Electrophysiological Evaluation of Oropharyngeal Dysphagia in Parkinson’s Disease
Article information
Abstract
Parkinson’s disease (PD) is a chronic, neurodegenerative movement disorder that typically affects elderly patients. Swallowing disorders are highly prevalent in PD and can have grave consequences, including pneumonia, malnutrition, dehydration and mortality. Neurogenic dysphagia in PD can manifest with both overt clinical symptoms or silent dysphagia. Regardless, early diagnosis and objective follow-up of dysphagia in PD is crucial for timely and appropriate care for these patients. In this review, we provide a comprehensive summary of the electrophysiological methods that can be used to objectively evaluate dysphagia in PD. We discuss the electrophysiological abnormalities that can be observed in PD, their clinical correlates and the pathophysiology underlying these findings.
INTRODUCTION
Parkinson’s disease (PD) is a common neurodegenerative disorder of the elderly, affecting approximately 1% of the population over 50 years of age. Early symptoms of PD may be present in as many as 10% of those over 60 years of age [1-4].
In PD patients, neurogenic dysphagia occurs very frequently. Some of the main consequences of neurogenic dysphagia includes apparent and silent aspirations to the airway that often results in pneumonia, malnutrition and dehydration, which are potentially fatal complications for dysphagic patients with PD [5,6].
The incidence of dysphagia in PD has been reported to be between 18.5% to 100% [7-9]. Not surprisingly, many PD patients report no swallow impairment, presumably due to their frequent lack of insight about their neurological changes [2,9,10].
Indeed, neurogenic dysphagia could be insidious and slowly progressive in PD. There may be a subclinical period before the onset of clinically overt dysphagia. Subjective dysphagia or subclinical dysphagia occurs in 35% of PD patients who live at home when swallowing is assessed by some objective diagnostic tests [11]. When objective swallowing assessment is performed in all PD patients, the estimated prevalence of dysphagia reaches 82% [1,3]. In advanced stages of PD, 95–100% of the patients will have dysphagia [3,9,12-14].
The diagnosis of dysphagia in the early subclinical period may be critical for the management of complications of dysphagia. This can also be important to study in elderly patients with or without clinically overt swallowing difficulties [15-17].
Patients with chronic, progressive neurological disorders, such as PD are submitted to invasive swallowing tests, especially for the objective diagnosis of aspiration. Invasive tests for dysphagia include videofluoroscopy (VFS), fiberoptic endoscopic examination of swallowing (FEES), manometry and manofluoroscopy. VFS is considered the gold standard for analysis of swallowing physiology and detection of aspiration [18]; however, this technique suffers from problems associated with reliability [19-21]. VFS may not be practical for severely handicapped neurological patients, such as those with advanced PD and dementia. Other concerns related to the VFS approach include its cost, qualitative rather than quantitative nature, and readily lack of easy repetitive follow-up testing.
Fiberoptic endoscopic examination of swallowing is chiefly performed by otorhinolaryngologists [22,23] and cannot be used to assess the oral phase of swallowing [24,25].
Parkinson’s disease cases with dysphagia may be evaluated and followed using non-invasive screening tests, such as water swallowing tests. One diagnostic approach is the 3 ounce water test [26-29], another is the swallowing speed/timed test [30-32], and finally, there are various electromyogram (EMG) methods that can objectively and readily assess swallowing, including the dysphagia limit, single-bolus analysis, sequential water swallowing (SWS) test and polygraphic recordings. Because our review focuses on the electrophysiological evaluation of swallowing and its disorders, invasive methods and water swallowing tests without EMG are beyond the scope of this review.
HOW TO EVALUATE OROPHARYNGEAL SWALLOWING USING EMG METHODS
It is well known that neurogenic dysphagia may occur frequently in both the early and late periods of PD. There are three important diagnostic points in PD patients with dysphagia.
1) The diagnosis of dysphagia should be objective and quantitative.
2) The severity or prognosis of dysphagia and the therapeutic effect of any type of treatment should be followed up during the chronic course of PD.
3) Spontaneous swallows should be identified clearly when patients cannot perform voluntary swallows.
For these purposes, we have developed a series of electrophysiological methods that are readily and safely applicable to PD patients.
When we look at the process of deglutition as a whole, there are three different locations for the swallowing process. One of them is known as the oropharynx, where all of the peripheral sensory-motor events of swallowing occur (Figure 1). The second location is the medullary swallowing center in and around the reticular network of nucleus tractus solitarius (NTS) and nucleus ambiguus (NA). There is a central pattern generator (CPG) for swallowing integration in and around these nuclei.
The medullary swallowing center and CPG receives all peripheral inputs and descending motor drives. The third location is the cerebral cortex [33-35] and some subcortical structures that are connected to the brainstem center via corticobulbar pathways. The cerebral cortex is mainly involved with triggering voluntary swallows [36]. Another type of swallowing, called spontaneous or reflexive swallowing, originates from the CPG of the bulbar swallowing center [37].
It has become convenient to divide swallowing into three phases: oral, pharyngeal, and esophageal. This conventional division of human swallowing is usually ascribed to Magendie (1825) [38]. Oral and pharyngeal phases are firmly related to each other at the end of the oral phases, and consequently, they are referred to as oropharyngeal swallowing. The majority of neurogenic dysphagia appears in this stage [39-42]. The oropharyngeal stage of swallowing will be the focus of this review.
In the oropharyngeal phase of swallowing, there are several successive events. The first event is the triggering of the swallowing reflex that occurs at the oropharyngeal cavities by the bolus, which produces sensory input that travels up to the brainstem and cortex [24,36,38,42-44]. The second event is the elevation and closure of the larynx and soft palate for airway protection together with contraction of the submental/suprahyoid muscles situated under the chin [40,45-49]. The third is the propulsive, pumping forces of the tongue [50] and sweeping function of the bolus on the pharyngeal constrictor muscles [51]. The fourth event is the relaxation and opening of the cricopharyngeal muscle of the upper esophageal sphincter (UES), which is normally closed at rest. Finally, the bolus is transported from the pharynx into the esophagus [40,41,52]. The oral phase of deglutition is under voluntary control, and the pharyngeal phase is a complex swallowing act. Both voluntary and involuntary motor events are coordinated during oropharyngeal swallowing [37,41].
ELECTROPHYSIOLOGICAL EVALUATION OF OROPHARYNGEAL SWALLOWING
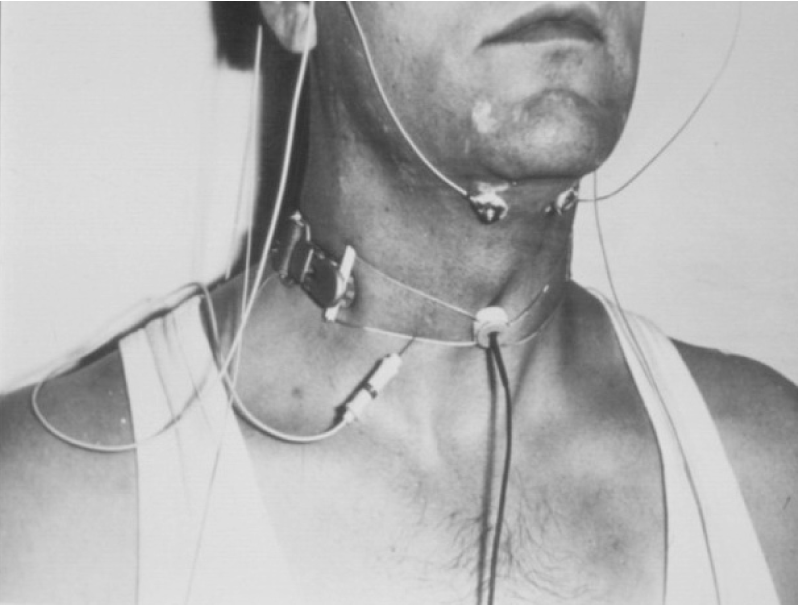
Figure 2 provides a basic demonstration of our electrophysiological method, which assesses oropharyngeal swallowing events and has been instrumental to our understanding of the nature of swallowing. First, the onset and duration of oropharyngeal swallowing are recorded from the suprahyoid muscles under the chin using surface electrodes. This method of registration is briefly called submental EMG (SM-EMG). The second vertical elevation of the larynx during a swallow is measured by a piezoelectric movement sensor (or laryngeal movement sensor) placed between the thyroid and cricoid cartilages at the midline and tied to the neck. Third, the cricopharyngeal sphincter muscle of the UES complex is reached percutaneously with a needle electrode, which is called cricopharyngeal EMG (CP-EMG) [53-57].
Cricopharyngeal EMG is recorded by a disposable concentric needle electrode that is inserted through the skin at the level of the cricoid cartilage approximately 1.5 cm lateral to its palpable lateral border in the postero-median direction. The sphincter recording is seldom necessary in our routine practice. All EMG records are always rectified and sometimes integrated and averaged. Because the perioral facial muscles are the first to be activated as the mouth closes before the pharyngeal phase, we often use the surface electrode placed bipolarly on the upper and lower lips near the corners of the mouth (not shown in Figure 2).
Our EMG methods can be used for three main purposes:
The first aim is to understand the pathogenesis of the oropharyngeal swallowing disorder in a patient or in a group of patients with the same disease etiology. This is accomplished by the “Single Bolus Analysis” [12,24,40,57,58]. The second aim is to diagnose and follow-up the dysphagic patients objectively and quantitatively. The “dysphagia limit” [55,59] and “SWS” [60-63] are the methods used to achieve this aim. The third aim is to identify spontaneous swallowing (SS) and distinguish it from voluntarily-induced swallows using polygraphic EMG methods. This can be important in the evaluation of oropharyngeal dysphagia in uncooperative patients, such as comatose, demented patients and in advanced cases of PD (for details of the single-bolus analysis and dysphagia limit methods, please refer to [52,55,59,63-66]).
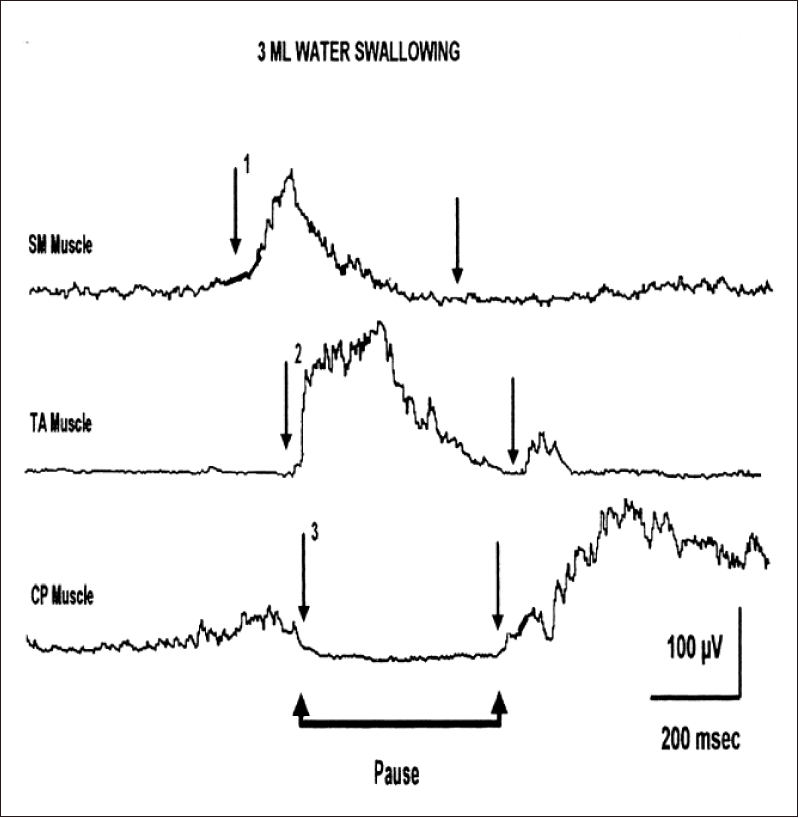
Figure 3 illustrates a sample recording of a single bolus analysis from a normal, healthy, adult participant. After the command of the examiner to “Swallow!” 3 mL of water, the following EMG traces appear. SM-EMG at the bottom trace begins approximately 300 msec earlier than the other traces. Subsequently, the laryngeal upward movement signal appears at the top trace (laryngeal movement sensor). The onset points of SM-EMG and the first upward laryngeal signal provide information about the motor aspect of the triggering time of swallowing reflex. The top trace shows the laryngeal movement signals demonstrating the upward and downward movement of the larynx. The time-span from the onset points of the first to the second laryngeal signals provides information about the timing of the laryngeal elevation and superior location. The larynx and airway are closed until the onset of the downward movement signal. After this second point, the larynx and airway are opened. Finally, the tonically active CP sphincter shown in the middle trace is relaxed and opened, and the bolus is transported into the esophagus.
Single bolus analysis. Three milliliters of water swallowed by a normal control. A: Laryngeal sensor signal. B: Cricopharyngeal sphincter muscle (CP-EMG). C: Submental EMG (SM-EMG). The EMG traces are the averages of five responses. Amplitude calibration: 100 μV and 20 μV in (B) and (C), respectively. Time calibration: 200 msec in all traces [52].
The laryngeal relocation time and laryngeal closure correlated well with the opening of the CP-sphincter or the so-called “CP-EMG pause”. This pause terminates just before or at the downward movement of the larynx. The bolus is transferred into the esophagus during the CP-EMG pause.
When we translate Figure 3 into the anatomo-functional basis of swallowing, the deflections could be expressed as follows: SM-EMG is generally composed of the mixture of EMG activities of mylohyoid, geniohyoid and anterior digastric muscles that fire concurrently to initiate a swallow and function as laryngeal elevators pulling the larynx upwards [5,38,47,52,55,64,67-69]. The rectified and integrated surface EMG activity of SM-EMG provides a considerable amount of information about the onset and duration of the pharyngeal phase.
Another important parameter related to the triggering of the reflex swallow is the interval between the onset of SM-EMG and the onset of the first deflection of the laryngeal sensor signal reflecting the upper movement of the larynx, which is one of the first events in pharyngeal reflex swallowing [5,38,39,67,70]. This interval can provide information about the temporal relationship between the instance of voluntary activation of the SM-muscle complex and of reflex triggering of the swallowing response [37,41,55,59]. Thus, triggering of the voluntary swallow, swallow duration and laryngeal closure time and pharyngeal duration can be easily measured using EMG techniques.
Laryngeal adductor muscle can also be investigated using EMG methods, which demonstrate that the larynx is closed during the pharyngeal phase of swallowing. The thyroarytenoid (TA) muscle of the vocal cord is the laryngeal adductor, and its contraction occurs with laryngeal closure, which is very important to protect the airway against pieces of bolus that escape during swallowing [64,70-75].
Figure 4 illustrates the intimate relationship between laryngeal and pharyngeal muscles during a single swallow of 3 mL of water. First, SM-EMG is activated and pulls up the larynx. This results in very dense EMG activity of the TA muscle and closure of the larynx. Concurrently, the CP-sphincter muscle of the UES complex is opened and closed during the swallow. The result is the fast transport of the bolus into the esophagus. One of the important events during the cascade of the swallowing reflex is the anterior-superior elevation of the larynx with closure of the vocal cords. As seen in Figure 4, the larynx and lower airways are protected from the swallowed bolus passing through the pharynx by closure of the vocal cords with the laryngeal adductor muscles, especially the TA muscle. This coordination between the laryngeal elevator and adductor muscles is clearly demonstrated using EMG methods [46,52,56,64,68,75-79].
SINGLE-BOLUS ANALYSIS IN PD PATIENTS
Oral phase swallowing abnormalities in PD include inefficient mastication, impaired bolus formation, hesitant deglutition and glottis tremor [3,10-12,80-85]. Other abnormalities of the oral stage of deglutition in PD, such as difficulty in bolus formation and delay in swallowing, have been suggested by many authors. The oral stage of deglutition abnormalities are strictly related to the pharyngeal phase of swallowing disorders, which are equally important and carry the potential risk of aspiration for the airway [12]. The pharyngeal phase of abnormalities in swallowing may not necessarily produce dysphagia symptoms unless laryngeal closure during swallowing becomes inadequate and the bolus enters the airway during and after a swallow [2,3,5,11,86]. The standard values for single-bolus analysis in normal subjects of different ages are shown in Table 1.
The symptoms and signs of dysphagia during the pharyngeal phase of swallowing often are not clinically recognized unless patients suffer overt clinical aspiration and choking during swallowing, which triggers the protective cough reflex. Unfortunately, airway aspiration can be silent without cough reflex, which can lead to aspiration pneumonia. Silent aspiration can be diagnosed by means of VFS and/or FEES [2,20,22,87-92], although silent aspiration and other abnormalities of pharyngeal phase of deglutition also are suggested by indirect EMG methods.
Figure 5 shows recordings obtained from three subjects [12]: a healthy subject, normal (A); a PD patient without dysphagia (B); and another PD patient with dysphagia (C). The triggering time of the swallowing reflex is shown by an arrow (A-0) and indicates the onset of SM-muscle activity (point A) and of the upward movement of the laryngeal sensor (Point 0) during swallowing of 3 mL of water. The triggering time of swallowing was delayed in two PD patients compared to the control subject. The mean laryngeal relocation time measured by the laryngeal piezoelectric signals also progressively increased from the control to PD without clinical dysphagia to PD with dysphagia. Prolongation of the laryngeal relocation time was associated with slowing of the pharyngeal reflex and was most pronounced in PD patients with dysphagia.

Single bolus analysis of Parkinson’s disease (PD) patients. Laryngeal sensor signals and integrated submental electromyogram (SM-EMG) obtained from a normal subject (A) and 2 PD patients with a normal dysphagia limit in (B) and with an abnormal dysphagia limit in (C) during water swallowing. The laryngeal relocation time (0–2 interval) and total duration of SM-EMG (A–C interval) are prolonged in the PD patient with dysphagia (arrows). All of the traces are the average of five responses [12].
Problems associated with the oral phase of swallowing in PD all seem to be related to akinesia, rigidity and tremor [3,9,11,12].
Akinesia is particularly important during simultaneous and sequential movements [93,94]. Consequently, there may be a deficiency in the transition from the voluntary tongue movements to the sequential, reflex movements of pharyngeal swallowing [55,85,93] similar to the deficits in other striated muscle movements [95]. Although the oral phase of swallowing is known to be under the control of the cerebral cortex, basal ganglia nuclei receive information from cortical areas [3,40]. Both the indirect influence of cortico-bulbar and extrapyramidal descending pathways might affect the oral phase in PD patients by manifesting inadequate bolus control in the mouth and tongue and delay of the swallowing reflex [2,3,12,81].
Prolongation of the SM-EMG and upward relocation of the larynx during the single-bolus assessment demonstrate two pathophysiological elements in the dysphagia of PD patients.
1) Slow and reduced laryngeal elevation and suspension as also detected by radiological methods [82,84,96-98].
2) Extreme prolongation of the pharyngeal stage of swallowing, which, together with the delay in triggering of reflex swallows, is highly specific to PD [12,55]. Delayed pharyngeal transit of the bolus has long been demonstrated by VFS (e.g.) [12,82-84,96,98].
These findings indicate that the electrophysiological abnormalities observed in PD are not directly associated with the mechanism of dysphagia but rather relate to the general pathophysiology of the disease. Such abnormal parameters should be regarded as risk factors for dysphagia and aspiration in PD. The abnormalities of CP-EMG and aspiration in PD will be discussed later under the subtitle “Cricopharyngeal sphincteric EMG”.
Some recent studies highlight new aspects in the pathophysiology of oropharyngeal dysphagia in PD. Pharyngeal muscles were found to display histopathological changes with evidence of chronic denervation and reinnervation in PD patients [99-103]. These pathological findings were caused by neurodegeneration of the motor branches of the pharyngeal nerves of PD patients, especially in dysphagic subjects. Synucleinopathy is also observed in cervical vagal nerve fibers [99,103]. These nerve fibers are mainly afferent in nature, and their Lewy body pathology was detected, especially in pharyngeal sensory nerve fibers [104]. In our experience, no EMG signs of chronic denervation could be detected by CP-EMG in PD subjects, although systematic comparison of EMG findings in PD patients and age-matched controls may reveal such degenerative changes in PD using qualitative and quantitative methods. The body of evidence presented in this subsection strongly suggests that the electromyographer should examine the oral and pharyngeal nerves and muscles in elderly normal and PD patients both with and without dysphagia.
DYSPHAGIA LIMIT AND PD
The methods used to evaluate the dysphagia limit were previously described [55,105]. During examination, the seated patient is instructed to hold his/her head in a neutral, upright position. To detect the vertical laryngeal movements during swallowing, a mechanical sensor that consists of a simple piezoelectric wafer with a 4 × 2.5-mm rubber bulge fixed at its center is placed on the skin between the thyroid and cricoid cartilage at the midline [106]. The sensor is taped onto the neck, and its output signal is bandpassed, filtered at 0.01 to 20 Hz, and fed into one channel of the EMG apparatus. Similar laryngeal sensors are commercially available worldwide.
Submental EMG activity is recorded by bipolar silver chloride electroencephalogram (EEG) electrodes taped under the chin, over the submental muscle complex (SM). The signal is bandpassed, filtered at 100 Hz to 10 kHz, amplified, rectified and sometimes integrated. The laryngeal sensor output shows two deflections during swallowing. The first deflection of the sensor represents the upper laryngeal movement. The leading or trailing edge of the first deflection is used to trigger the delay-line circuitry of the recording system so that all of the signals are time-locked to the same instant. This enables all of the electromechanical events to be displayed synchronously. Because SM-EMG activity coincides with laryngeal upward movement, this EMG activity is also time-locked to the laryngeal sensor signals [52,55,58,59,105,106].
Piecemeal deglutition refers to the division of the bolus into 2 or 3 successive swallows [107,108]. The dysphagia limit is the volume at which a second or more swallows becomes necessary to swallow the whole bolus [59]. In this method, the total sweep time of SMEMG is set at 10 seconds, and the delay-line is initiated at 2 seconds after the onset of the single sweep of the oscilloscope. Thus, after an amount of water is ingested, the effect of the bolus is followed for 8 seconds. Subjects are given 3, 5, 10, 15, and 20 mL of water in a stepwise manner, and they begin to swallow immediately after being instructed to do so by the examiner. They are asked to swallow all the liquid in a single effort, and the oscilloscope traces are started at the examiner’s order to “Swallow”. The laryngeal sensor signals and the activity of the SMEMG are recorded 2 seconds after the onset of each sweep. Any recurrence of two signals usually recorded at the beginning of the recording is accepted as piecemeal deglutition and a sign of dysphagia limit. Any duplication or multiplication of swallowing at or below 20 mL water is considered to be pathological, and the patient is considered to be dysphagic. The volume of water below 20 mL at which “Piecemeal Deglutition” [5,6,107] is observed is called the “Dysphagia Limit” (Figure 6) [55,59,105].

Dysphagia limit (DL). (A) is a normal adult subject, and DL is normal. (B) is a patient with dysphagia with stroke. DL is 10 mL and pathological. (C) is an amyotrophic lateral sclerosis (ALS) patient without dysphagia, and DL is normal. (D) is an ALS patient with dysphagia, and DL is 5 mL and pathological [55,65].
The examination is stopped if the patient shows any signs of subglottic aspiration, such as coughing or a wet voice. If there is any suspicion of piecemeal deglutition and/or recurrence of swallowing in the first attempt, the same procedure is repeated two or three times with the same volume of water.
The dysphagia limit seems to be a more sensitive test compared with the other 2 water swallowing tests (i.e., 3-ounce test and timed test). These two swallowing tests are fundamentally different from the dysphagia limit with respect to the physiological process they measure. The dysphagia limit tests a precise physiological phenomenon to assess voluntary swallowing and is therefore objective and quantifiable. It is also more comfortable and likely also safer, especially for older or debilitated patients, as compared with the two other swallow tests. The process measured by the Dysphagia limit approach is a single cortical drive that leads to the motor behavior of the discrete voluntary swallow via the “CPG” at the medullary level. Any disturbance within the system can be demonstrated by the fragmentation of a single swallow below the 20 mL water volume. In contrast, the other two swallowing tests measure voluntary SWS of a considerable bolus volume (90– 150 mL) [108] as fast as possible and without interruption. In this type of deglutition, the initiation of swallowing is also voluntary, but its timing, shaping and rhythmicity are mostly provided by the CPG of the medullary swallowing center [36,37,48,60]. Thus, these two swallowing tests likely measure different aspects of oropharyngeal swallowing compared to the “Dysphagia Limit”.
Because the dysphagia limit assesses a voluntary, single/discrete swallowing event and is more strictly controlled by connections between the cerebral cortex and oropharynx via the corticobulbar tract, brainstem CPG does not seem to be as critically involved in its control compared to that of SWS, which will be discussed below.
Dysphagia limit abnormalities can arise from dysfunction of structures along the cortex-corticobulbar tract-oropharyngeal muscle axis as follows:
1) Insufficient sensory information from the oral cavity can result in inadequate evaluation of the bolus at the level of central nervous system, which likely results in the division of a small bolus (< 20 mL water) into two or more swallows instead of a single normal swallow. This is most likely a compensatory or protective mechanism triggered by the lack of necessary and adequate information about the bolus [40,41,109]. The sensory abnormalities of the oral cavity or pharyngeal mucosae can result in abnormal findings in the dysphagia limit [109].
2) Motor dysfunction of the tongue due to central and/or peripheral causes is another important reason for an abnormal dysphagia limit and dysphagia [55]. This results from the inability of the tongue to function normally in the formation of the bolus and its transit and pumping from the mouth to the pharynx (Figure 6).
Figure 7 compares the dysphagia limits of 2 patients with PD: one nondysphagic (Patient-1) and the other dysphagic (Patient-2). It can be easily observed that in the PD patient without dysphagia, the dysphagia limit was normal. However, in the dysphagic patient, the dysphagia limit was 10 mL of water, which is pathological [12]. When the dysphagia limit is evaluated, there are three important facts for PD:

Dysphagia limit and patient with Parkinson’s disease (PD). Note that the water volume swallowed at one time can go up to 20 mL of water in a PD patient without dysphagia (A), whereas in a PD patient with dysphagia (B), the bolus is divided into two or more separate swallows during swallowing of 10–20 mL of water. Arrows denote the second and subsequent swallows [12].
1) In PD patients without clinical dysphagia, the dysphagia limit may be less than 20 mL water. These patients likely have subclinical dysphagia and should be followed clinically and electrophysiologically for oropharyngeal swallowing [11,12,110-112].
2) Many PD patients with dysphagia and low dysphagia limits also have clinically significant oropharyngeal dysfunction, such as wet voice and/or coughing after water swallowing and the accumulation of saliva and pieces of food in the mouth. These subjects have a more advanced clinical condition and longer disease duration.
3) Although the role of dopaminergic treatment for dysphagic PD patients has long been disputed [112,113], dopaminergic treatment during the first year after PD diagnosis can improve dysphagia [114-116]. These reports are in agreement with our experience that in newly diagnosed patients without prior treatment, dopaminergic treatment can improve subclinical and/or clinical dysphagia. However, the majority of the patients with PD treated previously with dopaminergic drugs do not show improvement of their dysphagia [8,115,117].
A pathological dysphagia limit in PD can be related to sensory-motor impairment, although less attention has been given to the contributions of sensory deficits in the airway towards dysphagia [118]. In contrast, the assessment of airway protection is not complete without evaluating laryngo-pharyngeal sensation [2]. Diminished sensation, whether chemical or mechanical, has been shown to contribute to the development of aspiration pneumonia [89,119]. For normal swallowing, the cough reflex should be intact, and irritation by the material escaping from the pharynx to the airway should activate the cough reflex. For normal sensation of irritation due to the escaped bolus, the afferent side of the airway, including the laryngeal and pharyngeal mucosae, should be intact, such that any escaped food material is thrown outside the airway by the cough reflex. If pharyngolaryngeal sensation is reduced, then the cough reflex threshold may increase, and silent aspiration may occur in any neurological disease [87,92,120]. Ebihara et al [120]. found that in the early stage of PD, motor control was impaired with preservation of sensory aspects. However, in later stages of disease, both motor and sensory components of the pharyngo-laryngeal region become less sensitive, producing silent laryngeal penetration that eventually led to aspiration pneumonia. Sensory impairments in PD have not been completely resolved; however, it is clear that this type of pathology may lead to lower dysphagia limits and sometimes result in aspiration.
However, the motor aspect of the oral phase of swallowing seems to be more involved in PD. Abnormally low dysphagia limits are due to insufficiency of bolus formation in the oral cavity and are not a cause of aspiration, especially during the early period of PD. Piecemeal deglutition is likely not only a pathological finding but also a potential compensatory mechanism in the presence of intraoral pathology. As a practical matter, when an abnormally low dysphagia limit is observed, it should be viewed as a potential precursor to aspiration. Such patients should at a minimum undergo an oropharyngeal rehabilitation program as soon as possible [15,39].
SEQUENTIAL WATER SWALLOWING AND PD
Continuous sequential swallowing from a cup or straw or SWS in humans has recently been recognized as a more physiologically appropriate approach to evaluate the physiology of deglutition and the diagnosis of dysphagia. SWS is a common daily occurrence characterized by multiple, successive, rapid swallows [60,61,88,121]. SWS is technically similar to the “3 Ounce test” and timed test but these two swallowing tests are performed by clinical observation during which 90–150 mL of water should be swallowed as fast as possible and without interruption [26,30,122,123]. SWS is assessed during either the collection of radiological [61,62] or physiological recordings [60,63,124]. During SWS, there are rhythmic or pseudorhythmic swallowing sequences without any clinical pause. When recordings of SWS are collected while using a nasal airway sensor for respiration and SM-EMG, we can observe approximately 1-second inter-swallow intervals between each couple of swallow deflections in SM-EMG, during which the respiratory recordings demonstrate breath-holding that results in a swallow apnea period in normal subjects.
The advantage of recording SWS is to closely and objectively follow the relationship between swallowing and breathing. This approach is also more advantageous for evaluating aspiration problems because after the first swallow in SWS, subsequent swallows are primarily initiated at the hypopharynx in normal subjects, who are the most prone to producing laryngeal penetration [61,62]. When the water volume is increased for SWS, laryngeal penetration or aspiration frequently occurs [71,88,125]. There are also some reports of an effect of aging on SWS with laryngeal penetration occurring more frequently in older adults [62,78]. This may be another contributing factor to dysphagia in PD, as most PD patients are above 50 years of age.
ELECTROPHYSIOLOGICAL INVESTIGATION METHODS OF SWS
1) Swallowing signals are recorded from the submental muscles with SM-EMG using silver-silver chloride (Ag-AgCl) EEG electrodes taped under the chin over the SM. Signals are filtered using a bandpass of 100 Hz–10 kHz, amplified, rectified and integrated. The total analysis time is adjusted to 20 seconds for each set of SWS.
2) Respiration is usually studied in another channel of the EMG apparatus on a 2 or 4-channel EMG. The respiratory signal is obtained via a nasal cannula that is placed at the entry of the nostrils and connected to the EMG machine. The direction of airflow is recorded as a negative polarity representative of inspiration and a positive polarity representative of expiration.
Submental EMG and respiration are simultaneously recorded before, during and after 50–100 mL of water swallowing from a cup. EMG filters for respiration channels are adjusted with a band pass of 0.2–30 Hz.
After the command to “Swallow”, normal adult subjects perform swallowing movements recorded from SM muscles that contract in a rhythmic fashion, and an apneic period is also registered during SWS. As shown in Figure 8, six swallows were sufficient to drink 100 mL of water from a cup. During sequential swallowing, breathing ceases, as seen in the swallowing apnea period (Trace 1-A and 3-A with different sweep speeds in Figure 8). During SWS, the swallows are rhythmically deflected on the trace (see inter-swallow intervals at 4-B in Figure 8), although the last inter-swallow interval was prolonged. Swallowing apnea is often initiated by inspiration/expiration, but the end of swallowing apnea is most frequently completed by expiration (Trace 3-B in Figure 8). The number of swallows and duration of SWS increased significantly with increasing water volume from 50 to 100 mL (Table 2).
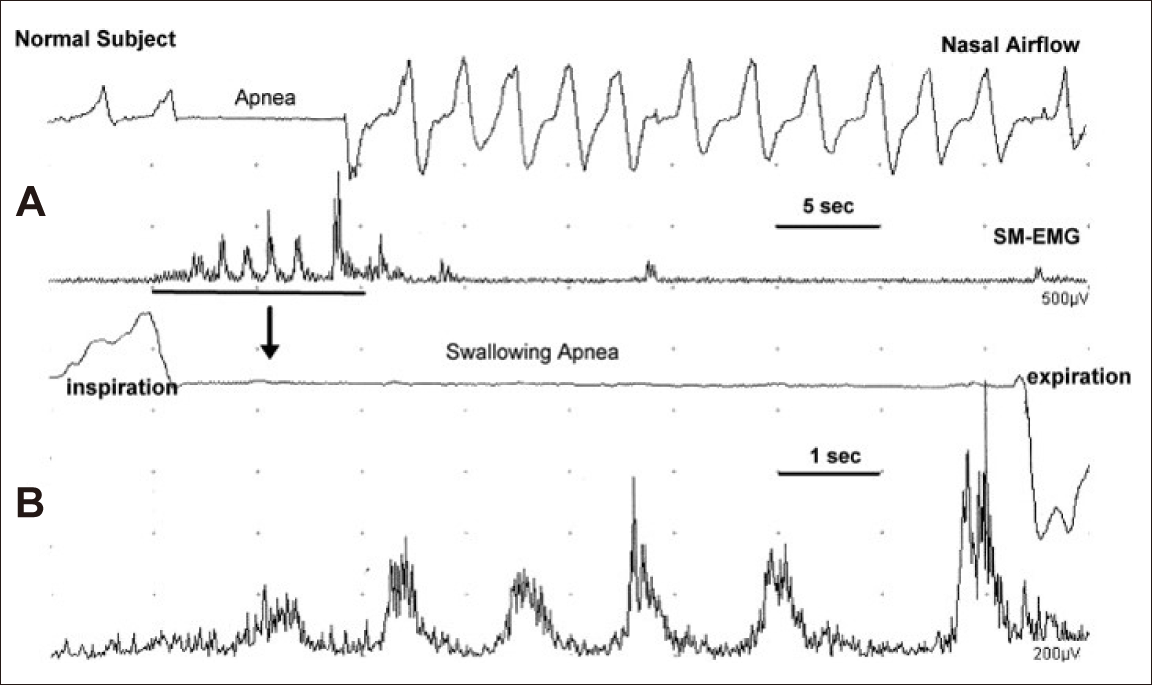
Sequential water swallowing (SWS). Nasal airflow signals (upper traces in A and B) and integrated submental electromyogram (SM-EMG) (lower traces in A and B) obtained from a normal subject during SWS of 100 mL of water. The 3rd and 4th traces were obtained from the first two traces using the window technique with a 10-second sweep time. Interruption of the nasal airflow signal that was consistent with the deglutitive apnea was covered during SWS. Swallowing apnea began at inspiration and ended at expiration during SWS [60].
The SM-EMG pattern of SWS of either 50 or 100 mL water volumes is regular and rhythmic deflections with similar inter-swallow intervals. However, the amplitude of the EMG of SM muscles varies among successive swallows with the trend of crescendo or crescendo/decrescendo amplitudes during SWS; the most recent swallows have longer interswallow intervals than those that occur the first raw sequential swallows. The majority of the normal controls show very late swallows, usually 5 seconds after the end of SWS.
The SWS pattern and apnea period are not clearly altered when SWS is repeated 2–3 times intra-individually in normal subjects who perform at almost the same speed of drinking.
Another interesting phenomenon encountered at the initiation of SWS in normal subjects is that the first SM-EMG burst shows some different features than those following swallow deflection in SMEMG. These initial SM-EMG bursts occur after the command of “Swallow”, and they almost always appear just before or during the last respiratory cycles and before the swallowing apnea. They are often smaller in amplitude than subsequent swallow deflections and are sometimes longer in duration then continued swallowing (Figure 9). We termed these bursts “Foreburst of SWS”, which are found in 86% of the normal subjects both with 50 and 100 mL water swallowing [63].
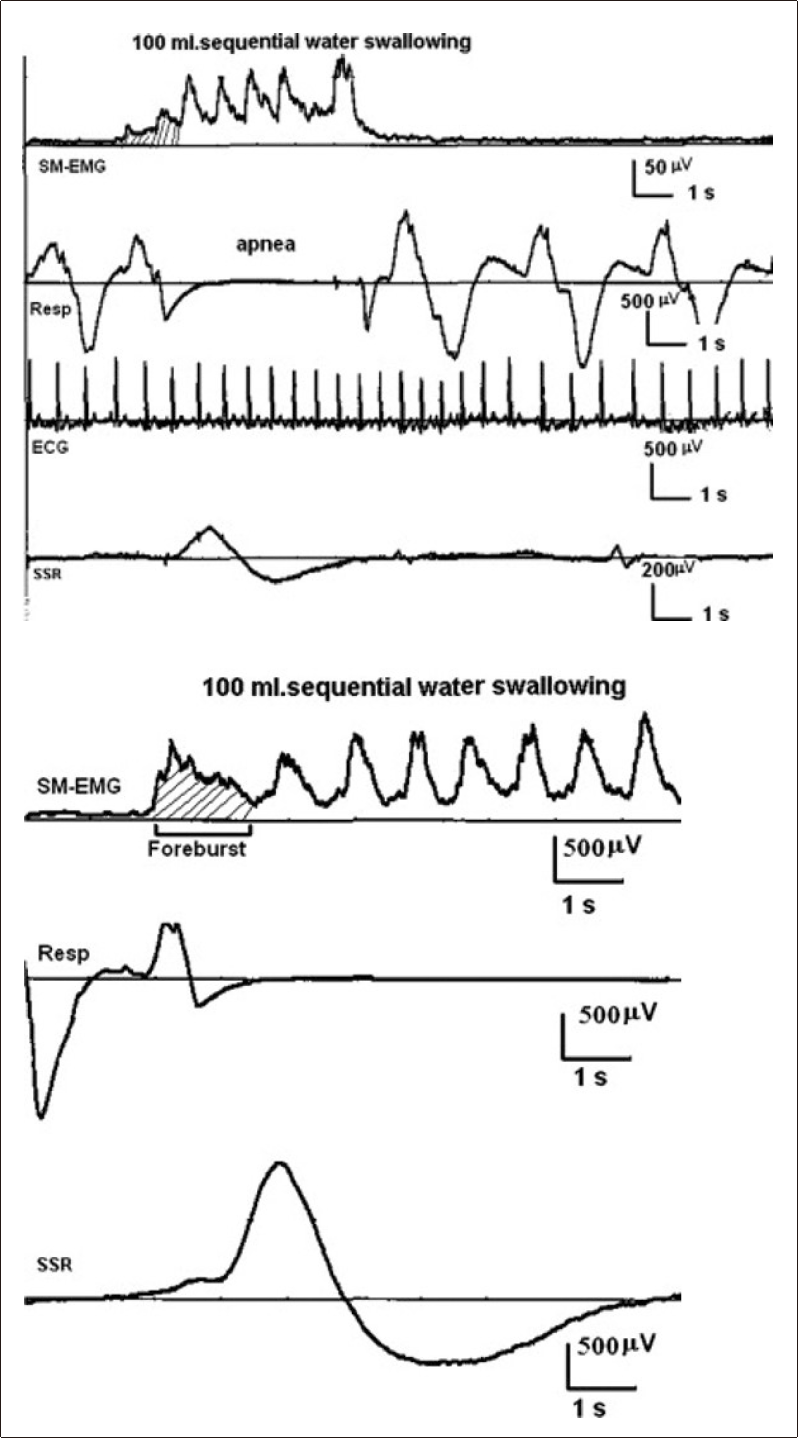
Sequential water swallowing (SWS) with foreburst. Upper four traces: SWS from a normal adult subject. The first channel is the submental electromyogram (SM-EMG), the second is the nasal cannula, the third is the electrocardiogram and the fourth is the electrodermal activity. The “Foreburst” occurs with the last respiratory cycle before the apnea interval. Lower three traces: SWS from another normal adult subject. The foreburst (lined) occurs concurrently with the last respiratory activity, and the evoked skin potentials appears slightly later in two cases [63].
The swallowing apnea period depends on the duration of SM-EMG of SWS. It starts and ends with expiration and expiration in approximately 67% of normal controls. While the expiration/inspiration pattern is found in approximately 33% of normal human subjects, the expiration/expiration pattern increases with aging, which suggests that this is most likely a type of protective mechanism against aspiration.
Another important observation during SWS is the “Compensatory Respiration Cycle” (inspiration/expiration) during the swallowing apnea period while participants are sequentially swallowing (Figure 10). It is not uncommon to record the compensatory respiratory cycle between the last inter-swallow interval of SWS. The last inter-swallow interval during SWS is slightly longer compared to the first swallow intervals. Compensatory respiration can also be recorded in the midst of SWS during swallowing apnea. Another feature of compensatory respiration is that the onset is mostly inspiration and followed immediately with expiration. The amplitudes of compensatory respiration events are often higher than those of the non-swallowing period. The compensatory cycle of respiration during SWS usually occurs in many normal controls with increasing frequency as the water volumes increase. However, compensatory respiratory cycles during the apnea period are generally recorded only once. In aging, the SWS duration and swallowing apnea are more prolonged than in the young-middle-aged group, which is also an important point for patients with PD.
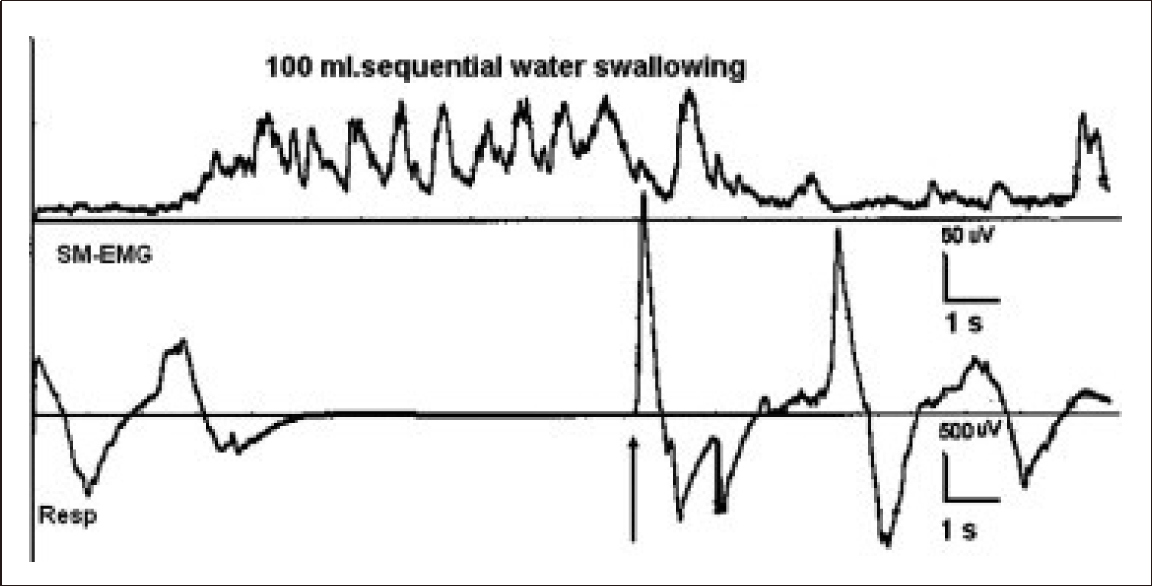
Sequential water swallowing (SWS) with 100 mL water. First channel: submental electromyogram (SM-EMG); second channel respiration and swallowing apnea from a normal subject. The compensatory respiratory cycle occurs in between the last swallows during the apnea period (arrow) [63].
In PD, all SWS parameters are expected to deviate from normal controls. The number of swallows and the duration of SWS are significantly increased in PD patients (Figure 11). In dysphagic PD patients, the number of swallow movements are increased, and the swallowing volume capacity is significantly decreased when compared with non-dysphagic PD subjects [60]. One of the important features of patients with PD is that inter-swallow intervals are considerably regular and rhythmic in the majority of cases, and only 7% of PD patients display an arrhythmic pattern. On the whole, PD patients with or without dysphagia complete the SWS by taking a longer time with more swallows than normal subjects. Some PD patients display a different SWS pattern of machine-like, regular sequences of swallows throughout the SM-EMG recording. The typical parkinsonian gait is slow with small regular steps; the same type of rhythmicity is also observed with the tongue movements. It has been demonstrated that in PD, the tongue muscles lose versatility and the movement pattern sometimes becomes rhythmic [8,12,81,86]. Apart from this abnormal pattern, inter-swallow intervals in PD do not differ significantly from those of normal subjects. The SWS patterns of swallowing and their values are not commensurate to the extent of swallowing problems in PD patients [60]. However, the coordination between swallowing and breathing has not been properly studied using electrophysiological SWS methods.
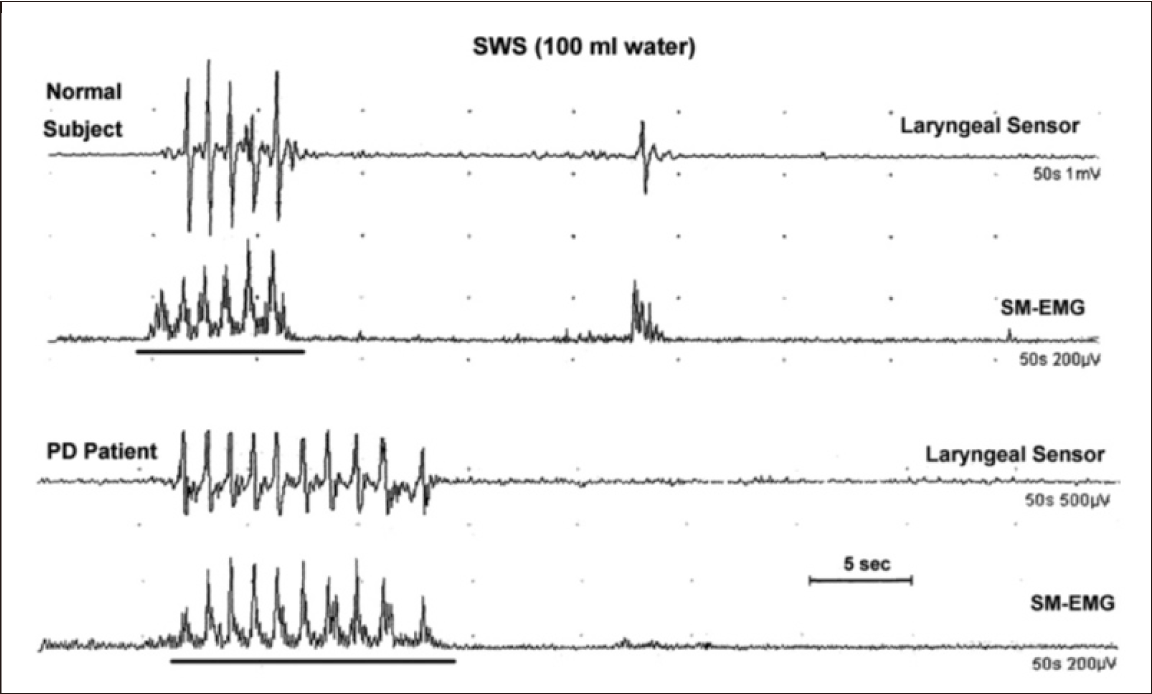
Sequential water swallowing (SWS) and Parkinson’s disease (PD). The number of swallows and duration of SWS are prominently increased in the patient with PD. Inter-swallow intervals are considerably regular and rhythmic during 100 mL of SWS [60].
Experimentally long-lasting, repetitive electrical stimulation of the superior laryngeal nerve can elicit a pattern of “Rhythmic” motor activities of swallowing [126-129]. The sequential and rhythmic patterns of swallowing are formed and organized by a CPG [40,66, 127,130-132]. In SWS, hypopharyngeal stimulation of the bolus sequences initiates the sensory input to the NTS, at first via a shorter afferent circuit, and then up to the cerebral cortex [36]. Subsequent swallows can be mostly controlled by the dorsal swallowing group (DSG) of neurons situated at the NTS. The DSG neurons fire a sequential and rhythmic pattern parallel to the motor pattern of oropharyngeal swallowing [127,132,133]. The sequential and/or rhythmic swallowing is operated by the CPG at the brainstem [126-128,130,134]. Lack of coordination between swallowing and respiration can be expected in severe dysphagic cases of PD. In such cases, swallowing is too long with several deflections, and the swallowing apnea period is frequently divided by 4–5 compensatory respiratory cycles. PD patients with this pattern of SWS likely have silent aspiration because there is no coughing despite severe abnormalities in the coordination of swallowing and respiration.
EVALUATION OF ELECTROMYOGRAPHY OF THE HUMAN CRICOPHARYNGEAL MUSCLE OF THE UPPER ESOPHAGEAL SPHINCTER
The CP is a striated muscle sphincter situated at the pharyngo-esophageal junction. The UES is composed of the striated CP muscle and nonmuscular components at the level of the cricoid cartilage. The major component of the UES is the CP muscle, which is attached to the lateral margins of the posterior aspect of the cricoid cartilage and encircles the lumen through the inferior pharyngeal constrictor. The striated muscles of the proximal esophagus may also contribute to the UES [77].
The CP sphincter muscle is tonically active at rest and relaxes during swallowing. Its main function is to control the anterograde and retrograde flow of contents between the pharynx and esophagus [69]. Following the important contributions of Doty and Bosma [42], EMG of the CP sphincter muscle has been studied mostly in a variety of non-human species to understand deglutition; however, EMG of the CPsphincter has been considerably less well studied in healthy humans and dysphagic patients [52-55,77,135-137].
The CP-sphincter muscle consists of the upper oblique (pars oblique) and lower horizontal (pars fundiformis) regions. The CP-muscle attaches to the cricoid cartilage and forms a C-shape muscle band. A concentric needle electrode is most likely inserted into the lower, horizontal part of the CP-muscle. The CP-muscle has dual innervations by the pharyngeal plexus of the vagus nerve and recurrent laryngeal nerve [101,138]. Motor neurons controlling the CP-muscle are found primarily within the semicompact and rostral compact portions of the NA, and the innervations are mostly ipsilateral [38,77].
The easiest way to approach the CP-muscle is the percutaneous approach. A concentric needle electrode is passed through the skin in a posterior and medial direction at the level of and just lateral to the cricoid cartilage. The point of insertion is approximately 1.5 cm lateral to the palpable cricoid cartilage. The needle electrode should be inserted while the subject is in a recumbent position with the head tilted posteriorly to prevent the rare possibility of arterial hypotension or syncope. After approximately 1 minute, the recording can be commenced. At any time during this process, if coughing occurs or the patient experiences an unpleasant feeling of pain, the needle electrode is removed. In our hands, this procedure has not led to any major complications or patient discomfort [52,77]. In a few subjects, a small subcutaneous hematoma occurred, as can also be seen in routine needle EMG applied to limb muscles.
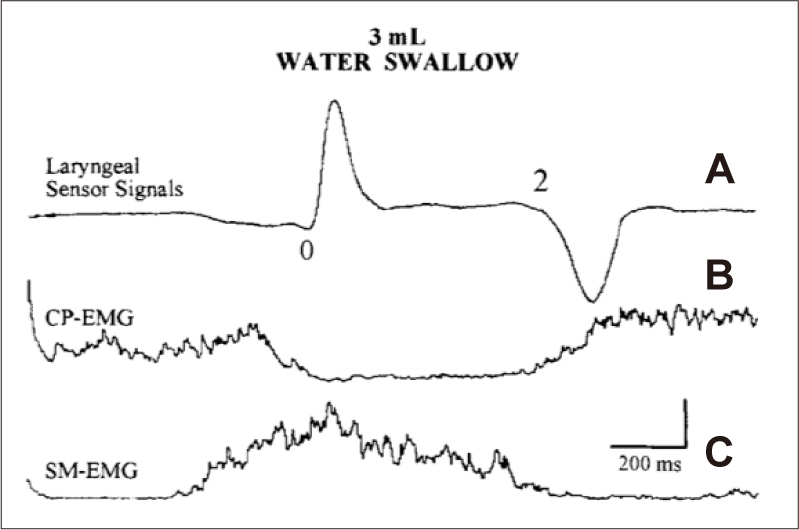
Figure 12 summarizes the EMG recording method for the CP sphincter (CP-EMG) and its relationship with SM-EMG. Normal participants swallowed 3 mL of water. During rest, there was continuous, high-frequency tonic EMG activity in the CP muscle (third trace). During swallowing, SM-EMG activity began first (first trace), and the tonic activity of the CP-muscle was switched off (pause in the second trace).
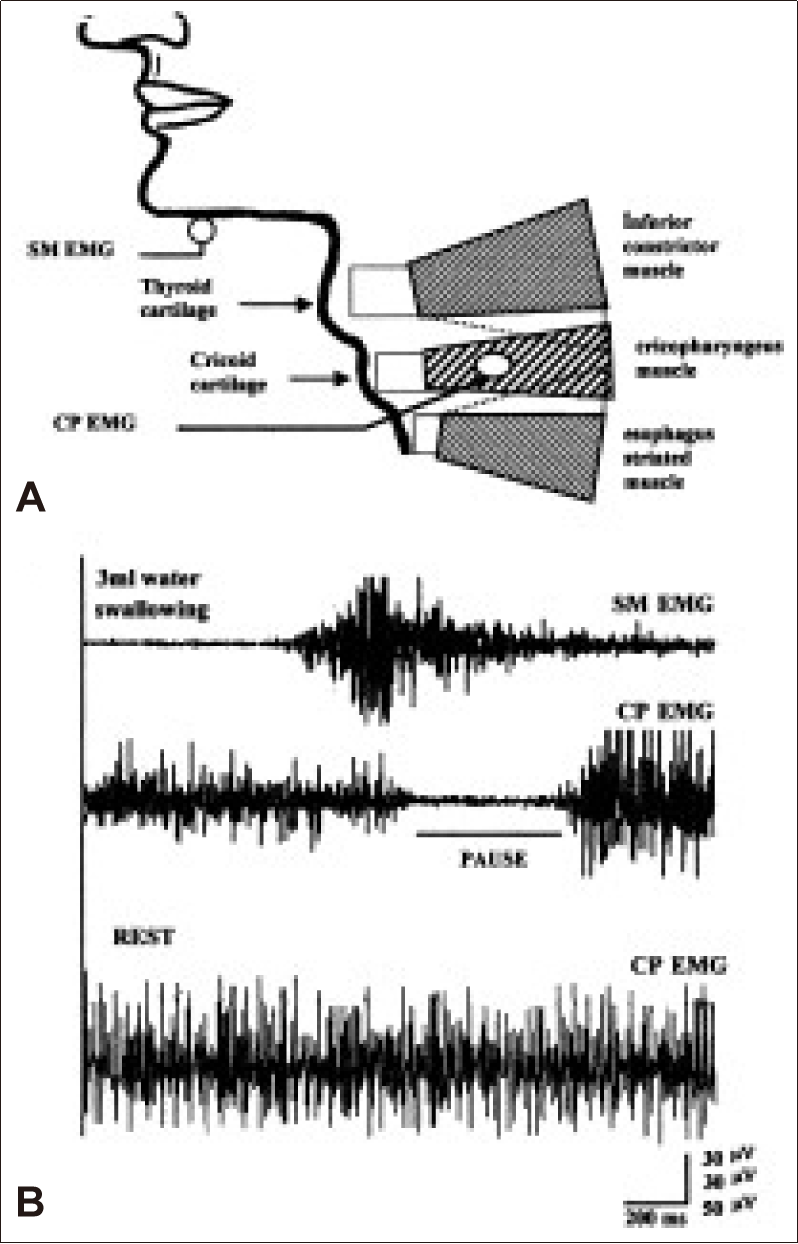
A: Schematic diagram showing electromyogram recording method for cricopharyngeal sphincter (CP-EMG) and submental muscles (SM-EMG). B: During rest, there is continuously high frequency tonic EMG activity in the CP muscle (third trace). During 3 mL of water swallowing, SM-EMG activity begins first, and the tonic activity of the CP muscle is switched off (pause) (first and second traces). A conventional EMG recording was used [77].
High-frequency tonic EMG activity appears on the oscilloscope screen, and a continuous high-frequency sound is emitted from the loudspeaker as the needle electrode tip penetrates the CP-muscle. During attempted swallowing, tonic EMG activity disappears for a short period of 400–600 msec in normal subjects. This CP-EMG pause during the swallow and the increase in tonic activity that often occurs prior to and always occurs following this pause is an indication of correct electrode entry into the CP-muscle [55,64,77]. The EMG signal can be conveniently recorded using a bandpass filtered between 100 Hz and 10 kHz, rectified, integrated and sometimes averaged during dynamic swallowing studies. The needle tip records the activity of the most lateral part of the pars fundiformis of the CP sphincter muscle. The entry path into the CP-muscle can sometimes differ, especially in obese, elderly and short-necked subjects. The depth of entry of the concentric needle into the CP-muscle is approximately 2–4 cm from the skin in most people, but a deeper approach is sometimes necessary. If the needle tip is unintentionally introduced into the neighboring muscles, then the swallowing pattern with the EMG pause is not obtained. If the laryngeal muscles are penetrated accidentally, then the EMG activity is mainly increased by phonation or forced breathing [77]. A hook wire electrode can also be used instead of the concentric needle electrode [46,47].
The principle CP-EMG finding is that the CP-sphincter muscle is tonically active at rest, and this continuous activity ceases during swallowing in human subjects. The CP-muscle maintains a continuous but varying basal tone that depends on the environmental conditions [53,54,135,138]. Numerous reflexes contribute to the tonic activity of the CP muscle. For example, pharyngeal stimulation is found to cause an increase in the UES tone and CP muscle EMG activity [139-144].
Two groups of motor units are present in the human CP-muscle. Small motor units are continuously discharged at rest, and larger motor units are recruited just before and after the CP-EMG pause during swallowing (Figure 13). These EMG findings are consistent with the histopathological findings for the CP-muscle of two groups of muscle fibers, i.e., type-I oxidative fibers, which constitute the majority of the muscle fibers, and type-II glycolytic muscle fibers [145,146]. These histochemical and morphometric characteristics of the human CP-muscle make it clearly adaptable to the physiological features of two types of motor units, i.e., small motor units that are continuously discharging at rest, and large motor units that discharge transiently just before and after deglutition.

Motor unit action potentials of the cricopharyngeal (CP) sphincter muscle. A: Tonic electromyogram (EMG) activity. B: Foreburst activity. C: Rebound burst activity. Note the increase in duration, amplitude and frequency of the motor units during the foreburst and rebound bursts before and after the CP-EMG pause recorded from a window, as demonstrated in the uppermost trace [77].
At rest, the tonic EMG activity of CP-muscle is such that the motor units are small and range from 1 to 10 msec in duration (mean: 4.9 msec) and between 50–120 μV in amplitude. They are often bi- or triphasic. It is difficult to determine the discharge frequency for a given motor unit due to technical and physiological factors.
During wet or dry swallowing, two bursts of increased EMG activity are clearly observed just before and after the CP-EMG pause. The motor units in the rebound burst are large, and biphasic or triphasic motor unit potentials may also be recorded in the initial burst. We postulate that the foreburst is a type of protective reflex that is strictly related to oropharyngeal function and does not necessarily take part in the sequential muscle activity of deglutition, whereas the rebound activity is an electrical event that is strictly bounded by the sequential muscle activity of the CPG [40,77]. The foreburst is recorded in approximately 70% of normal, adult subjects in contrast to the rebound burst, which is recorded in all subjects [77]. Reflex responses similar to those of CP are observed in the EMG of the laryngeal adductor muscles during swallowing as an oral-laryngeal reflex that precedes the contrast excitation of the laryngeal adductor, which closes the larynx [64,71,76].
Complete bioelectrical silence or CP-EMG pause is recorded in most normal subjects. However, in 36% of normal controls, the pause is not bioelectrically silent and there are some active motor units within the CP-EMG pause [40,64,77]. In such subjects, the CP-EMG pause sometimes displays two stages: in the first part of the pause, the bioelectric silence is obvious and electrical potentials higher than 20 μV are never observed except for mechanical artifacts. In the later part of the pause, some small motor unit potentials exceeding 20 μV can be seen. This EMG activity eventually increases in amplitude toward the onset of the rebound burst. These two stages are termed as the absolute and relative CP-EMG pauses, respectively (Figure 14). The absolute period occurs within the first 100–200 msec and constitutes approximately 40% of the CP-EMG pause during swallowing. The relative pause then follows and constitutes over 50% of the total pause. These normal variations are very important for the evaluation of patients with neurogenic dysphagia because some patients with cortico-bulbar [58,65,77] or extrapyramidal involvement, such as in Steele-Richardson-Olszewski syndrome and multiple system atrophy, can be differentiated from idiopathic PD based on their CP-EMG pause abnormalities [147].
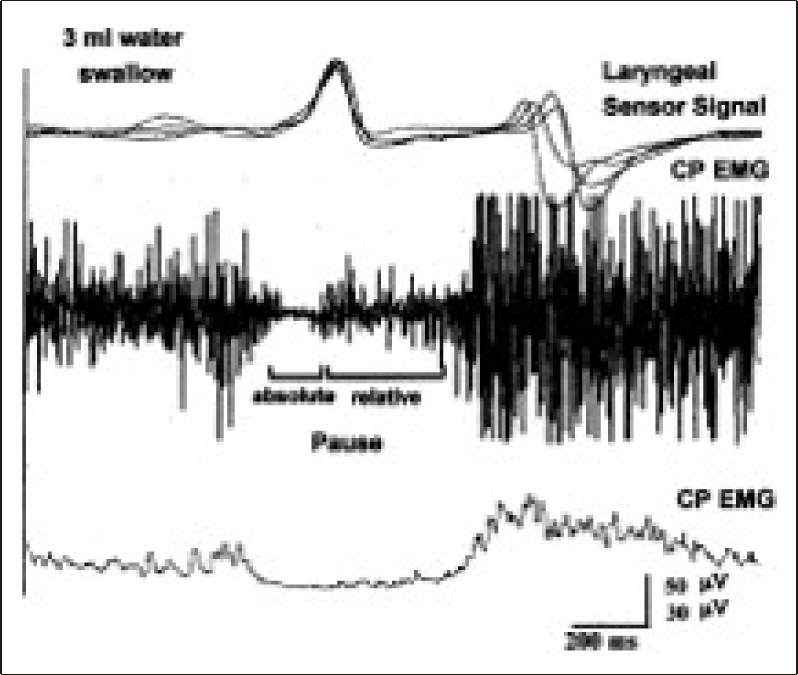
Laryngeal sensor signals and electromyogram (EMG) activities of the cricopharyngeal (CP) muscle during swallowing of 3 mL of water. Two stages of the CP-EMG pause (absolute and relative) are shown with conventional and integrated/averaged CP-EMG traces [77].
CP-SPHINCTER EMG IN PATIENTS WITH NEUROGENIC DYSPHAGIA AND EXTRA-PYRAMIDAL SYSTEM DISORDERS
Cricopharyngeus-sphincter EMG displays abnormalities in a number of diseases with neurogenic dysphagia.
“Hyperreflexic CP-EMG” is a type of EMG abnormality of the CP-sphincter that is mostly encountered in motor neuron diseases, i.e., amyotrophic lateral sclerosis (ALS) [55,58,65], suprabulbar palsy with lacunar infarct [65,148], and cranio-cervical dystonia [149]. Figure 15 illustrates the features of hyperreflexic CPEMG in patients with corticobulbar involvement. The duration of the CP-EMG pause is shorter in a patient with ALS (Figure 15B) than in a normal subject (Figure 15A). The CP-EMG pause ends prematurely before the larynx descends from the superior position (oblique arrow in Figure 15B). The unexpected burst of motor unit action potentials appear during the swallowing pause of CP-muscle. These changes prevent the transfer of the whole bolus from the pharynx into the esophagus, leading to food remnants in the pharyngeal spaces. The relationship between the laryngeal relocation time and the opening of the UES has been demonstrated clearly with different bolus volumes in normal subjects [105]. The correlation of the laryngeal elevation and relocation time with the opening of the CP sphincter disappears during voluntary swallows. As a result of the premature closure of the CP sphincter that occurs before the larynx descends from its upward position, the bolus is retained in the pharyngeal spaces. Subsequently, after the larynx has descended from its upper location, the bolus escapes into the airway, resulting in laryngeal penetration and even lower airway aspiration. The hyperreflexic CP sphincter is caused by the cortico-bulbar involvement in motor neuron diseases, such as ALS. This type of abnormality is likely a result of upper motor neuron involvement, which prevents the inhibitory influence on the UES, rather than any pathology of the lower motor neurons [55,58,64,65,77].

Hyperreflexic cricopharyngeal (CP) sphincter electromyogram (EMG). Two amyotrophic lateral sclerosis (ALS) patients with dysphagia (B and C). During swallowing, the duration of the CP-EMG pause is shorter, and the CP-EMG pause ends prematurely before the larynx descends from its superior position (in B). The unexpected burst of motor units during the CP-EMG pause of the CP sphincter is clearly recorded (in C) (see the arrows and compare them with the normal control shown in A). The last trace is the result of conventional EMG [65].
Electrophysiological investigations were reported in PD patients for the CP sphincter muscle [12]. During oropharyngeal swallowing in PD, the CP sphincter closed considerably earlier than the onset of downward movement of the larynx in 29% of the investigated PD patients. This finding is attributed to the delay in the onset of downward movement of the larynx (Figure 16). The mean duration of the CP-EMG pause is not significantly different in PD patients (444.8 ± 19.3 msec) compared with normal subjects (482.7 ± 27.1 msec). An increased duration of CP-EMG pause is rare in patients with PD. Thus, the CP-EMG pause, which indicates the relaxation and opening of the UES during swallowing, does not correlate with the clinical symptoms and the values for dysphagia limits in PD.
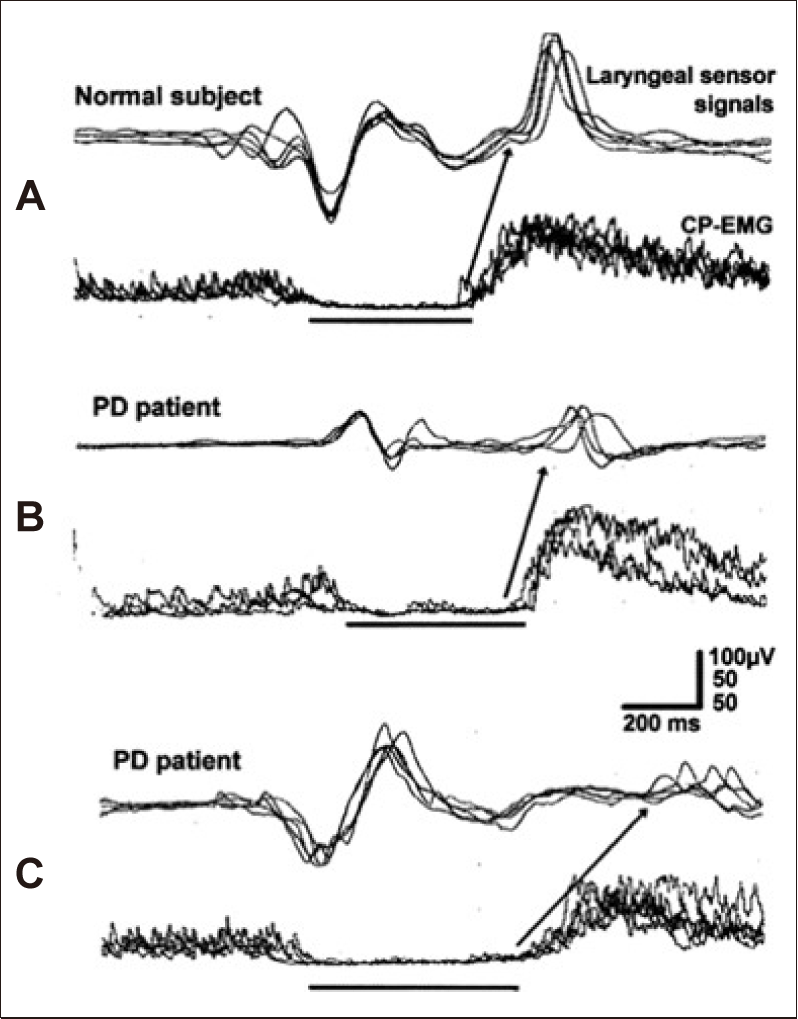
Cricopharyngeal (CP)-sphincter electromyogram (EMG) and patients with Parkinson’s disease (PD). In one of the two patients with PD but without dysphagia, the CP sphincter is normal (A), but in the second PD patient with dysphagia, the CP-EMG pause and the pharyngeal phase of the submental EMG are prolonged (B and C) [12].
Cricopharyngeus muscle dysfunction in PD has been the subject of considerable debate. Some studies suggested that CP dysfunction is the major abnormality in PD-related dysphagia [82,96,100,150], whereas others failed to describe any significant CP dysfunction [80,147,151].
In PD, a hyperreflexic CP sphincter muscle is not an electrophysiological abnormality, unlike in conditions such as ALS or pseudobulbar palsy with lacunar stroke as discussed above. In contrast, a hyperreflexic CP-sphincter pathology can be observed in cortico-bulbar palsy [55,58]. This distinction between PD and PD plus syndromes, such as multiple system atrophy and progressive supranuclear palsy [147], which show clinical signs of corticobulbar tract involvement [152,153], is helpful in differentiating these conditions. Additionally, in the Parkinson’s plus syndromes, early symptoms of dysphagia are common [154-156]. Alfonsi et al. [147] found that in these syndromes, the CP-EMG pause is not silent, and some unexpected motor units occur during the silent phase of the CP-EMG pause. It should be noted, however, that some normal subjects may also present motor unit potentials during the CP-EMG pause, although these signals display a small amplitude rather than tonic activity.
The last point of discussion of the CP-sphincter muscle is related to the sequential muscle activation of CPG [38,40,42,51,127]. As previously discussed, one of the sequential muscle activation events is the anteriorsuperior elevation of the larynx with closure of the vocal cords. By this mechanism, the larynx and lower airways are protected from the bolus that is passing through the pharynx and opening of the CPsphincter to the esophagus. Laryngeal adductor muscles, especially the TA muscle, play an important role in the closure of the vocal cord, i.e., glottis closure, and the opening of the CP-sphincter muscles because the primary function of the larynx during swallowing is to close the airway to prevent the entry of food or liquids [74]. Both glottis closure and relaxation and opening of the UES are found to have a close temporal relationship during swallowing [52,65,157]. It has been demonstrated that there is generally an overlap in glottis closure and opening of the CPmuscle [45,157-159]. This temporal relationship can be demonstrated by EMG methods if wire or concentric needle electrodes are inserted into the operating muscles [76,79,96]. The TA-EMG and CP-EMG pause are recorded by needle electrodes using locked delay-line circuitry of the EMG apparatus. The bolus volumes used are saliva and 3, 5, 10, and 15 mL of water in a comparative fashion. This test enables the detection of “Silent Aspiration” in the elderly, including in PD subjects. Figure 17 illustrates that in normal subjects, there are three different patterns for the sequence of TA-excitation and CP-relaxation. In Figure 17A, the delay of the onset of TAEMG; in Figure 17B, the overlapping onset of TAEMG; and in Figure 17C, an earlier onset of TAEMG in relation to the onset of the CP-EMG pause. The first pattern (Figure 17A) is the most risky condition and occurs with a delay of the onset of TAEMG that marks the excitation for closure (between 50–500 msec). This pattern is frequently recorded with saliva or a small volume of swallowed water, whereas the other two patterns are not observed with larger water volumes. This critical time period of 50–500 msec between CP opening followed by delayed activation of TA muscle is the time during which the bolus passes into the pharynx in normal adults. This physiological phenomenon could be a potential risk of aspiration in patients with neurogenic dysphagia, including in PD. Indeed, it has been reported that silent aspirations are volume-dependent, with smaller bolus volumes carrying a higher risk for silent aspiration [22]. Many patients with PD have drooling, including posterior drooling, which may occur in particular during sleep to produce silent aspiration. One mechanism of silent aspiration derives mostly from the sensory-motor abnormalities of the laryngeal structures [160,161].
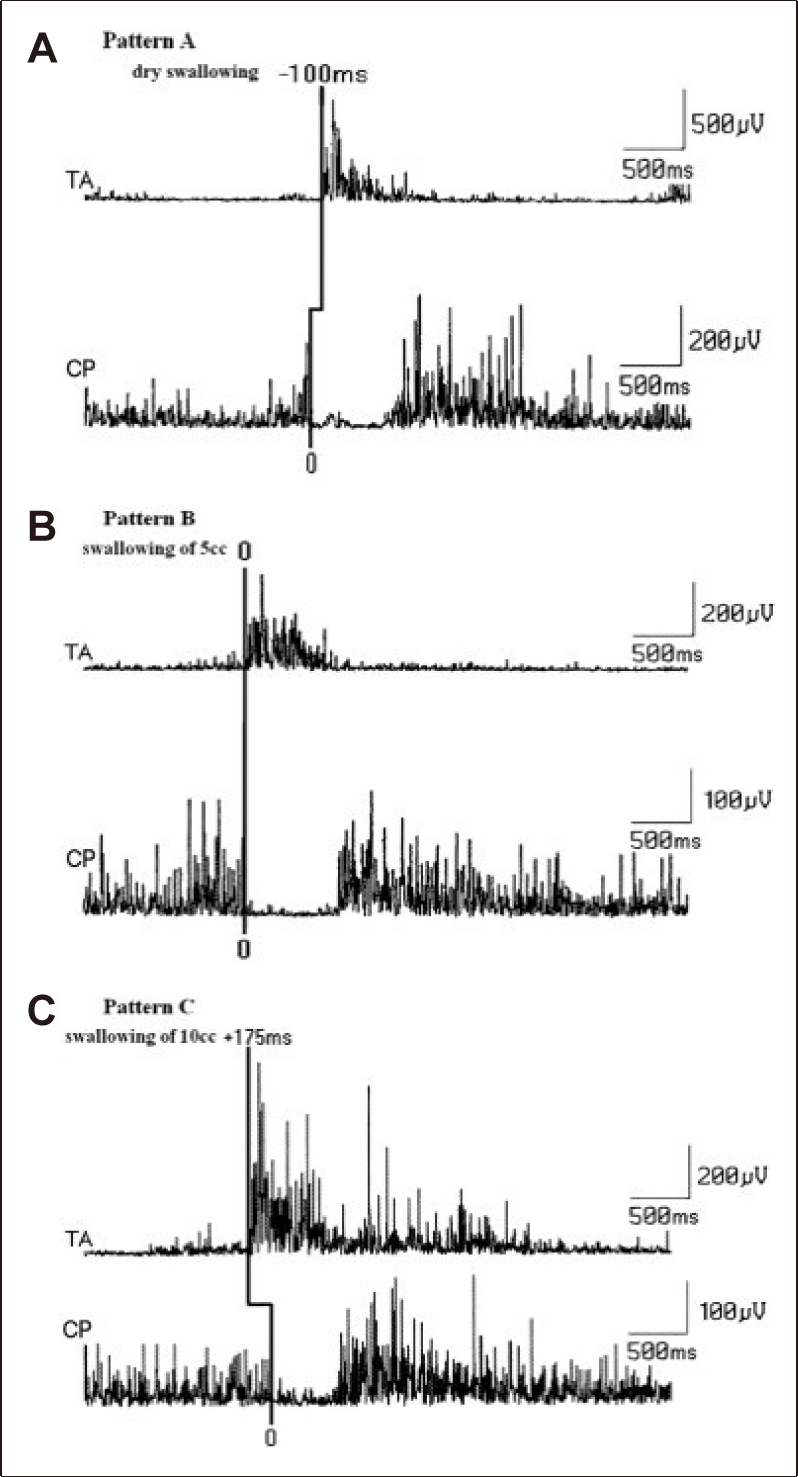
The relationship of laryngeal closure and cricopharyngeal (CP)-sphincter relaxation and opening with increasing water volumes (A, B, and C. See the text for details) [76].
EVALUATION OF SPONTANEOUS SWALLOWING: POLYGRAPHIC/EEG STUDIES
Voluntarily elicited swallowing often occurs with eating and drinking; however, many times during eating, the person is not concentrating on chewing and swallowing despite these functions occurring correctly without a detailed conscious effort. However, SS is composed of periodic swallowing movements spread out over 24 hours [37,106,162-164]. Therefore, the rate of SS is important both physiologically and clinically. During an awake state, healthy subjects swallow saliva frequently at a rate of about once a minute [37,162]. This rate may vary due to different factors, especially those influencing saliva production from the salivary glands around the oral cavity. For example, after a meal or drinking a cup of water, the rate of SS increases [106]. The rate of SS slows down during sleep, during slow-wave sleep [164-166]. In most adult subjects, SS is simultaneously recorded with some EEG arousal [166]. The mean number of swallows per hour during sleep was found to be about three in adult human [166].
The coordination of breathing and swallowing is especially important during sleep. The triggering mechanism is frequently imperfect, and it has been reported that most healthy adults experience nightly aspiration of pharyngeal secretion during sleep [167].
The pacemaker for SS seems to be related to the CPG of the medullary swallowing center [127,134,168]. However, in some situations, alteration of the SSs may be related to some subcortical regions. The automatic movement pattern at the oral cavity for cleaning the accumulated saliva is similar to the spontaneous blinking required for corneal cleaning in humans [106,169-171]. Just as blinking is influenced by the extrapyramidal system, it is likely that SS may also be linked to this system [12,106].
The rate of SS in the elderly is slower than that in young adults [171]. Therefore, in PD, the escape of food materials into the pharynx and saliva accumulation in the oral cavity become risky for airway penetration and aspiration, as is the case in some forms of neurogenic dysphagia.
The rate of SS has been investigated previously using several methods in normal subjects. Accurate measurement of the SS rate requires precise identification of the individual SS against the background activity from the upper aerodigestive tract, as previously reported by Crary et al. [172]. One method includes the use of accelerometry as a measure of structural movement or acoustic recording [173,174]. Another approach is the SM-EMG as a measure of SS activation [106,161,171,173] and respiratory recordings by using nasal airflow or other methods [161,170,173]. The other methods described previously are difficult to apply clinically and are mostly experimental [41,175]. Sleep EEG may be the most appropriate approach to measure the rate of SSs [164-166]. However, sleep EEG recordings to measure the rate of SS are time-consuming and not applicable to every dysphagic patient.
We have developed a technique for continuous evaluation of SS using one-hour polygraphic recordings [171].
POLYGRAPHIC RECORDING OF SS
The subjects lie on a comfortable examination table in the supine position with the head and neck resting at a 30° angle from the table surface. This position prevents dripping of saliva from the mouth. Adult humans are allowed to rest or fall asleep naturally. The room conditions should be calm, quiet and semidark. Any routine EEG apparatus can be used, but we used a twelve-channel EEG device (Nicolet one EEG®, Natus, CA, USA). Five channels are used to record the EMG signals: two channels for lateral/vertical ocular movements, two channels for EEG activity from parieto-occipital electrodes on both sides, one channel with a laryngeal sensor for vertical movements of the larynx during swallowing and the last two channels for electrocardiography and electrodermal activity. In most patients and in normal healthy subjects, a channel is used to record the respiratory signals from a nasal cannula (sleepsense). All of the EMG recordings track signals from the orbicularis oculi (OC), orbicularis oris (OR), SM, masseter (MSS) and anterior tibial muscles.
In the polygraphic recording, the paper speed is 30 mm/sec online. All of the channels are filtered at an approximate frequency response with a high cutoff at 35–1600 Hz, and all the sensitivities are 100 μV/cm. The data are recorded to an external memory, and the values can be changed in the off-line analysis. This enables the amplitude of each channel and the time base of all of the channels to be altered according to the examiner’s requirements. SS data are included according to the following criteria [171]:
1) The OR, MSS, and SM must fire altogether and sequentially in a burst that lasts 1–3 seconds. The SM-EMG is often highest in amplitude during this activity.
2) All three swallowing muscles mentioned above must be recorded in parallel with a synchronous, laryngeal sensor deflection indicating vertical movements of the larynx during swallowing.
3) During recording, one of the investigators ensures the upward and downward movement of the thyroid cartilage and the swallowing movements, preferably using a video-EEG recording.
4) In all three muscles, the response amplitudes for deglutition must be at least four times higher than baseline.
5) Because the nasal airway sensor is used, the swallowing activity should occur during the apnea period of respiration.
Figure 18A demonstrates a spontaneous swallow from a normal subject during the awake stage. OR, MSS, and SM muscles are simultaneously and sequentially recorded with the laryngeal sensor recording. Figure 18B shows a recording from another normal adult subject in whom the swallowing muscles are synchronously recorded within the apnea period of respiration and the laryngeal sensor provides signals during the awake stage. It is interesting to note that the OC are always excited together with the SS. Another important feature of the SS is that there is a tendency to observe double/triple swallows consecutively at intervals of 1–1.5 seconds. Double swallows can be recorded in almost all normal subjects, but their frequency is variable in a given case. Among all of the normal subjects, double swallows could be thought to fire once. Double swallows can appear both during awake and sleep stages, but they are mostly observed during awake stage. Double swallows are found in young and old normal subjects, and to date, they have no pathological meaning but are physiologically interesting.
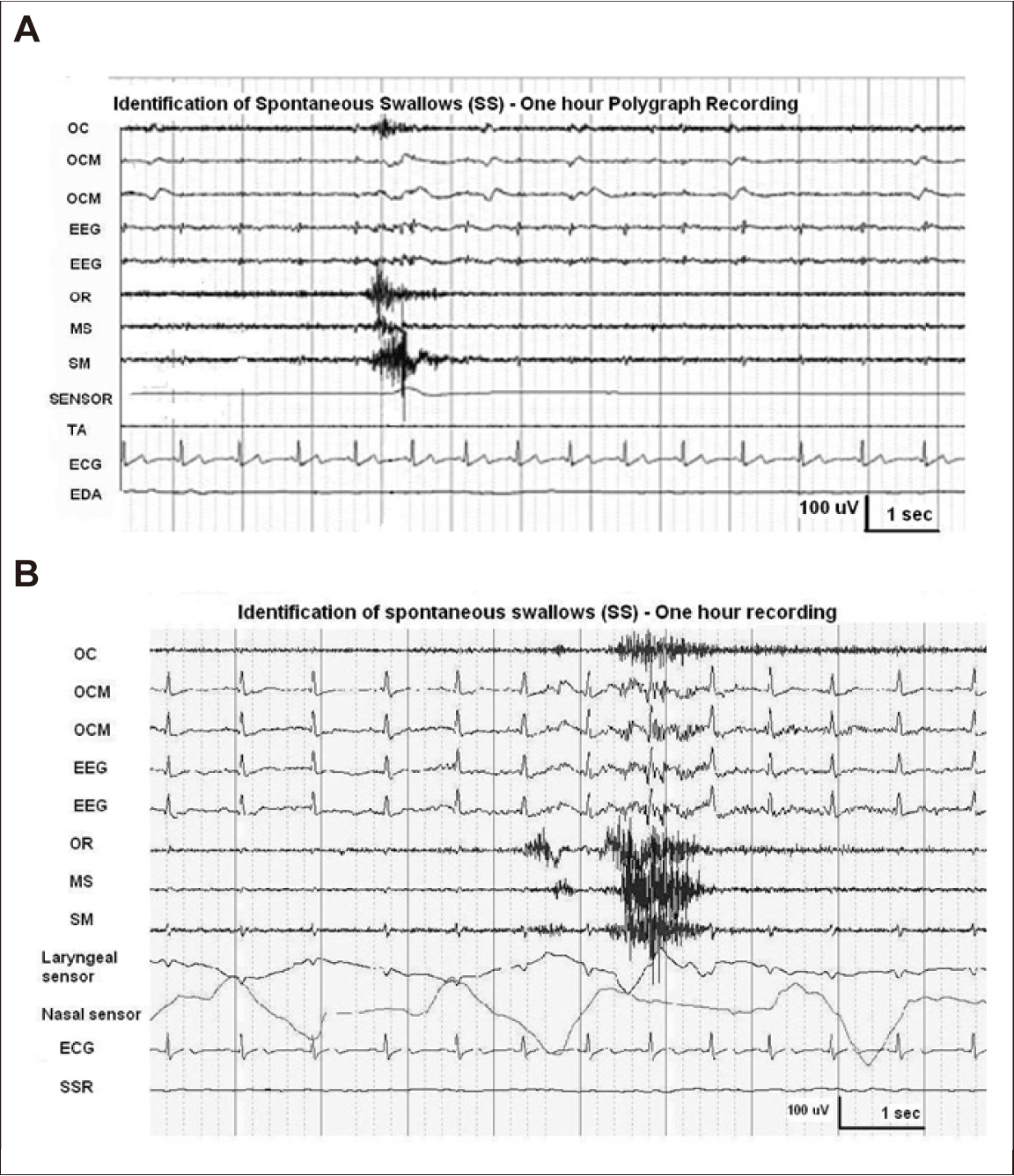
Spontaneous swallowing during a 1-hour polygraphic recording. (A) and (B) are from different normal subjects. A laryngeal sensor recording is clearly seen during spontaneous swallowing in (A). Swallowing apnea, provided by a nasal air sensor (a cannula placed in the nostrils) can be clearly seen during a spontaneous swallow (unpublished).
There are large variations in the SS rate among normal controls, with no significant differences recorded between young and old normal subjects. SS in one-hour recordings do not allow measurement of the rate accurately due to non-rhythmicity. In some normal subjects, the time interval between the two closest SS can exceed 5 minutes.
Spontaneous swallowing may occur at approximately 1 swallow per minute or less in the waking state and 1 swallow per 2 minutes or less during slow wave and REM sleep in normal subjects [171]. The total number of swallows and those in slow wave and REM sleep are significantly reduced in older normal control subjects.
PARKINSON’S DISEASESPONTANEOUS SWALLOWING
The SS rate can be increased in PD patients compared to normal subjects during the awake stage, which may be related to posterior drooling in PD patients who are likely to have increased saliva during wakefulness. In slow-wave sleep in PD, the SS rate is considerably reduced compared to the awake state and is also reduced compared to normal older participants. However, due to the tremendous variability in the SS rate inter-individually both in PD and normal controls, the SS rate is not a clinically sensitive method.
Nevertheless, some qualitative findings can be used for clinical purposes in patients with PD, some of which are clearly demonstrated during one-hour polygraphic recordings. Double swallows can be observed in normal subjects; however, in PD, more than 3 double swallows with an interval of approximately 1 second can be found in 32% of the PD patients investigated. SS numbers can vary between 4 to more than 20 in one triggering set. This type of consecutive swallowing is termed the salvo type of SS (Figure 19). The salvo type of swallowing is mostly but not always recorded in PD patients with drooling. Salvo SS can be recorded in PD patients without dysphagia or sialorrhea, and on one occasion, it was observed in a patient with complaints of dry mouth. Salvo type consecutive swallowing is not related to the head position or to posterior drooling. This is most likely due to a disturbance of the triggering mechanism of pharyngeal swallowing in PD patients. There are two lines of evidence for this pathophysiology based on one-hour studies of SS:

Salvo type of successive spontaneous swallows in Parkinson’s disease. The eight successive spontaneous swallows appear over approximately 17 seconds (see the text for more information) (unpublished).
1) Prior to any salvo-type swallowing, patients may wait several minutes without any SS.
2) Given that salvo SS can occur even in patients without sialorrhea or even in a subject complaining of dry mouth, it is likely that the formation of this type of consecutive swallowing is not related simply to head position or the amount of saliva.
Saliva accumulates in the pharyngeal spaces of PD patients mostly in those with drooling. The saliva accumulation results in anterior drooling during sitting and standing awake position. The saliva, which accumulates in the oral cavity, may flow posteriorly to the pharyngeal spaces. However, in PD, the sensory input from the pharynx may not properly activate the NTS. Indeed, pharyngeal sensory nerves are directly affected by the pathological process in PD patients [104]. Thus, insufficient sensory input in the oropharyngeal mucosae may be the reason for salvo type swallowing of saliva, which accumulates in the pharynx.
Another qualitative finding relates to relationship between cough and SS, which can be investigated objectively. In patients with PD, cough is encountered more commonly than in normal subjects. Under normal conditions, coughing has an obvious function in clearing the material that might otherwise be aspirated into the airway or is already present in the airway [176]. A higher incidence of coughing may indicate that the swallowing material can be transmitted into the airways during deglutition [87,120,127]. Dysphagia plus frequent coughing can occur in all stages of PD [91,120]. However, many individuals with PD can have silent aspirations with little awareness of their dysphagia and little or no cough response to aspiration (Figure 20) [90,91,98].

Spontaneous swallow (SS) and aspiration. In a patient with Parkinson’s disease (PD), during a 1-hour recording, first SS occurs. Five seconds after SS, cough appears, indicating laryngeal penetration. Finally, another SS is recorded (unpublished).
Taken together, we observe that dysphagic PD patients are more prone to saliva aspiration into the airway. We suspect from the polygraphic changes that some SS can produce silent aspiration without cough, although no clear polygraphic pattern can be identified for silent aspiration.
COORDINATION BETWEEN RESPIRATION AND SWALLOWING - EMG EVALUATION OF THE DIAPHRAGM-
The interaction between breathing and swallowing has been well investigated via different methods in various studies [177,178]. Several characteristics of this interaction have been clearly demonstrated as follows:
1) The timing of swallows in relation to the respiratory cycle is not random. Instead, the majority of swallows (approximately ≥ 80%) begin during the expiratory cycle.
2) Swallowing is associated with brief apnea with a duration of 1–2 seconds during discrete/single swallows. The swallowing apnea period becomes longer when SWS is performed.
This coordination between swallowing and breathing is vital for safe passage of the bolus because the route for air and nutrition is partially shared.
The methods used to record the association between respiration and swallowing are less well developed, and the results obtained are open to debate particularly in human studies. The devices employed include respiratory bands, which are nasal or oral masks with airflow or temperature-sensitive transducers that record respiratory activity when the subjects are administered a bolus of liquid or food [178]. Despite extensive studies, methodological difficulties and differences between reports have led to controversies regarding the precise coordination of breathing and swallowing in humans [178-180]. The diaphragm plays an essential role in respiration. Diaphragm movement data provide a more accurate and reliable assessment of post-swallowing expiration and the swallowing apnea period in studies of swallowing and breathing. To obtain diaphragm movement data, diaphragmatic needle EMG is one of the important approaches [179,180]. Figure 21 demonstrates how to record from the diaphragm muscle using the needle approach, and the results obtained from normal volunteers. The subjects remain still during the study with a curved pillow at a slope of 30° and hold this position during swallowing. SM-EMG is recorded using a surface electrode taped under the chin. A laryngeal movement sensor is placed over the region between the cricoid and thyroid cartilages at the midline. Diaphragm EMG activity is recorded with a disposable bipolar concentric needle electrode inserted between the 7th or 8th costae at the left midclavicular line [180].

Electromyogram (EMG) evaluation of the diaphragm for respiration and swallowing. The method used to evaluate diaphragm EMG is shown at the top of the figure. Expiration with swallowing and inspiration with swallowing are shown at the bottom of the figure (see the text for details) [180].
As shown in Figure 21, the recording during a swallow coinciding with expiration is associated with a smaller burst of EMG activity during expiration compared to inspiration (first box). However, the recording during a water swallow coinciding with inspiration demonstrates a prolonged inspiratory diaphragmatic EMG duration (second box). Thus, EMG activities recorded during voluntary swallows that coincide with the different phases of respiration have different features. A new burst of activity is recorded during expiration with swallowing. The duration of the expiratory phase is significantly prolonged compared to quiet respiration when swallowing takes place during expiration. It is possible that inspiration is also prolonged in duration when swallowing takes place during the inspiratory phase of respiration; however, additional EMG activity cannot be clearly differentiated from normal respiratory EMG activity. Despite this, the EMG activity of the diaphragm does not cease with swallowing during the inspiration phase [180]. A consistent increase in diaphragmatic EMG activity is also demonstrated during swallowing apnea [179]. Such diaphragmatic EMG activity and swallowing studies may indicate that medullary central neurons (or interneurons) play dual roles in regulating both breathing and swallowing, as suggested by animal experiments [127,168]. Diaphragmatic activity related to respiration and swallowing should be studied in selected patients with neurogenic dysphagia, particularly PD patients.
In summary, the various EMG methods discussed in this review can be used to diagnose and characterize dysphagia in PD patients, a frequent and yet overlooked topic of great importance. There are methods other than EMG that can be utilized to assess dysphagia in PD; however, they are not discussed herein given the scope of this review.
Notes
Conflicts of Interest
The author has no financial conflicts of interest.