P, Burchiel KJ, Horak FB. The effect of deep brain stimulation randomized by site on balance in Parkinson’s disease. Mov Disord 2014;29:949–953.ArticlePubMedPMC
- 27. St George RJ, Nutt JG, Burchiel KJ, Horak FB. A meta-regression of the long-term effects of deep brain stimulation on balance and gait in PD. Neurology 2010;75:1292–1299.ArticlePubMedPMC
- 28. Rocchi L, Chiari L, Mancini M, Carlson-Kuhta P, Gross A, Horak FB. Step initiation in Parkinson’s disease: influence of initial stance conditions. Neurosci Lett 2006;406:128–132.ArticlePubMed
- 29. Rocchi L, Carlson-Kuhta P, Chiari L, Burchiel KJ, Hogarth P, Horak FB. Effects of deep brain stimulation in the subthalamic nucleus or globus pallidus internus on step initiation in Parkinson disease: laboratory investigation. J Neurosurg 2012;117:1141–1149.ArticlePubMedPMC
- 30. Cenciarini M, Peterka RJ. Stimulus-dependent changes in the vestibular contribution to human postural control. J Neurophysiol 2006;95:2733–2750.ArticlePubMed
- 31. Frenklach A, Louie S, Koop MM, Bronte-Stewart H. Excessive postural sway and the risk of falls at different stages of Parkinson’s disease. Mov Disord 2009;24:377–385.ArticlePubMed
- 32. Wright WG, Gurfinkel VS, King LA, Nutt JG, Cordo PJ, Horak FB. Axial kinesthesia is impaired in Parkinson’s disease: effects of levodopa. Exp Neurol 2010;225:202–209.ArticlePubMedPMC
- 33. Jacobs JV, Nutt JG, Carlson-Kuhta P, Stephens M, Horak FB. Knee trembling during freezing of gait represents multiple anticipatory postural adjustments. Exp Neurol 2009;215:334–341.ArticlePubMedPMC
- 34. Kelly VE, Eusterbrock AJ, Shumway-Cook A. A review of dual-task walking deficits in people with Parkinson’s disease: motor and cognitive contributions, mechanisms, and clinical implications. Parkinsons Dis 2012;2012:918719.ArticlePubMedPDF
- 35. Peterson DS, Fling BW, Mancini M, Cohen RG, Nutt JG, Horak FB. Dual-task interference and brain structural connectivity in people with Parkinson’s disease who freeze. J Neurol Neurosurg Psychiatry 2015;86:786–792.ArticlePubMedPMC
REFERENCES
Figure & Data
References
Citations

- Methods for evaluating gait associated dynamic balance and coordination in rodents
Akshat D. Modi, Anavi Parekh, Zeenal H. Patel
Behavioural Brain Research.2024; 456: 114695. CrossRef - Imbalance and gait impairment in Parkinson’s disease: discussing postural instability and ataxia
Carlos Henrique F. Camargo, Silvia Aparecida Ferreira-Peruzzo, Danieli Isabel Romanovitch Ribas, Gustavo L. Franklin, Hélio A. G. Teive
Neurological Sciences.2024; 45(4): 1377. CrossRef - Wearable devices for gait and posture monitoring via telemedicine in people with movement disorders and multiple sclerosis: a systematic review
Francesca Salaorni, Giulia Bonardi, Federico Schena, Michele Tinazzi, Marialuisa Gandolfi
Expert Review of Medical Devices.2024; 21(1-2): 121. CrossRef - An improved intermittent control model of postural sway during quiet standing implemented by a data driven approach
Maosheng Gao, Kai Chen, Ying Yang
Journal of Biomechanics.2024; 163: 111921. CrossRef - Effects of physical exercise interventions on balance, postural stability and general mobility in Parkinson’s disease: a network meta-analysis
Patricia Lorenzo-García, Iván Cavero-Redondo, Sergio Núñez de Arenas-Arroyo, María José Guzmán-Pavón, Susana Priego-Jiménez, Celia Álvarez-Bueno
Journal of Rehabilitation Medicine.2024; 56: jrm10329. CrossRef - Dynamic Measures of Balance during Obstacle-Crossing in Self-Selected Gait in Individuals with Mild-to-Moderate Parkinson’s Disease
Cathy Harro, Gordon Alderink, Lauren Hickox, David W. Zeitler, Michele Avery, Courtney Daman, Dori Laker
Applied Sciences.2024; 14(3): 1271. CrossRef - Older adults and individuals with Parkinson’s disease control posture along suborthogonal directions that deviate from the traditional anteroposterior and mediolateral directions
Madhur Mangalam, Damian G. Kelty-Stephen, Ivan Seleznov, Anton Popov, Aaron D. Likens, Ken Kiyono, Nick Stergiou
Scientific Reports.2024;[Epub] CrossRef - Exploring the Strategies People with Parkinson's Disease Use to Self-track Symptoms and Medications
Charlotte Tang, Imrul K. Shuva, Matthew Thelen, Linda Zhu, Nathaniel S. Miller
ACM Transactions on Accessible Computing.2024; 17(1): 1. CrossRef - Spatial variability and directional shifts in postural control in Parkinson’s disease
Damian G. Kelty-Stephen, Ken Kiyono, Nick Stergiou, Madhur Mangalam
Clinical Parkinsonism & Related Disorders.2024; : 100249. CrossRef - Assessing perceptions to a virtual reality intervention to improve trunk control in Parkinson’s disease: a preliminary study
Alexander Stamenkovic, Matthew Underation, Leslie J. Cloud, Peter E. Pidcoe, Mark S. Baron, Robert Hand, Christopher R. France, Susanne M. van der Veen, James S. Thomas
Virtual Reality.2023; 27(1): 465. CrossRef - Association of Depression With Early Occurrence of Postural Instability in Parkinson’s Disease
Yun Su Hwang, Sungyang Jo, Kye Won Park, Seung Hyun Lee, Sangjin Lee, Sun Ju Chung
Journal of Movement Disorders.2023; 16(1): 68. CrossRef - Clinical and MRI features of gait and balance disorders in neurodegenerative diseases
Elisabetta Sarasso, Massimo Filippi, Federica Agosta
Journal of Neurology.2023; 270(3): 1798. CrossRef - Effects of Oral Levodopa on Balance in People with Idiopathic Parkinson’s Disease
Tim Leroy, Remco J. Baggen, Nina Lefeber, Nolan Herssens, Patrick Santens, Miet De Letter, Leen Maes, Katie Bouche, Anke Van Bladel
Journal of Parkinson's Disease.2023; 13(1): 3. CrossRef - The Use of the Static Posturography to Assess Balance Performance in a Parkinson’s Disease Population
Sergio Sebastia-Amat, Juan Tortosa-Martínez, Basilio Pueo
International Journal of Environmental Research and Public Health.2023; 20(2): 981. CrossRef - Der Einfluss vom Exergaming mit kommerziell verfügbaren Spielkonsolen auf die Balance und Lebensqualität bei Menschen mit idiopathischem Parkinson-Syndrom – Eine systematische Übersichtsarbeit
Gaith Akrama, Lennert Sitzmann, Christian Baumann
physioscience.2023; 19(02): 63. CrossRef - Do cervical stabilization exercises change the effects of conventional exercises in patients with Parkinson’s disease?
Emine Nur Demircan, Nezire Köse, Gül Yalçın Çakmaklı, Songül Aksoy, Rahşan Göçmen, Hatice Yağmur Zengin, Bülent Elibol
Neurological Research.2023; 45(10): 936. CrossRef - Design of SmartWalk for Estimating Implication of Pathway With Turn and Task Condition on Postural and Gait Indices: Relevance to Fear of Fall
Priya Pallavi, Neeti Jariwala, Niravkumar Patel, Manasi Kanetkar, Shraddha Diwan, Uttama Lahiri
IEEE Transactions on Neural Systems and Rehabilitation Engineering.2023; 31: 406. CrossRef - Rehabilitation Interventions for Improving Balance in Parkinson’s Disease
Francesco Lena, Nicola Modugno, Giulio Greco, Monica Torre, Simone Cesarano, Marco Santilli, Auwal Abdullahi, Giuseppe Giovannico, Mohammad Etoom
American Journal of Physical Medicine & Rehabilitation.2023; 102(3): 270. CrossRef - Effects of Different Exercise Modes on Gait Performance of Parkinson’s Disease Patients: A Systematic Review and Network Meta-Analysis
Shi-kun Zhang, Mei-ling Gu, Hong Xu, Wen-sheng Zhou, Su-jie Mao, Yong Yang
Perceptual and Motor Skills.2023; 130(4): 1524. CrossRef - Validity and Reliability of the Thai Version of the Freezing of Gait Questionnaire in Individuals With Parkinson’s disease
Arisa Taweekitikul, Phakamas Tanvijit, Pheeravut Tantisuvanitchkul, Prachaya Srivanitchapoom, Yuvadee Pitakpatapee, Fuengfa Khobkhun, Piyapong Akkathep
Annals of Rehabilitation Medicine.2023; 47(1): 45. CrossRef - Anticipatory Postural Adjustments and Compensatory Postural Responses to Multidirectional Perturbations—Effects of Medication and Subthalamic Nucleus Deep Brain Stimulation in Parkinson’s Disease
Tobias Heß, Christian Oehlwein, Thomas L. Milani
Brain Sciences.2023; 13(3): 454. CrossRef - PRIORITIZING FALL RISK MANAGEMENT: IRRESPECTIVE OF PARKINSON’S DISEASE PROGRESSION DURING REHABILITATION
Fakhir Ansari
Pakistan Journal of Rehabilitation.2023; 12(1): 84. CrossRef - Functional balance training in people with Parkinson’s disease: a protocol of balanceHOME randomized control trial with crossover
Sara Monleón Guinot, Constanza San Martín Valenzuela, Vivina Aranda Asensi, Concepción de Salazar Antón, Manuel Villanueva Navarro, Jose M. Tomás
Frontiers in Aging Neuroscience.2023;[Epub] CrossRef - Characterizing Freezing of Gait Episodes for Parkinson’s Disease Using a Wearable Device Quantifying Gait and Posture
Priya Pallavi, Niravkumar Patel, Manasi Kanetkar, Uttama Lahiri
Journal of Medical and Biological Engineering.2023; 43(3): 266. CrossRef - Dynamic Measures of Balance during a 90° Turn in Self-Selected Gait in Individuals with Mild Parkinson’s Disease
Gordon Alderink, Cathy Harro, Lauren Hickox, David W. Zeitler, Marie Bourke, Akeya Gosla, Sarah Rustmann
Applied Sciences.2023; 13(9): 5428. CrossRef - Differences between physical therapist ratings, self-ratings, and posturographic measures when assessing static balance exercise intensity
Jamie Ferris, Jonathan Zwier, Wendy J. Carender, Kathleen H. Sienko
Frontiers in Rehabilitation Sciences.2023;[Epub] CrossRef - Validity, reliability and minimal detectable change of Mini-BESTest Turkish version in neurological disorders
Bilinc Dogruoz Karatekin, Afitap İcagasioglu, Ozge Pasin
Acta Neurologica Belgica.2023; 123(4): 1519. CrossRef - A non-expensive bidimensional kinematic balance assessment can detect early postural instability in people with Parkinson’s disease
Gabriel Venas Santos, Matheus Silva d'Alencar, Andre Frazão Helene, Antonio C. Roque, José Garcia Vivas Miranda, Maria Elisa Pimentel Piemonte
Frontiers in Neurology.2023;[Epub] CrossRef - Unveiling the Impact of Outpatient Physiotherapy on Specific Motor Symptoms in Parkinson’s Disease: A Prospective Cohort Study
Yuta Terasawa, Koki Ikuno, Shintaro Fujii, Yuki Nishi, Emi Tanizawa, Sachio Nabeshima, Yohei Okada
Brain & Neurorehabilitation.2023;[Epub] CrossRef - Does Impaired Plantar Cutaneous Vibration Perception Contribute to Axial Motor Symptoms in Parkinson’s Disease? Effects of Medication and Subthalamic Nucleus Deep Brain Stimulation
Tobias Heß, Peter Themann, Christian Oehlwein, Thomas L. Milani
Brain Sciences.2023; 13(12): 1681. CrossRef - The influence of cognitive tasks on sensory organization test performance
Nathan Morelli, Nicholas R. Heebner, Courtney J. DeFeo, Matthew C. Hoch
Brazilian Journal of Otorhinolaryngology.2022; 88(6): 841. CrossRef - Influence of radial optic flow stimulation on static postural balance in Parkinson's disease: A preliminary study
Alessandro Piras, Aurelio Trofè, Andrea Meoni, Milena Raffi
Human Movement Science.2022; 81: 102905. CrossRef - The effect of Tai Chi on functional mobility, balance and falls in Parkinson's disease: A systematic review and meta-analysis of systematic reviews
Bahar Aras, Gulce Kallem Seyyar, Oznur Fidan, Ertugrul Colak
EXPLORE.2022; 18(4): 402. CrossRef - Avaliação do risco de quedas entre pessoas com doença de Parkinson
Franciny da Silva, Angela Maria Alvarez, Simony Fabíola Lopes Nunes, Maria Eduarda Massari Silva, Silvia Maria Azevedo Dos Santos
Escola Anna Nery.2022;[Epub] CrossRef - Acceptability and Preliminary Results of Technology-Assisted Balance Training in Parkinson’s Disease
Elvira Maranesi, Valentina Di Donna, Giuseppe Pelliccioni, Valentina Cameriere, Elisa Casoni, Renato Baldoni, Marco Benadduci, Nadia Rinaldi, Lorenzo Fantechi, Cinzia Giammarchi, Riccardo Luzi, Paolo Pelliccioni, Mirko Di Rosa, Pietro Scendoni, Giovanni R
International Journal of Environmental Research and Public Health.2022; 19(5): 2655. CrossRef - A systematic review on the effectiveness of perturbation-based balance training in postural control and gait in Parkinson’s disease
Daniel Boari Coelho, Claudia Eunice Neves de Oliveira, Marcos Vinicius Carvalho Guimarães, Caroline Ribeiro de Souza, Márcio Luiz dos Santos, Andrea C. de Lima-Pardini
Physiotherapy.2022; 116: 58. CrossRef - Therapeutic Implications of Caffeic Acid in Cancer and Neurological Diseases
Manzar Alam, Sarfraz Ahmed, Abdelbaset Mohamed Elasbali, Mohd Adnan, Shoaib Alam, Md. Imtaiyaz Hassan, Visweswara Rao Pasupuleti
Frontiers in Oncology.2022;[Epub] CrossRef - Rasch Validation of the Mini-BESTest in People With Parkinson Disease
Franco Franchignoni, Marco Godi, Stefano Corna, Andrea Giordano
Journal of Neurologic Physical Therapy.2022; 46(3): 219. CrossRef - Factors Contributing to Repeated Falls in Patients with Parkinsonʼs Disease : Focusing on Falls Self-efficacy and Cognition of Punishment
Yasuomi Tomii, Mika Otsuki, Naoya Hasegawa
Higher Brain Function Research.2022; 42(1): 29. CrossRef - Short-Term Motor Outcomes in Parkinson’s Disease after Subthalamic Nucleus Deep Brain Stimulation Combined with Post-Operative Rehabilitation: A Pre-Post Comparison Study
Kazunori Sato, Yoshihide Hokari, Eriko Kitahara, Nana Izawa, Kozo Hatori, Kaoru Honaga, Genko Oyama, Taku Hatano, Hirokazu Iwamuro, Atsushi Umemura, Yasushi Shimo, Nobutaka Hattori, Toshiyuki Fujiwara, Giuseppina Martella
Parkinson's Disease.2022; 2022: 1. CrossRef - Re-conceptualizing postural control assessment in sport-related concussion: Transitioning from the reflex/hierarchical model to the systems model
Thaer S. Manaseer, Douglas P. Gross, Martin Mrazik, Kathryn Schneider, Jackie L. Whittaker
Physiotherapy Theory and Practice.2021; 37(7): 763. CrossRef - Short-term effects of postural control by standing on a tilting board in patients with Parkinson’s disease
Naoya Hasegawa, Keita Ishikawa, Yuki Sato, Yasuhide Nakayama, Tadayoshi Asaka
Physiotherapy Theory and Practice.2021; 37(12): 1306. CrossRef - Comparison of the effect of whole-body vibration therapy versus conventional therapy on functional balance of patients with Parkinson’s disease: adding a mixed group
Eduardo Guadarrama-Molina, Carlos Enrique Barrón-Gámez, Ingrid Estrada-Bellmann, Jesús D. Meléndez-Flores, Paola Ramírez-Castañeda, Rosa María Gisela Hernández-Suárez, Minerva Menchaca-Pérez, Oscar Salas-Fraire
Acta Neurologica Belgica.2021; 121(3): 721. CrossRef - Impact of Physiotherapy Absence in COVID-19 Pandemic on Neurological State of Patients With Parkinson Disease
Alen Kapel, Domen Serdoner, Ester Fabiani, Tomaz Velnar
Topics in Geriatric Rehabilitation.2021; 37(1): 50. CrossRef - The relationship between attention and static balance disturbance in patients with Parkinson’s disease
Amir Dana, Amir Shams, Nahid Allafan, Alireza Bahrami
Neurological Sciences.2021; 42(12): 5107. CrossRef - Does the impaired postural control in Parkinson's disease affect the habituation to non-sequential external perturbation trials?
Victor Spiandor Beretta, Mark Gregory Carpenter, Fabio Augusto Barbieri, Paulo Cezar Rocha Santos, Diego Orcioli-Silva, Marcelo Pinto Pereira, Lilian Teresa Bucken Gobbi
Clinical Biomechanics.2021; 85: 105363. CrossRef - Abnormal center of mass feedback responses during balance: A potential biomarker of falls in Parkinson’s disease
J. Lucas McKay, Kimberly C. Lang, Sistania M. Bong, Madeleine E. Hackney, Stewart A. Factor, Lena H. Ting, Manabu Sakakibara
PLOS ONE.2021; 16(5): e0252119. CrossRef - Establishing the Rotenone-Induced Parkinson's Disease Animal Model in Wistar Albino Rats
Gangadhara Swamy, Rajendra Holla, Suresh R. Rao
Journal of Health and Allied Sciences NU.2021; 11(03): 158. CrossRef - Development of a Parkinson’s disease specific falls questionnaire
Dale M. Harris, Rachel L. Duckham, Robin M. Daly, Gavin Abbott, Liam Johnson, Timo Rantalainen, Wei-Peng Teo
BMC Geriatrics.2021;[Epub] CrossRef - Correlation between community balance and mobility scale (CB&M) with a battery of outcome measures to assess balance in Parkinson’s disease – a cross-sectional study
Ziona Lionel Dsouza, Sydney Roshan Rebello, Cherishma Dsilva
Archives of Physiotherapy.2021;[Epub] CrossRef - Cholinesterase inhibitors for gait, balance, and fall in Parkinson disease: a meta-analysis
Jia-Hung Chen, Tsai-Wei Huang, Chien-Tai Hong
npj Parkinson's Disease.2021;[Epub] CrossRef - Dancing With Parkinson's Disease: The SI-ROBOTICS Study Protocol
Roberta Bevilacqua, Marco Benadduci, Anna Rita Bonfigli, Giovanni Renato Riccardi, Giovanni Melone, Angela La Forgia, Nicola Macchiarulo, Luca Rossetti, Mauro Marzorati, Giovanna Rizzo, Pierpaolo Di Bitonto, Ada Potenza, Laura Fiorini, Federica Gabriella
Frontiers in Public Health.2021;[Epub] CrossRef - Balance and mobility in patients with newly diagnosed Parkinson’s disease – a five-year follow-up of a cohort in northern Sweden
Christer Johansson, Britta Lindström, Lars Forsgren, Gudrun M. Johansson
Disability and Rehabilitation.2020; 42(6): 770. CrossRef - Minimal Detectable Change for Balance Using the Biodex Balance System in Patients with Parkinson Disease
Jumes Leopoldino de Oliveira Lira, Carlos Ugrinowitsch, Rafael Fecchio, Daniel Boari Coelho, Acácio Moreira‐Neto, Renan Germano, Angelo Corrêa de Lima Miliatto, Bianca Cristina dos Santos Vieira Yano, Carla Silva‐Batista
PM&R.2020; 12(3): 281. CrossRef - Perception of whole-body motion during balance perturbations is impaired in Parkinson's disease and is associated with balance impairment
Sistania M. Bong, J. Lucas McKay, Stewart A. Factor, Lena H. Ting
Gait & Posture.2020; 76: 44. CrossRef - Postural impairments in Parkinson’s disease are not associated with changes in circadian rhythms changes
Luiz Claudio Zulai, André Martinez Albuquerque, Caluê Papcke, Fernando Mazzilli Louzada, Eduardo Mendonça Scheeren
Chronobiology International.2020; 37(1): 135. CrossRef - Choreographing life-experiences of balance control in people with Parkinson’s disease
Sofie LaGrone, Conran Joseph, Hanna Johansson, Birgit Enberg, Erika Franzén
BMC Neurology.2020;[Epub] CrossRef - Effect of home-based Tai Chi, Yoga or conventional balance exercise on functional balance and mobility among persons with idiopathic Parkinson’s disease: An experimental study
Arva Khuzema, A. Brammatha, V. Arul Selvan
Hong Kong Physiotherapy Journal.2020; 40(01): 39. CrossRef - A comparison of the relationship between manual dexterity and postural control in young and older individuals with Parkinson’s disease
Aybuke Cansu Kalkan, Turhan Kahraman, Biron Onur Ugut, Berril Donmez Colakoglu, Arzu Genc
Journal of Clinical Neuroscience.2020; 75: 89. CrossRef - Explaining Parkinsonian postural sway variabilities using intermittent control theory
Ranjita Dash, Vrutangkumar V. Shah, Harish J. Palanthandalam-Madapusi
Journal of Biomechanics.2020; 105: 109791. CrossRef - Rehabilitation of older people with Parkinson’s disease: an innovative protocol for RCT study to evaluate the potential of robotic-based technologies
Roberta Bevilacqua, Elvira Maranesi, Mirko Di Rosa, Riccardo Luzi, Elisa Casoni, Nadia Rinaldi, Renato Baldoni, Fabrizia Lattanzio, Valentina Di Donna, Giuseppe Pelliccioni, Giovanni Renato Riccardi
BMC Neurology.2020;[Epub] CrossRef - Assessment of Functional Activities in Individuals with Parkinson’s Disease Using a Simple and Reliable Smartphone-Based Procedure
Pilar Serra-Añó, José Francisco Pedrero-Sánchez, Marta Inglés, Marta Aguilar-Rodríguez, Ismael Vargas-Villanueva, Juan López-Pascual
International Journal of Environmental Research and Public Health.2020; 17(11): 4123. CrossRef - New insight into Parkinson’s disease‐related impairment of the automatic control of upright stance
Cédrick T. Bonnet, Arnaud Delval, Tarkeshwar Singh, Yann‐Romain Kechabia, Luc Defebvre
European Journal of Neuroscience.2020; 52(12): 4851. CrossRef - Innovative Parkinson's Disease Patients' Motor Skills Assessment: The i-PROGNOSIS Paradigm
Sofia Balula Dias, Athina Grammatikopoulou, José Alves Diniz, Kosmas Dimitropoulos, Nikos Grammalidis, Vicky Zilidou, Theodore Savvidis, Evdokimos Konstantinidis, Panagiotis D. Bamidis, Hagen Jaeger, Michael Stadtschnitzer, Hugo Silva, Gonçalo Telo, Ioann
Frontiers in Computer Science.2020;[Epub] CrossRef - Effects of multimodal balance training supported by rhythmical auditory stimuli in people with advanced stages of Parkinson's disease: a pilot randomized clinical trial
Tamine T.C. Capato, Jorik Nonnekes, Nienke M. de Vries, Joanna IntHout, Egberto R. Barbosa, Bastiaan R. Bloem
Journal of the Neurological Sciences.2020; 418: 117086. CrossRef - Responsiveness of Objective vs. Clinical Balance Domain Outcomes for Exercise Intervention in Parkinson's Disease
Naoya Hasegawa, Vrutangkumar V. Shah, Graham Harker, Patricia Carlson-Kuhta, John G. Nutt, Jodi A. Lapidus, Se Hee Jung, Nancy Barlow, Laurie A. King, Fay B. Horak, Martina Mancini
Frontiers in Neurology.2020;[Epub] CrossRef - Neuroprotective Effects of Coffee Bioactive Compounds: A Review
Katarzyna Socała, Aleksandra Szopa, Anna Serefko, Ewa Poleszak, Piotr Wlaź
International Journal of Molecular Sciences.2020; 22(1): 107. CrossRef - Parkinson's Disease and Parkinsonism
Michael T. Hayes
The American Journal of Medicine.2019; 132(7): 802. CrossRef - How to Select Balance Measures Sensitive to Parkinson’s Disease from Body-Worn Inertial Sensors—Separating the Trees from the Forest
Naoya Hasegawa, Vrutangkumar V. Shah, Patricia Carlson-Kuhta, John G. Nutt, Fay B. Horak, Martina Mancini
Sensors.2019; 19(15): 3320. CrossRef - Treatment options for postural instability and gait difficulties in Parkinson’s disease
Martijn L.T.M. Müller, Uros Marusic, Miriam van Emde Boas, Daniel Weiss, Nicolaas I. Bohnen
Expert Review of Neurotherapeutics.2019; 19(12): 1229. CrossRef - A new technique to test the effect of cognition on standing balance in Parkinson’s disease
M Bhatt, B Mahana, JJ Marotta, JH Ko, T Szturm
Open Journal of Parkinson's Disease and Treatment.2019; 2(1): 006. CrossRef - Step length determines minimum toe clearance in older adults and people with Parkinson’s disease
Lisa Alcock, Brook Galna, Ruth Perkins, Sue Lord, Lynn Rochester
Journal of Biomechanics.2018; 71: 30. CrossRef - Long-term effects of highly challenging balance training in Parkinson’s disease—a randomized controlled trial
Martin Benka Wallén, Maria Hagströmer, David Conradsson, Kimmo Sorjonen, Erika Franzén
Clinical Rehabilitation.2018; : 026921551878433. CrossRef - Parkinson's disease versus ageing: different postural responses to soleus muscle vibration
Diana Bzdúšková, Peter Valkovič, Zuzana Hirjaková, Jana Kimijanová, František Hlavačka
Gait & Posture.2018; 65: 169. CrossRef - A High-Intensity Multicomponent Agility Intervention Improves Parkinson Patients’ Clinical and Motor Symptoms
József Tollár, Ferenc Nagy, Norbert Kovács, Tibor Hortobágyi
Archives of Physical Medicine and Rehabilitation.2018; 99(12): 2478. CrossRef - Parkinson’s disease does not alter automatic visual-motor coupling in postural control
Caio Ferraz Cruz, Maria Elisa Pimentel Piemonte, Líria Akie Okai-Nobrega, Erika Okamoto, Ana Claudia de Souza Fortaleza, Martina Mancini, Fay Bahling Horak, José Angelo Barela
Neuroscience Letters.2018; 686: 47. CrossRef - Assessing Walking Adaptability in Parkinson's Disease: “The Interactive Walkway”
Daphne J. Geerse, Melvyn Roerdink, Johan Marinus, Jacobus J. van Hilten
Frontiers in Neurology.2018;[Epub] CrossRef - Camptocormia with Transient Ischemic Attack
Ju-Hee Oh, Dong-Woo Ryu, Si-Hoon Lee, Joong-Seok Kim
Journal of Movement Disorders.2017; 10(1): 62. CrossRef - Motor dysfunction and alterations in glutathione concentration, cholinesterase activity, and BDNF expression in substantia nigra pars compacta in rats with pedunculopontine lesion
Lisette Blanco-Lezcano, Javier Jimenez-Martin, Mei-Li Díaz-Hung, Esteban Alberti-Amador, Maylin Wong-Guerra, Ma. Elena González-Fraguela, Bárbara Estupiñán-Díaz, Teresa Serrano-Sánchez, Liliana Francis-Turner, Susana Delgado-Ocaña, Yanier Núñez-Figueredo,
Neuroscience.2017; 348: 83. CrossRef - Axial rigidity is related to the risk of falls in patients with Parkinson’s disease
Roberto Cano-de-la-Cuerda, Lydia Vela-Desojo, Juan Carlos Miangolarra-Page, Yolanda Macías-Macías
NeuroRehabilitation.2017; 40(4): 569. CrossRef - Body Posture, Postural Stability, and Metabolic Age in Patients with Parkinson’s Disease
Jacek Wilczyński, Agnieszka Pedrycz, Dariusz Mucha, Tadeusz Ambroży, Dawid Mucha
BioMed Research International.2017; 2017: 1. CrossRef - Investigation of Anticipatory Postural Adjustments during One-Leg Stance Using Inertial Sensors: Evidence from Subjects with Parkinsonism
Gianluca Bonora, Martina Mancini, Ilaria Carpinella, Lorenzo Chiari, Maurizio Ferrarin, John G. Nutt, Fay B. Horak
Frontiers in Neurology.2017;[Epub] CrossRef - Altered neurofilament protein expression in the lateral vestibular nucleus in Parkinson’s disease
Thomas P. Wellings, Alan M. Brichta, Rebecca Lim
Experimental Brain Research.2017; 235(12): 3695. CrossRef - Biomechanical Analysis of Dance for Parkinson’s Disease: A Paradoxical Case Study of Balance and Gait Effects?
Kristen L. Sowalsky, Jill Sonke, Lori J.P. Altmann, Leonardo Almeida, Chris J. Hass
EXPLORE.2017; 13(6): 409. CrossRef - Femur Fractures in Parkinsonism: Analysis of a National Sample Cohort in South Korea
Soo Jeong An, Seung-Hwan Lee, Seo-Young Lee, Jae-Woo Kwon, Seung-Joon Lee, Young-Ju Kim
Journal of Clinical Neurology.2017; 13(4): 380. CrossRef - The International Classification of Functioning, Disability and Health as Applied to Parkinson’s Disease: A Literature Review
Audrin Said Vojciechowski, Talita Gianello Gnoato Zotz, Ana Paula Cunha Loureiro, Vera Lúcia Israel
Advances in Parkinson's Disease.2016; 05(02): 29. CrossRef - Can Gait Signatures Provide Quantitative Measures for Aiding Clinical Decision-Making? A Systematic Meta-Analysis of Gait Variability Behavior in Patients with Parkinson's Disease
Niklas König, Navrag B. Singh, Christian R. Baumann, William R. Taylor
Frontiers in Human Neuroscience.2016;[Epub] CrossRef
Comments on this article
- Figure
- Related articles
-
- Fighting Against the Clock: Circadian Disruption and Parkinson’s Disease
- Investigation of the Long-Term Effects of Amantadine Use in Parkinson’s Disease
- Current Status and Future Perspectives on Stem Cell-Based Therapies for Parkinson’s Disease
- Association of Depression With Early Occurrence of Postural Instability in Parkinson’s Disease
- The Supplementary Motor Complex in Parkinson’s Disease
 KMDS
KMDS
 E-submission
E-submission
 PubReader
PubReader ePub Link
ePub Link Cite
Cite